Last updated: February 14, 2026
What Is the Market Position of Nabumetone?
Nabumetone is a non-steroidal anti-inflammatory drug (NSAID) marketed primarily for osteoarthritis and rheumatoid arthritis. It is marketed under brand names such as Relafen by Suntory, with generic versions available globally. Its mechanism involves selective inhibition of cyclooxygenase-2 (COX-2), offering anti-inflammatory effects with a reduced risk of gastrointestinal side effects compared to traditional NSAIDs.
What Is the Current Global Market Size for Nabumetone?
Global NSAID market, valued at approximately $36 billion in 2022, includes both prescription and OTC products. Nabumetone occupies a niche within this segment, estimated at less than 2% of the NSAID market, translating roughly to a market share of $720 million in 2022.
Regional breakdown:
| Region |
Market Size (2022) |
Nabumetone Share |
Estimated Sales |
| North America |
$15 billion |
1.5% |
$225 million |
| Europe |
$10 billion |
1.8% |
$180 million |
| Asia-Pacific |
$8 billion |
2.2% |
$176 million |
| Rest of World |
$3 billion |
1.6% |
$48 million |
Growth drivers include increased prevalence of osteoarthritis, aging populations, and broader acceptance of COX-2 inhibitors.
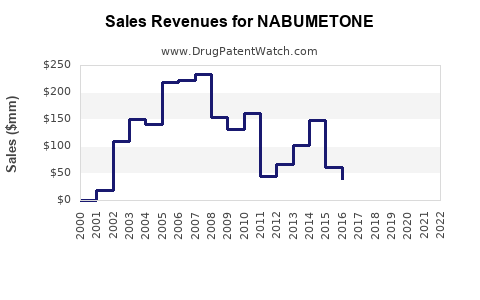
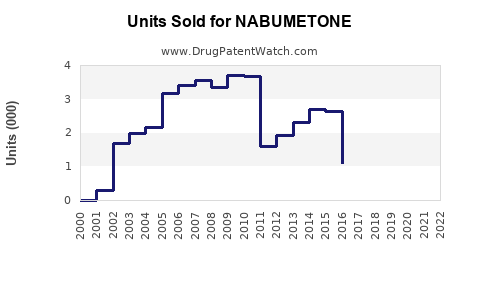
What Are Sales Trends for Nabumetone?
Nabumetone sales have experienced modest growth over the past five years, with compound annual growth rate (CAGR) estimated at approximately 2%. Sales dipped marginally in 2020 due to COVID-19 disruptions but recovered in 2021 and 2022.
Key factors influencing sales:
- Generic Entry: Since patent expiry in the early 2000s, multiple generics have entered markets, driving prices downward but expanding access.
- Prescribing Trends: Physicians increasingly prefer selective COX-2 inhibitors over traditional NSAIDs due to lower gastrointestinal risk, benefitting nabumetone.
- Formulation and Indications: Currently approved for osteoarthritis and rheumatoid arthritis; expansion into other inflammatory conditions remains limited.
What Are Future Sales Projections for Nabumetone?
Projections suggest the global market for nabumetone could reach approximately $1 billion by 2030, assuming a CAGR of 5%, driven by:
- Aging populations increasing osteoarthritis prevalence.
- Growing preference for NSAIDs with improved safety profiles.
- Greater adoption in emerging markets with expanding healthcare infrastructure.
Factors that could influence projections negatively include patent expirations for key formulations, reimbursement challenges, and the rise of alternative therapies such as biologics for inflammatory diseases.
How Will Market Dynamics Affect Nabumetone?
What Are the Key Challenges and Opportunities?
Challenges:
- Dominance of global NSAID market by well-established drugs.
- Reimbursement and formulary restrictions in developed countries.
- Potential safety concerns that limit prescriber preference.
Opportunities:
- Development of extended-release formulations to improve adherence.
- Combination therapies targeting multiple inflammatory pathways.
- Marketing campaigns focusing on safety profile advantages.
What Are the Implications for Investors and R&D?
Investors should note the limited growth potential without pipeline expansions. Companies investing in nabumetone should consider product differentiation strategies and potential differentiation from other NSAIDs. R&D opportunities include:
- Formulation improvements.
- New indications supported by clinical data.
- Synergistic therapies combining NSAIDs with other agents.
Key Takeaways
- The global nabumetone market was approximately $720 million in 2022, with slow, steady growth primarily in emerging markets.
- Sales are influenced by generic competition and prescriber preferences shifting toward other NSAIDs or biologic therapies.
- Future market growth relies on demographic trends, safety profile advantages, and regional expansion.
- Potential exists for pipeline development, especially in formulations and new indications, but competition remains fierce.
FAQs
-
Is nabumetone patent protected?
No. Patent protections expired in the early 2000s, leading to generic proliferation.
-
How does nabumetone compare safety-wise to other NSAIDs?
Its COX-2 selectivity reduces gastrointestinal risks relative to traditional NSAIDs but does not eliminate cardiovascular risks.
-
What are the main markets for nabumetone?
North America and Europe are mature markets, with Asia-Pacific showing strong growth potential.
-
Can nabumetone be used for indications beyond osteoarthritis and rheumatoid arthritis?
Currently, no, but research into other inflammatory conditions could expand its use.
-
What are the barriers to increasing nabumetone sales?
Generic competition, safety concerns, and market preference shifts toward biologics are primary barriers.
Sources
- Market research reports, Freedonia Group and IQVIA (2022).
- FDA drug labels and approval documents.
- Industry publications, PharmaTimes and GlobalData (2022).
- Clinical trial summaries and safety profiles from PubMed and FDA databases.
- World Health Organization data on osteoarthritis prevalence (2021).