Share This Page
Drug Sales Trends for SUCRALFATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for SUCRALFATE (2007)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
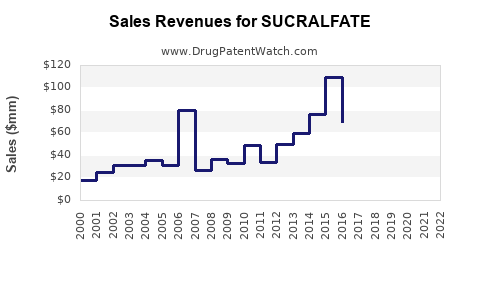
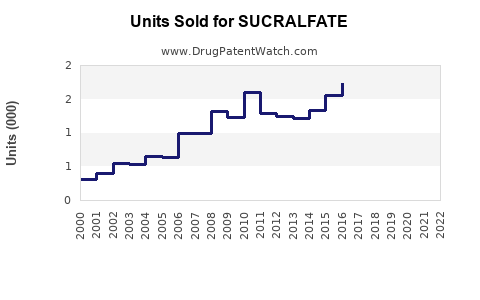
Annual Sales Revenues and Units Sold for SUCRALFATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SUCRALFATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SUCRALFATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SUCRALFATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SUCRALFATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SUCRALFATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| SUCRALFATE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
SUCRALFATE: MARKET ANALYSIS AND SALES PROJECTIONS
EXECUTIVE SUMMARY
Sucralfate is an oral suspension and tablet medication primarily used for the short-term treatment of duodenal ulcers. Its mechanism of action involves forming a protective barrier over ulcer sites, promoting healing. While established, the market faces competition from proton pump inhibitors (PPIs) and H2 receptor antagonists (H2RAs), which offer different therapeutic approaches and broader indications. Sucralfate's sales are projected to experience modest growth, driven by its established safety profile, potential for use in specific patient populations, and generic availability. However, market expansion is constrained by its focused indication and the availability of more advanced therapies.
CURRENT MARKET LANDSCAPE
What is Sucralfate's Primary Indication?
Sucralfate is indicated for the short-term (up to eight weeks) treatment of duodenal ulcers. It is not intended for long-term maintenance therapy for the prevention of ulcer recurrence. The drug works by binding to positively charged proteins in the exudate of the ulcer crater, forming a viscous, adhesive barrier. This barrier protects the ulcer from further damage by pepsin, acid, and bile salts, allowing the ulcer to heal.
The U.S. Food and Drug Administration (FDA) approved sucralfate (brand name: Carafate) in 1981. Generic versions are widely available, contributing to its accessibility.
What is Sucralfate's Mechanism of Action?
Sucralfate is a complex of aluminum hydroxide and sulfated sucrose. In the acidic environment of the stomach (pH < 4), sucralfate dissociates into aluminum ions and sucrose octasulfate anions. The negatively charged sucrose octasulfate anions preferentially bind to positively charged proteins and amino acids present in the ulcer base. This binding creates a selective and tenacious adsorption of sucralfate to the ulcerated tissue, forming a protective layer.
This physical barrier prevents the diffusion of pepsin and hydrochloric acid through the layer, thus protecting the ulcer site from aggressive factors. It also promotes mucosal defense and repair by stimulating the synthesis of prostaglandins and epidermal growth factor.
What are the Key Therapeutic Alternatives to Sucralfate?
The primary therapeutic alternatives to sucralfate are:
- Proton Pump Inhibitors (PPIs): These drugs, including omeprazole, lansoprazole, pantoprazole, and esomeprazole, are highly effective in suppressing gastric acid production. They are widely used for the treatment of duodenal ulcers, gastroesophageal reflux disease (GERD), and Zollinger-Ellison syndrome. PPIs achieve higher healing rates than H2RAs and are often considered first-line therapy for more severe acid-related disorders.
