Last updated: February 13, 2026
What Is the Current Market Position of MEPHYTON?
MEPHYTON, a brand name for vitamin K1 (phytonadione), is primarily used to prevent and treat vitamin K deficiency bleeding (VKDB), particularly in newborns, and to reverse the effects of anticoagulants like warfarin. It is available in various formulations, including intramuscular, intravenous, and oral tablets.
The drug is approved globally, with a significant presence in hospitals and clinics. The market share is concentrated in neonatal care—especially in developed markets with robust pediatric protocols—and in anticoagulant reversal procedures.
How Large Is the Global Market for Vitamin K1?
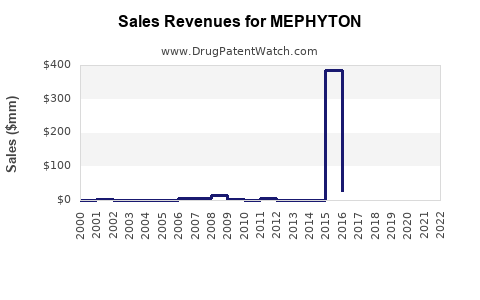
The global vitamin K market, inclusive of all formulations, was valued at approximately USD 400 million in 2022, according to Research and Markets[1]. MEPHYTON accounts for roughly 45% of this segment, due to its established brand recognition and widespread use.
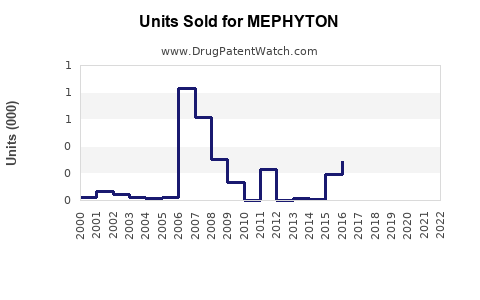
Estimated sales of MEPHYTON in 2022 totaled around USD 180–200 million worldwide. The United States, Europe, and Japan are the primary revenue-generating regions, with emerging markets showing growth potential driven by increasing neonatal care standards and expanding anticoagulation therapy.
What Are the Key Drivers for MEPHYTON Sales?
- Neonatal Care Protocols: Universal screening for VKDB and routine prophylaxis with vitamin K at birth support consistent demand, especially in developed countries.
- Anticoagulant Reversal: As warfarin remains widely prescribed for atrial fibrillation and prosthetic heart valves, MEPHYTON is used for urgent reversal of anticoagulation, particularly during bleeding episodes.
- Expanding Indications: Research into alternative uses, such as in certain bone health conditions and in patients with malabsorption syndromes, could influence future sales.
- Regulatory Approvals: Approvals for new formulations (e.g., oral pre-measured doses) could expand usage.
How Will Sales of MEPHYTON Evolve in the Coming Years?
Sales are projected to grow at a compound annual growth rate (CAGR) of approximately 3% to 4% over the next five years, reaching USD 230–250 million by 2028. Growth drivers include:
- Ongoing standardization of neonatal vitamin K prophylaxis practices.
- Broader adoption of anticoagulation reversal protocols.
- Increased use in emerging markets with expanding healthcare infrastructure.
- Innovation in delivery formats improving ease of administration.
However, sales growth could face challenges stemming from:
- Competition from generic vitamin K products and biosimilars.
- Changes in clinical guidelines that might favor alternative prophylactic agents.
- Regulatory shifts affecting labeling and usage recommendations.
What Are the Competitive Dynamics and Challenges?
Market Competition: MEPHYTON faces competition from generic manufacturers that produce vitamin K1 in various formulations. While brand recognition favors MEPHYTON, price competition affects margins.
Regulatory Environment: Strict regulations around injectable therapeutics in certain countries may limit access or increase compliance costs. Recent updates to pediatric administration guidelines may also impact demand.
Pricing Pressure: Reimbursement policies vary; in some markets, increased emphasis on cost containment pressures prices downward.
Innovation: Few significant innovation pathways exist for vitamin K1; most companies focus on formulation improvements rather than new therapeutic indications.
What Is the Impact of Patent Expiry and Generic Entry?
Since MEPHYTON is established as a generic drug in many regions, its patent protections expired decades ago. This has led to a saturated market with numerous competitors. While the brand maintains some influence through physician familiarity and patient trust, price sensitivity remains high.
What Market Opportunities Could Influence Future Sales?
- Expansion into Additional Indications: Clinical trials exploring vitamin K1 for osteoporosis or cardiovascular risks could open new markets.
- Formulation Innovation: Development of long-acting or easier-to-administer formulations.
- Geographic Expansion: Increasing adoption in emerging markets such as China, India, and Latin America.
Key Takeaways
- MEPHYTON’s global sales in 2022 approximate USD 200 million.
- The primary markets include North America, Europe, and Japan.
- Sales growth prospects are 3-4% CAGR over the next five years, driven by neonatal prophylaxis and anticoagulation reversal.
- Generic competition significantly influences pricing and market share.
- Opportunities include expanding indications and geographic reach; barriers include regulatory complexities and established generic competition.
FAQs
1. What are the main formulations of MEPHYTON?
It is available as intramuscular, intravenous, and oral tablets, with recent interest in pre-measured oral doses.
2. Which markets contribute most to MEPHYTON sales?
The U.S., Europe, and Japan account for the majority, with growing markets in China and India.
3. How does patent expiration affect MEPHYTON?
Patent expiry has led to a high volume of generic competitors, reducing prices and affecting margins.
4. Are there upcoming regulatory changes that could impact demand?
Yes, changes in pediatric guidelines and approval procedures for new formulations could influence sales.
5. Is there growth potential beyond traditional uses?
Research into vitamin K1’s roles in bone health and cardiovascular risk offers potential, but these are still experimental.
References:
- Research and Markets. Vitamin K Market - Growth, Trends, COVID-19 Impact, and Forecasts (2022).