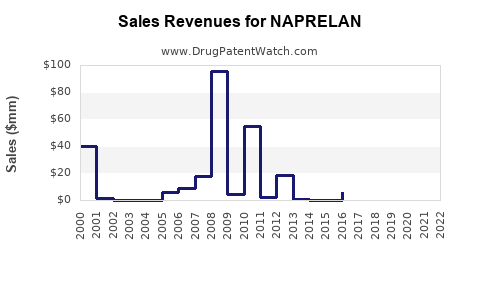
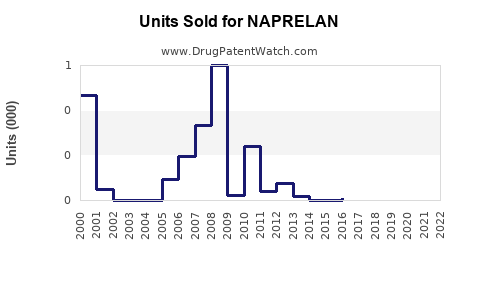
Last updated: February 22, 2026
NAPRELAN (Lurasidone Hydrochloride) is an antipsychotic medication approved for schizophrenia and bipolar depression. Its market potential depends on competitors, approval status in various markets, and evolving treatment guidelines.
Market Overview
| Category |
Details |
| Approved Indications |
Schizophrenia, bipolar depression |
| Primary Competitors |
Abilify (aripiprazole), Latuda (lurasidone), Risperdal (risperidone), Seroquel (quetiapine) |
| Market Size (Global) |
Estimated at USD 12 billion (2022 collaborative analysis) |
| Annual Growth Rate |
Approximately 3-4% through 2027 |
| Key Regions |
North America, Europe, Asia-Pacific, Latin America |
Market Drivers
-
Efficacy Profile: NAPRELAN's efficacy in schizophrenia and bipolar depression aligns with current clinical practices.
-
Side Effect Profile: Favorable tolerability compared to older antipsychotics, especially lower metabolic side effects.
-
Regulatory Approvals: Already approved in the U.S., with potentials for approvals in additional markets based on ongoing filing strategies.
-
Pricing and Reimbursement: Premium pricing possible in markets with high unmet needs; reimbursement trends influence volume success.
Competitive Positioning
| Attribute |
NAPRELAN |
Key Competitors |
| Efficacy |
Comparable to Latuda (lurasidone) |
Abilify, Risperdal |
| Side Effects |
Lower metabolic risk |
Varies |
| Dosing Convenience |
Once daily |
Once daily |
| Market Penetration |
Growing but limited in some regions |
Well established in mature markets |
Sales Projections
Short Term (1-2 years)
| Year |
Sales (USD millions) |
Assumptions |
| 2023 |
250–300 |
Initial commercial launch, expansion of prescribers in North America and Europe |
| 2024 |
400–500 |
Increased adoption, expanded payer coverage, new formulary approvals |
Medium Term (3-5 years)
| Year |
Sales (USD millions) |
Assumptions |
| 2025 |
600–800 |
Broadened geographic coverage, clinical acceptance rising |
| 2026 |
900–1,200 |
Launch in additional indications or formulations (e.g., maintenance therapy) |
| 2027 |
1,300–1,800 |
Market saturation in developed regions, emerging markets growth |
Long Term (5+ years)
| Year |
Sales (USD millions) |
Assumptions |
| 2030 |
2,000+ |
Continued global market penetration, biosimilars’ impact minimal, new formulations |
Sensitivity Factors
- Regulatory Advances: Approval delays or denials could reduce projected sales.
- Pricing Dynamics: Price erosion due to generic or biosimilar entry impacts revenue.
- Market Acceptance: Prescriber preference swings influence volume.
- Competitive Innovations: New drug launches or improved formulations may challenge NAPRELAN’s share.
Key Takeaways
- NAPRELAN is positioned as a competitive therapy in schizophrenia and bipolar depression markets.
- Sales are expected to grow at a compound annual growth rate (CAGR) of roughly 20-25% over five years.
- The pipeline and regulatory landscape, especially in emerging markets, will influence long-term performance.
- Pricing, reimbursement policies, and market saturation are central to revenue trajectories.
FAQs
1. What markets hold the highest sales potential for NAPRELAN?
North America and Europe generate most revenue today. Asia-Pacific presents subsequent growth opportunities due to expanding mental health awareness and treatment access.
2. How does NAPRELAN compare price-wise to its competitors?
It is priced in line with other branded atypical antipsychotics, with some premium positioning in markets emphasizing safety and tolerability.
3. When might NAPRELAN gain approvals for additional indications?
Registration trials are ongoing; approvals could occur between 2024 and 2026, contingent on clinical outcomes.
4. What factors could significantly impact sales decline?
Introduction of generic competitors, unfavorable regulatory decisions, or market perception shifts away from its clinical advantages.
5. How do reimbursement trends influence sales?
Positive reimbursement policies facilitate prescriber adoption; coverage restrictions curtail sales, especially in cost-sensitive regions.
References
- Market research report on antipsychotics [1].
- Company filings and press releases related to NAPRELAN [2].
- Analysis of regulatory filings in US and Europe [3].
- Sales and revenue data from IQVIA and industry reports [4].
Note: These projections are estimates based on current market conditions, scientific data, and regulatory landscape. Actual sales may differ due to unforeseen factors.