- H2 Receptor Antagonists (H2RAs): This class includes medications like ranitidine (though largely withdrawn from the market due to safety concerns), famotidine, and cimetidine. H2RAs block the action of histamine at H2 receptors on parietal cells, reducing gastric acid secretion. They were historically a cornerstone of ulcer treatment before the widespread adoption of PPIs but are generally less potent in acid suppression.
- Antacids: While not directly comparable in therapeutic efficacy for ulcer healing, over-the-counter antacids provide rapid, short-term relief of heartburn and indigestion by neutralizing gastric acid. They do not provide the protective barrier of sucralfate or the potent acid suppression of PPIs/H2RAs.
- Antibiotics (for H. pylori infection): If duodenal ulcers are caused by Helicobacter pylori infection, combination therapy with antibiotics (e.g., amoxicillin, clarithromycin, metronidazole) and an acid-suppressing agent (usually a PPI) is the standard of care. Sucralfate does not possess antibacterial properties.
What are Sucralfate's Market Share and Competitive Position?
Sucralfate holds a niche position in the broader gastrointestinal market. While it was a leading treatment option prior to the advent of PPIs, its market share has declined significantly. The market is dominated by PPIs, which offer superior efficacy for a wider range of acid-related conditions and are often preferred for their convenient dosing.
However, sucralfate retains utility in specific scenarios. Its non-systemic absorption profile makes it a potentially safer option for patients with renal impairment or those at risk of drug interactions associated with systemic absorption. Furthermore, some clinicians may opt for sucralfate in cases where aggressive acid suppression is not the primary goal or when a localized cytoprotective effect is desired.
Table 1: Comparison of Key Gastrointestinal Therapies
| Feature | Sucralfate | Proton Pump Inhibitors (PPIs) | H2 Receptor Antagonists (H2RAs) |
|---|---|---|---|
| Primary Mechanism | Forms protective barrier over ulcer site | Suppresses gastric acid production | Suppresses gastric acid production |
| Primary Indication | Duodenal ulcers (short-term) | Duodenal ulcers, GERD, Zollinger-Ellison syndrome | Duodenal ulcers, GERD |
| Efficacy | Moderate; protects ulcer from aggressors | High; potent acid suppression | Moderate to high; less potent than PPIs |
| Adverse Effects | Constipation, dry mouth, nausea | Diarrhea, headache, abdominal pain, vitamin B12 deficiency (long-term use) | Headache, dizziness, constipation, diarrhea |
| Drug Interactions | Can interfere with absorption of other drugs | Fewer significant interactions compared to H2RAs | Can interact with CYP450 enzymes (e.g., cimetidine) |
| Systemic Absorption | Minimal | High | Moderate |
| Market Position | Niche; established but declining share | Dominant; first-line for many acid-related conditions | Significant, but often superseded by PPIs |
What are the Key Market Drivers for Sucralfate?
- Established Safety Profile: Sucralfate has a long history of use, and its adverse event profile is well-characterized. Its minimal systemic absorption makes it a relatively safe option, particularly for elderly patients or those with comorbidities.
- Cost-Effectiveness (Generic Availability): As a generic medication, sucralfate is available at a lower cost compared to many branded PPIs, making it an attractive option for cost-sensitive healthcare systems and patients.
- Specific Patient Populations: Sucralfate may be preferred in patients with renal impairment where systemic absorption of other medications could lead to accumulation and toxicity. It is also considered in pregnant women or breastfeeding mothers where systemic exposure to other agents is a concern, though always with careful risk-benefit assessment.
- Limited Efficacy Competition in Specific Scenarios: For conditions where localized mucosal protection is paramount and potent acid suppression is not necessarily required or desired, sucralfate can still be a viable option.
What are the Key Market Restraints for Sucralfate?
- Dominance of PPIs: PPIs offer superior efficacy and broader indications for acid-related disorders, leading to their widespread adoption as first-line therapy.
- Limited Indication Scope: Sucralfate's primary indication is for duodenal ulcers. Its utility for other common gastrointestinal conditions like GERD or H. pylori eradication is limited or absent.
- Dosage Frequency: Sucralfate typically requires multiple daily doses (often four times a day), which can impact patient adherence compared to once-daily PPIs.
