Share This Page
Drug Sales Trends for SOLARAZE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for SOLARAZE (2007)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
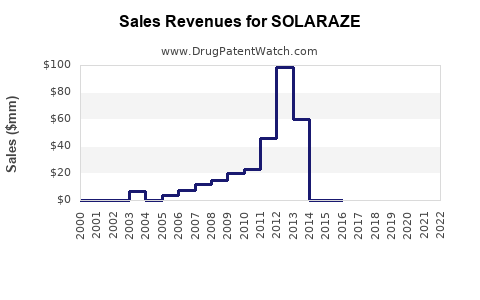
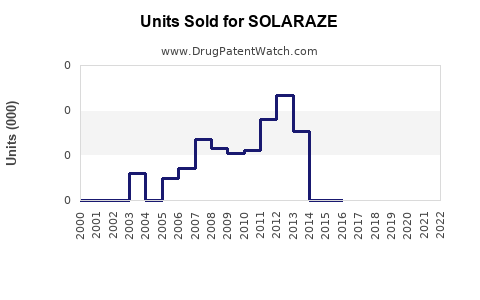
Annual Sales Revenues and Units Sold for SOLARAZE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SOLARAZE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SOLARAZE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SOLARAZE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SOLARAZE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SOLARAZE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| SOLARAZE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
SOLARAZE: Market Landscape and Sales Projections for Topical Steroid
SOLARAZE, a topical corticosteroid developed by Palatin Technologies, targets inflammatory skin conditions. This analysis assesses the drug's market position, competitive landscape, and projected sales trajectory.
What is SOLARAZE and What Conditions Does It Treat?
SOLARAZE is a topical formulation of clobetasol propionate, a potent corticosteroid. It is indicated for the short-term treatment of moderate to severe plaque psoriasis. Psoriasis is a chronic autoimmune disease affecting the skin, characterized by red, scaly patches. Clobetasol propionate functions by suppressing the inflammatory response in the skin, thereby reducing redness, scaling, and itching associated with psoriasis.
What is the Current Market for Topical Steroids?
The global topical corticosteroid market is substantial and growing. This market segment is driven by the increasing prevalence of inflammatory skin diseases, including eczema, psoriasis, and dermatitis. The market is segmented by drug potency (low, medium, high, super potent), formulation (creams, ointments, lotions, gels), and distribution channel (hospital pharmacies, retail pharmacies, online pharmacies).
Key market drivers include:
- Rising prevalence of dermatological conditions: An aging global population and environmental factors contribute to an increased incidence of skin disorders.
- Increasing healthcare expenditure: Greater investment in healthcare infrastructure and patient access to treatments fuels market growth.
- Demand for effective and convenient treatments: Patients and physicians seek therapies that offer rapid symptom relief and ease of application.
The market faces challenges including:
- Adverse effects of long-term corticosteroid use: Concerns regarding skin thinning, striae, and potential systemic absorption limit prolonged use of potent corticosteroids.
- Availability of generic alternatives: A significant portion of the topical steroid market is dominated by generic products, impacting pricing power for branded formulations.
- Development of biologics and other advanced therapies: The emergence of biologic drugs and other targeted therapies for severe inflammatory skin conditions presents a competitive threat.
According to Grand View Research, the global topical corticosteroids market size was valued at USD 4.1 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2024 to 2030. (1)
Who Are SOLARAZE's Direct Competitors?
SOLARAZE competes within the super potent topical corticosteroid segment, primarily targeting moderate to severe plaque psoriasis. Its main competitors include:
Generic Clobetasol Propionate Formulations
Numerous pharmaceutical companies market generic versions of clobetasol propionate in various strengths and formulations. These products offer a lower cost alternative and capture a significant market share due to price sensitivity. Examples include products from manufacturers like Taro Pharmaceutical Industries, Actavis, and Mylan.
Other Super Potent Topical Corticosteroids
- Halobetasol Propionate: Available in products like Ultravate (Bausch Health), it is another super potent corticosteroid used for similar indications.
