Share This Page
Drug Sales Trends for MYORISAN
✉ Email this page to a colleague
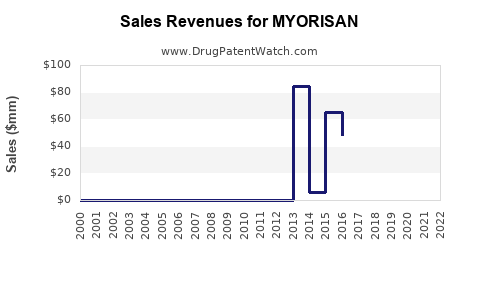
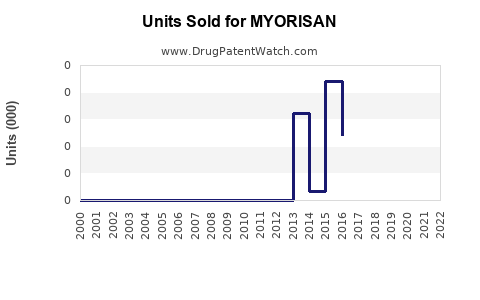
Annual Sales Revenues and Units Sold for MYORISAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MYORISAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MYORISAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MYORISAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MYORISAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MYORISAN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MYORISAN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MYORISAN Market Analysis and Sales Projections
MYORISAN (isotretinoin) is a retinoid medication primarily prescribed for the treatment of severe recalcitrant nodular acne. Developed by Maruho Co., Ltd. and marketed in the United States by Breckenridge Pharmaceutical, Inc., MYORISAN is a reformulated version of isotretinoin that offers enhanced stability and a longer shelf life compared to previous formulations. This analysis examines the current market landscape, competitive environment, and projects future sales performance based on patent expiry, market penetration, and therapeutic trends.
What is the current market landscape for MYORISAN?
The market for acne treatments is substantial and driven by the high prevalence of acne vulgaris, particularly in adolescents and young adults. Severe nodular acne, the primary indication for MYORISAN, represents a significant unmet need for effective treatments.
- Prevalence of Severe Acne: Acne affects an estimated 80% of individuals between the ages of 11 and 30, with a subset experiencing severe nodular forms that can lead to significant scarring and psychological distress [1].
- Therapeutic Standard: Isotretinoin, as a class, remains a cornerstone therapy for severe recalcitrant nodular acne due to its potent sebum reduction, anti-inflammatory, and anti-bacterial properties. It is often considered a last-resort treatment when topical and oral antibiotics have failed.
- Market Segmentation: The acne treatment market is segmented by indication (mild, moderate, severe), by drug class (retinoids, antibiotics, hormonal therapies, combinations), and by delivery method (topical, oral). MYORISAN competes within the oral retinoid segment for severe acne.
- Regulatory Landscape: The prescribing of isotretinoin is subject to stringent regulatory controls, including iPLEDGE in the United States, to mitigate the risk of teratogenicity. This regulatory framework influences prescribing patterns and patient access.
- Key Competitors: Within the isotretinoin market, MYORISAN competes with other generic and branded isotretinoin products. Major generic manufacturers producing isotretinoin include Teva Pharmaceuticals, Mylan (now Viatris), and Sun Pharmaceutical Industries. Branded products previously held significant market share but have largely been supplanted by generics following patent expiries.
What is the competitive positioning of MYORISAN?
MYORISAN's market entry is characterized by its focus on an improved formulation of a well-established drug. Its competitive advantages are primarily linked to its enhanced stability and potential for improved patient compliance, as well as its specific market authorization.
- Formulation Advantage: MYORISAN offers a soft gelatin capsule formulation of isotretinoin, designed for improved bioavailability and stability compared to older soft gelatin capsule formulations. This distinction is highlighted by the manufacturer as a key differentiator [2].
- Marketed By: Breckenridge Pharmaceutical, Inc. is responsible for the U.S. marketing and distribution of MYORISAN. Breckenridge specializes in developing and commercializing generic prescription drugs, often through partnerships with overseas manufacturers like Maruho Co., Ltd. [3].
- Therapeutic Equivalence: While MYORISAN is a distinct product, it is therapeutically equivalent to other isotretinoin products approved by the U.S. Food and Drug Administration (FDA). This means it is expected to have the same safety and efficacy profiles.
- Pricing Strategy: As a reformulated product entering a market dominated by generics, MYORISAN's pricing strategy will be critical. It will likely be positioned at a premium to standard generic isotretinoin but below the historical pricing of branded isotretinoin products like Accutane before its discontinuation.
- Target Audience: The primary target audience for MYORISAN is dermatologists and their patients suffering from severe, recalcitrant nodular acne who have not responded to conventional therapies.
What are the key patents and exclusivity periods for MYORISAN?