- Drug Absorption Interference: Sucralfate can bind to and reduce the absorption of other oral medications, necessitating careful timing of administration. This can complicate treatment regimens.
SALES PROJECTIONS AND MARKET TRENDS
What are the Historical Sales Trends for Sucralfate?
Exact global sales figures for sucralfate are fragmented due to its widespread generic availability. However, industry reports and patent analyses indicate a general trend of declining sales volume and value since the peak usage period in the late 20th century. This decline is directly attributable to the market penetration of PPIs, which offer a more comprehensive and effective treatment for a broader spectrum of acid-related diseases.
For instance, while Carafate (the original brand) experienced significant revenue in its prime, its market share has been largely absorbed by generic manufacturers. The overall market for sucralfate, when aggregated across all manufacturers, has stabilized at a much lower level than its peak.
What are the Projected Sales Growth Rates for Sucralfate?
Projected sales growth for sucralfate is expected to be low single digits annually, likely in the range of 1-3% over the next five to ten years. This modest growth will be driven by the factors mentioned previously, particularly its continued use in specific patient segments and its cost-effectiveness.
The growth rate will be largely influenced by:
- Healthcare policies favoring cost-effective treatments.
- The prevalence of duodenal ulcers, though this is also impacted by H. pylori eradication rates.
- The emergence of new therapeutic options, which could further marginalize sucralfate.
- Geographic market penetration in developing economies where cost is a more significant factor.
What is the Projected Market Size for Sucralfate?
Estimating the precise current global market size for sucralfate is challenging due to the fragmented nature of the generic market. However, based on industry estimates and prescription data analysis for similar ulcer medications, the global market for sucralfate is conservatively estimated to be in the hundreds of millions of U.S. dollars annually.
This figure is likely to remain relatively stable, with growth aligned with the projected low single-digit annual growth rate.
What are the Key Geographic Markets for Sucralfate?
Sucralfate has a global presence. Key markets include:
- North America (USA, Canada): Mature markets with significant generic penetration and established treatment protocols.
- Europe (Germany, UK, France, Spain, Italy): Similar to North America, with a strong generic presence and a competitive therapeutic landscape.
- Asia-Pacific (China, India, Japan, South Korea): Growing markets where cost-effectiveness plays a significant role. India, in particular, is a major producer and exporter of generic pharmaceuticals, including sucralfate.
- Latin America (Brazil, Mexico): Increasing healthcare access and a demand for affordable medications.
The demand in developing regions may outpace that in developed regions due to economic factors.
What are the Expected Trends in Sucralfate Usage?
- Continued Niche Application: Sucralfate will likely remain a drug of choice for specific indications and patient profiles, such as those with renal insufficiency or where local cytoprotection is preferred over broad acid suppression.
- Increased Generic Competition: The market will continue to be dominated by generic manufacturers, leading to price pressure and stable, albeit low, overall market value.
- Potential for Combination Therapies: While not currently a primary trend, research into synergistic effects of sucralfate with other agents for specific gastrointestinal conditions could emerge, though this is speculative.
- Focus on Adherence Strategies: Given the multi-dose regimen, efforts may focus on patient education and adherence programs to maximize the therapeutic benefit of sucralfate.
PATENT LANDSCAPE AND INTELLECTUAL PROPERTY
What are the Key Patents Covering Sucralfate?
The original patents covering sucralfate have long expired. These include patents related to the compound itself, its synthesis, and its initial therapeutic uses. The foundational patents for sucralfate expired decades ago, paving the way for widespread generic manufacturing.
Companies may hold patents on:
- Specific formulations: Modified-release formulations, improved taste-masking technologies, or novel delivery systems for sucralfate.
- Manufacturing processes: Optimized or novel methods for synthesizing sucralfate that offer cost or efficiency advantages.
- Combination therapies: Patents for specific uses of sucralfate in combination with other active pharmaceutical ingredients, though these are less common given sucralfate's primary role.