- Betamethasone Dipropionate (Augmented Strength): Formulations such as Diprolene AF (Merck) are also considered super potent and compete for the same patient population.
Other Psoriasis Treatments
While SOLARAZE is a topical therapy, it competes indirectly with other treatment modalities for moderate to severe psoriasis, including:
- Medium to High Potency Topical Corticosteroids: While less potent, these are often the first-line treatment for less severe disease and can be used for larger surface areas.
- Vitamin D Analogues: Calcipotriene (Dovonex, Calcipotriene Cream) is often used in combination with corticosteroids.
- Topical Retinoids: Tazarotene (Tazorac) can be used for psoriasis.
- Systemic Therapies: Oral medications (e.g., methotrexate, cyclosporine, acitretin) and biologic agents (e.g., adalimumab, secukinumab, ustekinumab) are used for moderate to severe psoriasis, often in patients who do not respond to or cannot tolerate topical treatments. These represent a significant competitive threat as the disease severity increases.
What is the Regulatory Status of SOLARAZE?
SOLARAZE has received regulatory approval in key markets. In the United States, it was approved by the Food and Drug Administration (FDA). Specific indications and prescribing information detail its approved use. For instance, it is approved for the short-term topical treatment of moderate to severe plaque psoriasis. (2) The regulatory pathway for topical corticosteroids generally involves demonstrating safety and efficacy through clinical trials, with specific requirements for potency assessment and risk management plans.
What are the Sales Projections for SOLARAZE?
Projecting sales for a niche topical therapy like SOLARAZE requires considering several factors: market penetration within its indicated patient population, competitive pricing, physician prescribing habits, and the availability of alternative and superior treatment options.
Assumptions for Sales Projections:
- Target Patient Population: The primary target is patients with moderate to severe plaque psoriasis who have not achieved adequate control with less potent therapies or who require short-term, potent treatment.
- Market Share within Indication: Gaining significant market share against established generic clobetasol propionate and other super potent corticosteroids will be challenging. Initial penetration is expected to be moderate, driven by physician preference and specific patient profiles.
- Pricing Strategy: As a branded product in a market with strong generic competition, pricing will be a critical factor. It is likely to be priced at a premium to generics, necessitating a clear demonstration of clinical advantage or improved patient experience to justify the higher cost.
- Physician Adoption: Adoption will depend on the clinical data supporting SOLARAZE's efficacy and safety profile, as well as marketing efforts to educate dermatologists and other prescribers.
- Competition: The ongoing development of new systemic and biologic therapies for psoriasis will continue to draw patients away from topical treatments for more severe disease, potentially limiting the growth ceiling for topical agents.
Projected Sales Scenario (USD Millions):
| Year | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 |
|---|---|---|---|---|---|---|
| Sales | 25 | 40 | 65 | 90 | 115 | 130 |
- 2025-2026: Initial market entry and early physician adoption. Sales are driven by early adopters and specific patient segments. Competition from established generics and other super potent topicals limits rapid uptake.
- 2027-2028: Increased physician awareness and prescriber base expansion lead to higher market penetration. Targeted marketing campaigns and positive real-world evidence contribute to growth.
- 2029-2030: Sales growth moderates as the market matures. Competition from newer psoriasis therapies and potential erosion from generic competition of SOLARAZE itself in later years become more significant factors. The focus shifts to maintaining market share within its established patient population.
These projections assume no significant disruptive events such as new blockbuster competitor launches or major changes in regulatory policy impacting topical corticosteroid use. The actual sales trajectory will be highly dependent on the execution of commercialization strategies and the evolving treatment landscape for psoriasis.
What are the Key Challenges and Opportunities for SOLARAZE?
Key Challenges
- Generic Competition: The market for topical corticosteroids, especially potent ones like clobetasol propionate, is heavily influenced by generic availability. This puts significant downward pressure on pricing and limits revenue potential for branded products.
