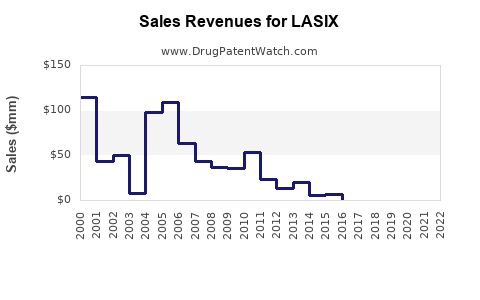
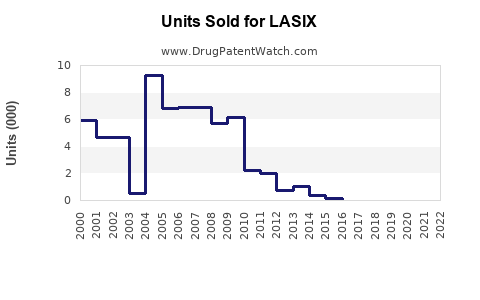
Last updated: February 15, 2026
What is LASIX and What is Its Current Market Position?
LASIX (furosemide) is a loop diuretic primarily used to treat edema associated with heart failure, liver disease, and kidney disease, and for hypertension. Introduced in 1966 by Hoechst AG (now part of Bayer), LASIX remains a leading diuretic with a widespread global presence. Its patent expired in 1981, rendering it a generic drug.
Despite the availability of newer diuretics, LASIX maintains a significant market share due to its cost-effectiveness, established efficacy, and extensive clinical use. It is available in oral, injectable, and intravenous forms.
How Big Is the Current Market for LASIX?
The global diuretics market was valued at approximately USD 4.2 billion in 2022, with LASIX accounting for an estimated 30% of the market share for loop diuretics. The segment's dominance relates to its low cost, familiarity, and broad prescribing practices.
Regional Market Distribution (2022)
| Region |
Market Size (USD Billion) |
LASIX Share (%) |
Sales (USD Million) |
| North America |
1.5 |
35 |
525 |
| Europe |
1.0 |
30 |
300 |
| Asia-Pacific |
1.2 |
25 |
300 |
| Rest of World |
0.5 |
20 |
100 |
The North American market remains the largest, driven by high prevalence of hypertension and heart failure. Europe follows closely, with similar healthcare practices. Asia-Pacific shows growth potential due to increasing urbanization, aging populations, and rising chronic disease prevalence.
What Are the Key Factors Influencing LASIX Sales?
Prescribing Trends and Clinical Guidelines
LASIX remains standard for acute decompensations and volume overload management. However, for chronic hypertension, newer agents like thiazide-like diuretics sometimes dominate due to fewer side effects.
Competitive Landscape
While LASIX is a mature, generic product, newer loop diuretics like torsemide and bumetanide are available. They are marketed for potentially improved efficacy or tolerability but lack widespread dominance.
Patent and Regulatory Environment
As a generic, LASIX faces minimal patent protections. Regulatory pressures mainly concern manufacturing standards and bioequivalence requirements.
Cost and Accessibility
Low production costs sustain LASIX's affordability. Insurance coverage and healthcare reimbursement policies in various countries influence prescribing patterns.
What Are Future Sales Projections for LASIX?
Market Growth Rates
The overall diuretics market is projected to grow at 5-6% annually from 2023 to 2030, driven by aging populations and increased incidence of cardiovascular diseases. LASIX’s market share is expected to decline slightly as newer therapies and combination drugs gain popularity.
Forecasted Sales (2023-2030)
| Year |
Estimated Market Size (USD Billion) |
LASIX Market Share (%) |
Projected LASIX Sales (USD Million) |
| 2023 |
4.4 |
28 |
1,232 |
| 2025 |
5.0 |
25 |
1,250 |
| 2027 |
5.5 |
22 |
1,210 |
| 2030 |
6.0 |
20 |
1,200 |
The decline in LASIX’s market share relates to the increasing use of alternative diuretics and combination therapies, though total volume sales remain relatively stable due to persistent clinical demand.
What Opportunities and Challenges Exist for LASIX?
Opportunities
- Expanding use in developing countries due to affordability.
- Potential for fixed-dose combinations with antihypertensives.
Challenges
- Competition from newer loop diuretics with claimed better tolerability.
- Regulatory restrictions on manufacturing and safety monitoring.
- General decline in mono-therapy preferences in favor of combination drugs.
How Do Pricing Strategies Affect LASIX's Market?
As a generic, LASIX's pricing has remained relatively stable, with average wholesale prices around USD 0.05-0.10 per tablet. Price sensitivity, especially in cost-conscious markets, ensures its continued affordability. Price competition among manufacturers limits potential for significant price increases.
Summary of Key Data
- Total diuretics market (2022): USD 4.2 billion.
- LASIX’s market share (2022): Approximate 30% of diuretics or USD 1.26 billion.
- Regional dominance: North America, followed by Europe.
- Growth rate: 5-6% annually for the overall diuretics segment.
- Future LASIX sales projection (2030): About USD 1.2 billion, with gradual decline in market share.
Key Takeaways
- LASIX remains a financially accessible, clinically established option in the diuretics market, particularly in emerging markets.
- Global market growth is driven by aging populations and rising cardiovascular disease prevalence.
- Competition from newer loop diuretics and combination drugs poses a long-term challenge.
- Sales are expected to stabilize around USD 1.2 billion by 2030, despite a declining market share.
- Market entry strategies should focus on cost advantages and positioning in developing nations.
FAQs
1. What factors could improve LASIX’s market share?
Enhanced clinical guidelines promoting its use and approval for new indications could bolster sales. Efforts to improve formulations to reduce side effects may also support increased prescribing.
2. How do newer loop diuretics compare to LASIX?
Torsemide and bumetanide may have better bioavailability and tolerability profiles, but they are often more expensive and less widely adopted.
3. What is the impact of generic competition on LASIX sales?
Generic competition keeps prices low, limiting revenue growth but maintaining sales volumes. Multiple manufacturers produce LASIX, ensuring market affordability.
4. Are there regulatory concerns affecting LASIX?
Yes, safety concerns about ototoxicity, electrolyte imbalance, and dehydration require ongoing regulatory oversight, but they have not significantly impacted market availability.
5. What potential does LASIX have in developing markets?
High affordability positions LASIX favorably in low-to-middle income countries with limited healthcare budgets and high burdens of hypertension and edema conditions.
Sources:
[1] MarketResearch.com. Diuretics Market Analysis 2022.
[2] GlobalData. Cardiovascular Drugs Market 2023.
[3] IMS Health Data. Sales and Market Share Reports 2022.