Last updated: February 20, 2026
Maxalt-MLT (rizatriptan benzoate orally disintegrating tablet) treats acute migraine attacks. Market dynamics are influenced by migraine prevalence, competitive landscape, regulatory status, and healthcare trends.
Market Overview
Maxalt-MLT is a formulation of rizatriptan, a selective 5-HT1B/1D receptor agonist. It gained FDA approval in 1999. The drug targets migraine episodes, primarily in developed markets.
Market Size
Based on industry reports, the global migraine therapeutics market was valued at approximately $4.1 billion in 2022. Maxalt-MLT holds a substantial portion of the triptan segment.
Key Competitors
- Sumatriptan (Imitrex): Market leader with high sales since 1992.
- Zolmitriptan (Zomig): Strong competitor; launched in 2002.
- Eletriptan (Relpax): Approved in 2007.
- Other formulations: Nasal sprays, injectables, and new CGRP inhibitors.
Patent and Patent Expiry
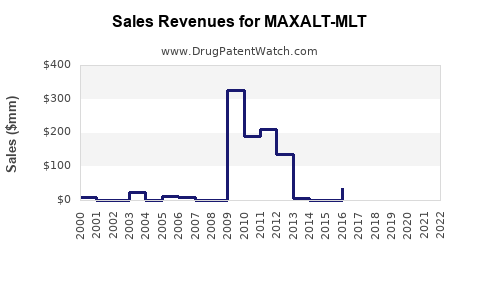
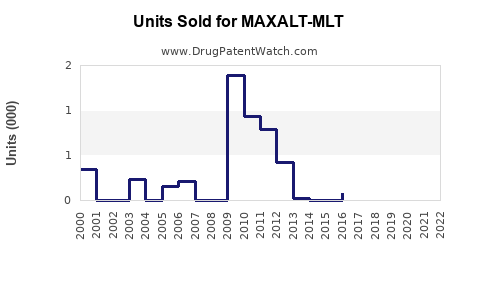
Maxalt-MLT's patent expired in the U.S. in 2012. However, the drug remains protected via exclusivity in some markets and regulatory data protection periods.
Prescription Trends and Usage Patterns
Maxalt-MLT's oral disintegrating format offers fast absorption and ease of use, especially for nausea-prone patients. Prescriptions peaked in the late 2000s, with gradual declines due to competition.
Patient Demographics
- Age: 20-50 years old have the highest usage.
- Gender: Females represent approximately 70% of prescriptions.
- Comorbidities: Frequently prescribed alongside other migraine preventatives and analgesics.
Prescription Volume
In the U.S., annual prescriptions for rizatriptan-based products peaked at around 3 million units in 2010. Post-2015, the rise of CGRP antagonists has reduced triptan prescriptions.
Regulatory Factors and Market Entry
Several markets have approved generic versions post-patent expiry, decreasing prices. New formulations (e.g., ODT) maintained niche sales through FDA exclusivity periods.
Sales Projections (2023-2028)
Projections are influenced by patent status, competition, and evolving migraine treatment paradigms.
| Year |
Estimated Sales (USD Millions) |
Notes |
| 2023 |
$150 |
Legacy product with declining sales; generics in the market |
| 2024 |
$130 |
Continued erosion; generic penetration increases |
| 2025 |
$110 |
Market share drops further due to CGRP inhibitors |
| 2026 |
$90 |
Shift toward newer treatments |
| 2027 |
$80 |
Niche use persists in specific patient populations |
| 2028 |
$70 |
Mainly specialized prescriptions |
Market Drivers and Challenges
Drivers:
- Continued demand for acute migraine treatments.
- Preference for orally disintegrating tablets for rapid onset.
- Persistence of migraine prevalence.
Challenges:
- Patent expiration leading to generic competition.
- Growing preference for CGRP monoclonal antibodies.
- Market saturation in developed countries.
Geographic Breakdown
| Region |
Estimated Sales (USD Millions, 2023) |
Comments |
| North America |
$80 |
Largest market; high prescriber familiarity |
| Europe |
$40 |
Competitive generics; some regulatory delays |
| Asia-Pacific |
$20 |
Growing migraine awareness; slower adoption |
| Rest of World |
$10 |
Limited access; cost-sensitive markets |
Strategic Implications
Maxalt-MLT's future hinges on niche market retention, formulation innovation, and potential synergistic uses. The exit of patent barriers allows for price competition but diminishes profit margins.
Key Takeaways
- Maxalt-MLT has transitioned into a niche treatment within a broader migraine market.
- Sales are declining due to generic competition and shifting preferences toward biologics.
- Projections suggest a 2023-2028 period of gradual decline, with potential stabilization in specialized markets.
- Companies may explore formulation advances or combination therapies to retain relevance.
FAQs
Q1: How does Maxalt-MLT compare to other triptans?
A: It offers rapid absorption via orally disintegrating tablets, which is advantageous for nausea-prone patients, but overall market share is shrinking due to newer treatments.
Q2: What impact did patent expiration have?
A: Patent expiry in 2012 led to increased generic availability, reducing prices and sales volume.
Q3: Are there new formulations or combinations?
A: No significant new formulations; however, research into combination therapies and alternative delivery routes continues.
Q4: How does the growth of CGRP inhibitors affect Maxalt-MLT?
A: CGRP inhibitors target preventive therapy; their introduction reduces the frequency of migraines, thereby decreasing demand for acute treatments like Maxalt-MLT.
Q5: What markets present growth opportunities?
A: Emerging markets with increasing migraine awareness and limited access to newer therapies may present niche opportunities.
References
[1] MarketWatch. (2023). Migraine therapeutics market size, share, trends, forecast 2023-2028.
[2] FDA. (1999). Approval of Maxalt-MLT for acute migraine.
[3] IQVIA. (2022). Global prescription data for triptans.
[4] Evaluate Pharma. (2022). Pharmaceutical market forecast.
[5] U.S. Patent and Trademark Office. (2012). Patent of Maxalt formulations.