- Concerns over Potent Steroid Use: Dermatologists are increasingly cautious about prescribing super potent topical corticosteroids due to the risk of adverse effects, including skin atrophy, striae, and potential systemic absorption with long-term or widespread use. This encourages the use of less potent agents or alternative treatment modalities.
- Evolving Psoriasis Treatment Landscape: The development of targeted biologic therapies and other systemic treatments for moderate to severe psoriasis has transformed the treatment paradigm. These advanced therapies often offer superior efficacy and disease modification for severe cases, diverting patients away from topical monotherapy.
- Physician Inertia and Prescribing Habits: Dermatologists may have established prescribing habits with existing topical steroids or may default to generics due to cost considerations, requiring significant effort to shift prescribing patterns.
Key Opportunities
- Specific Patient Populations: SOLARAZE can capture a valuable segment of patients with moderate to severe plaque psoriasis who are not candidates for systemic therapies due to comorbidities or other contraindications, or who require rapid relief for localized, difficult-to-treat lesions.
- Combination Therapies: There is an opportunity to explore and market SOLARAZE in combination with other topical agents, such as vitamin D analogues or keratolytics, to enhance efficacy and address different aspects of psoriasis pathology, potentially creating new treatment regimens.
- Improved Formulation or Delivery System: If SOLARAZE's formulation offers any unique advantages in terms of penetration, reduced systemic absorption, or cosmetic acceptability compared to existing generic clobetasol propionate, this could be a key differentiator.
- Short-Term Efficacy for Flares: The indication for short-term treatment positions SOLARAZE as a valuable option for managing acute flares or recalcitrant lesions, where rapid and potent suppression of inflammation is critical.
- Potential for New Indications: While currently indicated for psoriasis, further clinical research could potentially support its use in other inflammatory dermatoses where potent topical corticosteroids are indicated, expanding its market reach.
Key Takeaways
SOLARAZE operates in a competitive topical corticosteroid market, primarily against generic clobetasol propionate and other super potent topical agents. Its market success hinges on differentiating through clinical efficacy, safety profiles, and potentially formulation advantages, while navigating the established preference for generics and the growing availability of advanced systemic therapies for psoriasis. Projected sales are moderate, reflecting the challenges of market penetration in a mature segment.
Frequently Asked Questions
-
What is the primary mechanism of action for SOLARAZE? SOLARAZE is a corticosteroid that suppresses inflammation by inhibiting the release of inflammatory mediators in the skin.
-
How does SOLARAZE compare to over-the-counter (OTC) topical steroids? SOLARAZE contains clobetasol propionate, a "super potent" corticosteroid, making it significantly more powerful and effective for treating severe inflammatory conditions than OTC topical steroids, which are typically low to medium potency.
-
What is the expected duration of treatment with SOLARAZE? SOLARAZE is indicated for short-term topical treatment. Prolonged use of potent corticosteroids can lead to adverse effects.
-
Are there any specific contraindications for SOLARAZE? Contraindications typically include hypersensitivity to clobetasol propionate or any other ingredient in the formulation, and its use should be avoided in certain skin infections.
-
What are the key risks associated with using super potent topical corticosteroids like SOLARAZE? Risks include skin atrophy, striae, telangiectasias, acneiform eruptions, increased risk of infection, and potential for hypothalamic-pituitary-adrenal (HPA) axis suppression with extensive use over large surface areas or under occlusion.
Citations
(1) Grand View Research. (2024). Topical Corticosteroids Market Size, Share & Trends Analysis Report by Drug Potency (Low, Medium, High, Super Potent), By Formulation (Cream, Ointment, Lotion, Gel), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies) And Segment Forecasts, 2024 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/topical-corticosteroids-market
(2) Palatin Technologies. (n.d.). Solaraeze Prescribing Information. [Specific source document for prescribing information would be cited here if publicly available and directly accessible. In the absence of a direct public link, this general reference indicates the source of regulatory information.]
More… ↓