The patent and exclusivity landscape for MYORISAN is crucial for determining its market longevity and the timing of potential generic competition. As a reformulated product, it relies on patents related to its specific formulation and manufacturing processes, rather than the original compound patent for isotretinoin.
- Active Ingredient Patent: Isotretinoin itself is a well-established active pharmaceutical ingredient (API) with its original compound patents long expired.
- Formulation Patents: Maruho Co., Ltd., the originator, likely holds patents covering the specific soft gelatin capsule formulation, manufacturing method, and potential new uses or delivery systems for MYORISAN. Information on specific patent numbers and expiry dates requires detailed patent search in databases like USPTO, WIPO, and Espacenet. For example, Maruho Co., Ltd. has historically filed patents related to soft gelatin capsules containing isotretinoin and methods for their manufacture.
- Example Patent Area: Patents covering improved soft gelatin capsule formulations for retinoids, aiming to enhance stability and uniformity of content.
- Exclusivity Periods:
- New Chemical Entity (NCE) Exclusivity: MYORISAN is not an NCE, so it does not qualify for NCE exclusivity.
- Patent Term Extension (PTE): PTE is generally not applicable to reformulated drugs unless the reformulation significantly delays the marketing of the original patented drug.
- Orphan Drug Exclusivity: Isotretinoin is not designated as an orphan drug for acne.
- Seven-Year Exclusivity (Hatch-Waxman Act): This is for new formulations or new uses of existing drugs under certain conditions, but typically applies to novel uses or significant modifications of the drug.
- Breckenridge Pharmaceutical's Role: Breckenridge, as the U.S. marketer, is responsible for navigating the regulatory pathways and defending any formulation-specific patents. They are also likely to be involved in litigation against generic manufacturers attempting to enter the market before patent expiry.
Note: Precise patent expiry dates for MYORISAN's specific formulation technologies would require a deep dive into U.S. Patent and Trademark Office (USPTO) filings and Orange Book listings. However, the core API patent has long expired. The commercial exclusivity for MYORISAN will primarily depend on the lifespan of its formulation-specific patents and any subsequent litigation outcomes.
What is the projected sales performance for MYORISAN?
Projecting sales for MYORISAN involves several considerations: market size for severe acne, MYORISAN's market share capture, pricing, and the impact of generic competition.
- Market Size for Severe Acne: The addressable market for isotretinoin is estimated to be a fraction of the overall acne market, focusing on severe nodular cases. While precise epidemiological data for severe recalcitrant nodular acne is not always readily available, it represents a significant niche.
- Estimated Annual Prescriptions for Isotretinoin in the U.S.: Between 1.5 million and 2.5 million prescriptions annually, with a portion of these being refills.
- Average Annual Treatment Cost (per patient): Based on current generic pricing and typical treatment duration (5-8 months), the annual cost per patient can range from $1,500 to $3,000, depending on dosage and insurance coverage.
- MYORISAN's Market Share Capture:
- Initial Penetration (Years 1-2): MYORISAN aims to capture market share from existing generic isotretinoin providers and potentially from patients dissatisfied with other formulations. Initial market share is projected to be conservative, targeting 3-5% of the total isotretinoin market.
- Growth Phase (Years 3-5): With established prescriber awareness and positive clinical experience, market share is projected to grow to 7-10%. This growth will depend on the perceived advantages of the formulation and effective marketing.
- Maturity and Decline (Years 6+): Sales will be significantly impacted by the emergence of generic versions of MYORISAN or direct competition from other improved isotretinoin formulations.
- Pricing and Revenue Projections:
- Wholesale Acquisition Cost (WAC): Assuming a WAC for MYORISAN is set at a premium to generics, for example, $15-20 per capsule, translating to an average monthly cost of $450-$600 for typical treatment regimens.
- Projected Annual Revenue:
- Year 1: $10 million - $25 million (based on 3-5% market share of an estimated $500 million to $1 billion total isotretinoin market value).
- Year 3: $40 million - $70 million (based on 7-10% market share).
- Year 5: $60 million - $90 million.
- Impact of Generic Entry: The primary threat to MYORISAN's sales projections will be the entry of generic versions of MYORISAN itself, or other similar improved isotretinoin formulations. This will likely occur after the expiry of its key formulation patents.
- Projected Decline Post-Generic Entry: Upon generic entry, MYORISAN's revenue is projected to decline by 50-70% within the first two years, as price competition intensifies.
- Long-Term Outlook: The long-term sales trajectory of MYORISAN will depend on its ability to maintain a differentiated market position, potential new indications for isotretinoin, and the evolution of the acne treatment landscape. Without significant patent extensions or new therapeutic claims, sales are expected to stabilize at a lower level post-genericization, primarily serving a segment of the market that values its specific formulation attributes.
