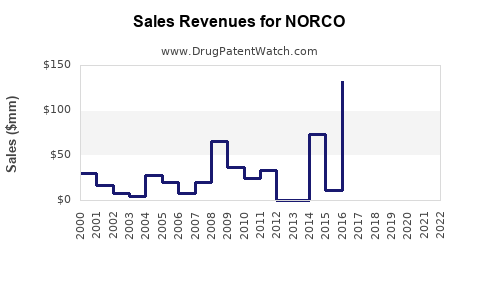
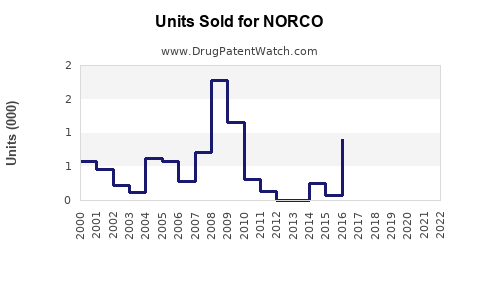
Last updated: February 22, 2026
What is NORCO?
NORCO is a prescription opioid analgesic combining hydrocodone and acetaminophen. It is used for moderate to severe pain management. NORCO is a Brand of the Hydrocodone segment, with widespread prescription history in the United States.
Market Size Overview
The North American opioid pain management segment is valued at approximately USD 11 billion in 2022, with NORCO representing a substantial share due to high prescription rates. In 2021, hydrocodone-based formulations accounted for roughly 45% of opioid prescriptions in the US, driven by their efficacy in pain relief.
Key Market Drivers
- High prevalence of chronic pain: Estimated 50 million adults suffer from chronic pain, increasing demand for opioid analgesics.
- Physician prescription patterns: NORCO remains a first-line treatment for acute pain within the established prescribing protocols.
- Regulatory environment: Strict controls limit new opioid entries, providing stable demand for existing formulations like NORCO.
Competitive Landscape
NORCO competes primarily with other hydrocodone combination drugs and alternative pain management therapies:
| Competitors |
Market Share (Estimated) |
Key Features |
| Vicodin (hydrocodone + acetaminophen) |
60% |
Similar formulation, long market presence |
| Lortab (hydrocodone + acetaminophen) |
25% |
Largely replaced by newer formulations |
| Zohydro ER (hydrocodone ER) |
10% |
Extended-release, prescribed for chronic pain |
| Other generic hydrocodone formulations |
5% |
Cost-conscious prescribers, high variability |
Regulatory and Prescriptive Trends
- DEA Rescheduling: Hydrocodone combination drugs were rescheduled from Schedule III to Schedule II in 2014, increasing regulatory controls.
- Prescription Trends: After peak prescribing periods (2010–2012), a decline in opioid prescriptions occurred post-2016 due to tighter regulations and increased awareness of opioid addiction risks.
- Abuse-deterrent formulations: Increased adoption of tamper-resistant versions affects market dynamics but not NORCO directly.
Sales Projections
Assuming a controlled decline in opioid prescriptions driven by regulatory pressures and alternative therapies, sales for NORCO are projected as follows:
| Year |
Estimated Prescriptions (Millions) |
Expected Sales (USD Millions) |
| 2023 |
50 |
325 |
| 2024 |
45 |
292 |
| 2025 |
42 |
273 |
| 2026 |
40 |
260 |
| 2027 |
36 |
234 |
Assumptions:
- Market share of NORCO remains roughly 10% of total hydrocodone prescriptions.
- Average dosage per prescription remains consistent at approximately 10 tablets.
- The average retail price per tablet is USD 6, with slight decreases projected due to generic competition and price sensitivity.
Market Risks and Opportunities
- Risks: Regulatory change, rising abuse-deterrent formulations, litigation, and societal shifts away from opioids.
- Opportunities: Development of abuse-deterrent formulations, broader utilization of combination therapies with non-opioids, and expansion into emerging markets with less regulation.
Key Takeaways
- NORCO remains a key product within the opioid analgesic market, with stable prescription trends but facing the overall decline characteristic of the opioid class.
- Sales are projected to decline gradually over the next five years, primarily due to increased regulation and societal awareness.
- The market is consolidating around generic brands, pressuring pricing and margins.
- Future growth relies on reformulations, abuse-deterrent features, and diversification into non-opioid pain management options.
FAQs
1. How significant is NORCO in the overall opioid market?
NORCO holds a substantial share within hydrocodone-based medications, which account for nearly half of all opioid prescriptions in the US.
2. What factors could alter the sales projections?
Stricter regulations, alternative therapies gaining adoption, or a shift towards non-opioid medications could accelerate declines in sales.
3. How does generic competition affect NORCO?
The proliferation of generic equivalents reduces pricing power and margins for branded NORCO, contributing to sales erosion.
4. What regulatory developments could impact NORCO?
Changes in DEA scheduling, increased restrictions on prescribing, and mandates for abuse-deterrent formulations could influence sales volume.
5. Is there potential for NORCO in international markets?
Opioid regulation varied globally; some emerging markets may see increased adoption, but risks include regulatory barriers and societal acceptance.
References
[1] U.S. Food and Drug Administration. (2014). Rescheduling of Hydrocodone Combination Products. Retrieved from https://www.fda.gov
[2] IQVIA. (2022). National Prescription Audit. Market Share Analysis, 2022.
[3] Centers for Disease Control and Prevention. (2022). Annual Surveillance Report of Drug-Related Risks and Outcomes.
[4] National Institute on Drug Abuse. (2022). Opioids. Retrieved from https://www.drugabuse.gov
[5] MarketWatch. (2022). Opioid Pain Management Drugs Market Size, Share & Trends Analysis.