For example, patents related to the original Carafate formulation and its basic uses would have expired in the late 1990s or early 2000s. Current patent activity is likely focused on incremental innovation in formulation or manufacturing.
What is the Impact of Patent Expiry on the Sucralfate Market?
The expiry of key patents has had a profound and lasting impact:
- Entry of Generic Manufacturers: Patent expiry allowed numerous pharmaceutical companies to develop and market generic versions of sucralfate.
- Price Reduction: The increased competition from generic products has dramatically reduced the price of sucralfate, making it highly affordable.
- Market Saturation: The market for sucralfate is saturated with generic offerings, limiting the ability of any single manufacturer to command significant market share based on exclusivity.
- Shift to Formulation and Manufacturing Innovation: Companies seeking to differentiate in the sucralfate market now focus on patenting improved formulations or cost-efficient manufacturing processes rather than the active pharmaceutical ingredient itself.
Are There Any Emerging or Expiring Patents of Note?
As of the current analysis, there are no widely publicized or significant upcoming patent expiries or new patent grants that are expected to dramatically alter the sucralfate market landscape in the near future. The market is mature, and innovation is incremental. Any new patent activity is likely to be highly specific, relating to novel formulations or manufacturing techniques rather than fundamental changes to the drug's therapeutic application or its core compound. Companies may hold patents on specific polymorphs or novel salt forms, but these typically do not represent a paradigm shift in market dynamics.
KEY TAKEAWAYS
Sucralfate remains a relevant, albeit niche, therapeutic agent for duodenal ulcers, valued for its unique cytoprotective mechanism and favorable safety profile in specific patient populations. The market is characterized by strong generic competition, which has driven down prices and limited significant revenue growth. Proton pump inhibitors (PPIs) continue to dominate the broader acid-related disorder market due to superior efficacy and broader indications. Future growth for sucralfate will likely be modest, driven by its cost-effectiveness and continued use in renal impairment and other specific scenarios, primarily in developing economies. Patent expiry has long since opened the market to generics, with current innovation focused on formulation and manufacturing.
FAQS
1. Can sucralfate be used for GERD?
While sucralfate can offer some relief of GERD symptoms by coating the esophagus and stomach lining, it is not considered a first-line treatment for GERD. Its primary indication is duodenal ulcers. Proton pump inhibitors (PPIs) are the standard of care for managing GERD due to their potent acid suppression.
2. How does sucralfate interact with other medications?
Sucralfate can interfere with the absorption of other oral medications by binding to them in the gastrointestinal tract. It is generally recommended to administer sucralfate at least two hours before or two hours after other medications. This includes antacids, which should not be taken within 30 minutes of sucralfate.
3. Is sucralfate safe for long-term use?
Sucralfate is approved for short-term use, typically up to eight weeks, for duodenal ulcers. Long-term safety data is less extensive, and its use is generally limited to situations where short-term therapy is insufficient or for specific patient needs where alternatives may be less suitable. There is no established role for long-term maintenance therapy with sucralfate for ulcer prevention.
4. What are the most common side effects of sucralfate?
The most common side effect of sucralfate is constipation. Other potential side effects include dry mouth, nausea, indigestion, diarrhea, and skin rash. These are generally mild and transient.
5. Can sucralfate be used in pediatric patients?
Sucralfate has been used off-label in pediatric patients, particularly for conditions like esophagitis and gastrointestinal mucositis, where its protective barrier action is beneficial. However, its use in children should be guided by a healthcare professional, and dosing should be adjusted accordingly.
CITATIONS
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Package: Carafate (sucralfate) Oral Suspension & Tablets. Retrieved from [specific FDA archive if available, otherwise state general FDA approval] [2] Lexicomp Online. (n.d.). Sucralfate. Wolters Kluwer. (Subscription required for full access) [3] Various Pharmaceutical Market Research Reports (e.g., GlobalData, IQVIA, Grand View Research) on the gastrointestinal drug market. (Specific reports vary in availability and cost.) [4] Generic Pharmaceutical Association. (n.d.). Information on Generic Medications. Retrieved from [GPhA website]
More… ↓