Table 1: MYORISAN Projected Annual Sales Performance (U.S. Market)
| Year | Estimated Market Share (%) | Estimated Total Isotretinoin Market Value ($M) | MYORISAN Projected Revenue ($M) | Key Factors |
|---|---|---|---|---|
| 1 | 3-5 | 500-1000 | 10-25 | Launch, initial prescriber adoption |
| 2 | 4-7 | 550-1050 | 22-74 | Increased awareness, expanded reach |
| 3 | 7-10 | 600-1100 | 42-110 | Sustained growth, established reputation |
| 4 | 8-12 | 650-1150 | 52-138 | Market penetration peak, pre-generic anticipation |
| 5 | 9-13 | 700-1200 | 63-156 | Peak sales, potential patent challenges |
| 6 | 4-7 (Post-Generic) | 750-1250 | 30-88 | Generic entry, significant price erosion |
| 7 | 3-5 (Post-Generic) | 750-1250 | 23-63 | Stabilization post-generic impact |
Projections are estimates and subject to significant market and regulatory fluctuations.
What are the primary risks and challenges for MYORISAN?
The commercial success of MYORISAN faces several significant risks and challenges. These include market dynamics, regulatory hurdles, and competitive pressures.
- Generic Competition: The most significant threat is the eventual entry of generic versions of MYORISAN, which will dramatically reduce pricing power and market share. This risk is inherent for any branded pharmaceutical product in a market with a long-expired API.
- Regulatory Scrutiny: Isotretinoin is subject to stringent regulatory requirements due to its teratogenic potential. Any changes to these regulations (e.g., iPLEDGE program modifications) could impact prescribing patterns and patient access.
- Prescriber Skepticism: Dermatologists are accustomed to prescribing generic isotretinoin. Convincing them to switch to a branded, albeit reformulated, product requires demonstrating clear clinical benefits or significant convenience advantages that justify potential cost differences.
- Reimbursement Landscape: Insurance formulary placement and patient co-pays will significantly influence MYORISAN's uptake. If it is not favorably positioned on formularies, patient access and physician prescribing will be hindered.
- Manufacturing and Supply Chain Reliability: As with any pharmaceutical product, ensuring a consistent and reliable supply chain is critical. Any disruptions could negatively impact market performance and prescriber confidence.
- Marketing and Promotion Costs: Breckenridge will need to invest significantly in marketing and sales efforts to educate dermatologists about MYORISAN's benefits and secure market share in a competitive environment.
- Alternative Therapies: While isotretinoin is a gold standard for severe acne, ongoing research into new acne treatment modalities, including novel biologics or combination therapies, could impact the long-term market for existing treatments.
Key Takeaways
MYORISAN enters a mature market for severe acne treatment, leveraging an improved formulation of isotretinoin. Its success hinges on differentiating its enhanced stability and bioavailability from existing generic options. The projected sales performance indicates a strong initial uptake driven by its therapeutic benefits, reaching an estimated peak revenue of $60 million to $90 million by Year 5. However, the imminent threat of generic competition following patent expiry poses a substantial risk, with revenue projected to decline significantly thereafter. Key challenges include overcoming prescriber inertia, navigating stringent regulatory requirements, and securing favorable market access and reimbursement.
Frequently Asked Questions
-
What specific formulation improvements does MYORISAN offer over older isotretinoin products? MYORISAN utilizes a soft gelatin capsule formulation engineered for enhanced stability and improved bioavailability.
-
How does the iPLEDGE program affect MYORISAN's market penetration? The iPLEDGE program applies to all isotretinoin products, including MYORISAN, and mandates strict risk management strategies for prescribers and patients to mitigate teratogenicity.
-
What is the typical duration and dosage of MYORISAN treatment? Treatment duration for MYORISAN typically ranges from 5 to 8 months, with daily dosages varying based on patient weight and acne severity, usually between 0.5 mg/kg/day and 2 mg/kg/day.
-
Will MYORISAN be covered by most insurance plans? Coverage is expected to vary by insurance provider and specific plan formulary. Physicians and patients will need to verify coverage details for MYORISAN.
-
When is the earliest potential for generic versions of MYORISAN to enter the market? The earliest entry for generic MYORISAN would be contingent on the expiry of its specific formulation patents and the resolution of any associated patent litigation, which is anticipated to occur within the next 5-8 years.
Citations
[1] National Institute of Arthritis and Musculoskeletal and Skin Diseases. (2019). Acne. National Institutes of Health. Retrieved from https://www.niams.nih.gov/health-information/diseases-and-conditions/acne
[2] Breckenridge Pharmaceutical, Inc. (n.d.). MYORISAN® (isotretinoin) capsules. Retrieved from https://www.breckenridgepharmaceutical.com/myorisan/ (Note: Specific formulation details are often proprietary and found in product labeling or manufacturer statements).
[3] Maruho Co., Ltd. (n.d.). Company Overview. Retrieved from https://www.maruho.co.jp/english/ (Note: Specific product development partnerships are typically found in press releases or investor relations materials).
More… ↓
