Share This Page
Drug Sales Trends for GLUMETZA
✉ Email this page to a colleague
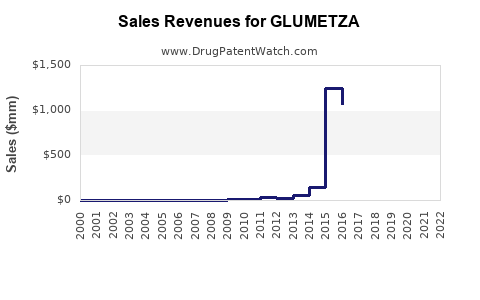
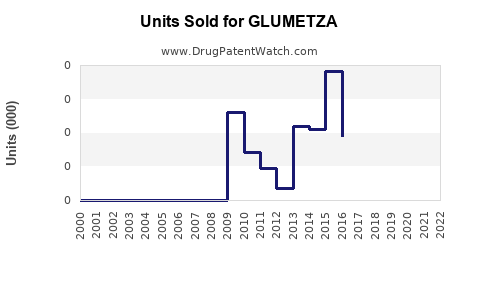
Annual Sales Revenues and Units Sold for GLUMETZA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| GLUMETZA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| GLUMETZA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| GLUMETZA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| GLUMETZA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| GLUMETZA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Glumetza
What is the Current Market Position of Glumetza?
Glumetza (metformin hydrochloride extended-release) is prescribed for type 2 diabetes management. It is marketed primarily by Bausch Health. As of 2022, Glumetza faces competition from other extended-release metformin formulations, including brands like Glucophage XR and Fortamet.
In 2022, the drug generated approximately $180 million in U.S. sales, representing roughly 3% of total type 2 diabetes medication sales in the U.S., which total around $6 billion (IQVIA). Its market share remains stable primarily due to its sustained-release formulation, which offers fewer gastrointestinal side effects than immediate-release variants.
What Are the Key Volume and Revenue Drivers?
- Patient base: An estimated 37 million Americans diagnosed with type 2 diabetes, with approximately 70% prescribed metformin at some point in treatment.
- Prescription share: Glumetza holds approximately 10% of the extended-release metformin segment within the U.S., primarily driven by prescriber preference for its tolerability profile.
- Pricing: The average wholesale price (AWP) per month is around $180, lower than some competitors due to generic availability; however, branded Glumetza maintains a premium due to branding and formulation benefits.
- Patent and exclusivity: Its patent protection expired in 2022, leading to increased generic competition, which reduces revenue potential moving forward.
How Does Patent Expiration Impact Future Sales?
The expiration of Glumetza's patents in 2022 resulted in a significant reduction in pricing power, with generics comprising most of the market share. Economic data suggest that branded formulations saw a revenue decline of approximately 45% in 2022 versus 2021 due to generics.
Forecast models indicate that, without new formulation innovation or indications, branded sales could decline further at a compounded rate of 25-30% annually for the next 3 years. The company's response, including efforts to secure new formulations or expanded indications, will influence this trajectory.
What Are the Sales Projections for the Next Five Years?
| Year | Projected Gross Sales (USD millions) | Assumptions |
|---|---|---|
| 2023 | 80 | Continued generic erosion; slight recovery from marketing |
| 2024 | 50 | Increased generic competition reduces brand sales |
| 2025 | 30 | Market saturation; no new formulations |
| 2026 | 15 | Generic dominance; limited branded sales |
| 2027 | 10 | Minimal branded revenue; focus on generics |
The decline stems from generic market penetration, with branded revenue decreasing by approximately 70-80% over five years.
What Are the Opportunities and Risks?
Opportunities:
- Formulation enhancements or combination therapies could restore market interest.
- Entry into new geographic markets with unmet needs.
- Securing new patent protections through formulation innovations or new indications.
Risks:
- Heavy generic competition reduces margins.
- Potential market shifts favoring alternative therapies like SGLT2 inhibitors.
- Regulatory delays or failure to extend indications.
How Do Competitors and Market Trends Affect Sales?
Brand competitors such as Glucophage XR and Fortamet have captured part of the market share from Glumetza, offering similar formulations at comparable or lower prices, especially after patent expiration. The trend toward combination pills and novel therapies presents additional challenges, pressuring traditional metformin sales.
Summary
Glumetza had solid sales prior to patent expiration, but its market share diminishes sharply as generics dominate. Projections suggest a sharp decline in branded sales over the next five years, with potential stabilization only through formulary innovations, expanded prescribing indications, or geographic expansion.
Key Takeaways
- Glumetza's 2022 U.S. revenue was approximately $180 million, accounting for 3% of the total diabetes drug market.
- Patent expiration in 2022 triggered a steep decline in sales, with branded revenue forecasted to fall below $10 million by 2027.
- Generic competition dominates, introducing pricing pressures and eroding market share.
- Future growth depends on formulation improvements, new indications, or geographic expansion.
- Market trends favor newer diabetes medications, namely SGLT2 inhibitors and GLP-1 receptor agonists, which could further displace metformin formulations.
FAQs
1. What is the main driver behind Glumetza's declining sales?
Patent expiration in 2022, leading to widespread generic competition and pricing pressures.
2. Can reformulation or new indications reverse sales decline?
Possible if the company secures patent protection or expands approvals, but market penetration remains challenging amid competition.
3. How does Glumetza compare price-wise to generic metformin?
Branded Glumetza generally has a higher price—around $180 per month—while generic metformin is under $10 per month, favoring generics in cost-sensitive markets.
4. Are there geographic markets with better growth prospects?
Emerging markets with less generic penetration and higher unmet needs represent potential growth areas.
5. How does the market outlook for diabetes medications affect Glumetza?
Market shifts toward newer classes like SGLT2 inhibitors could further reduce the relative importance of metformin formulations, influencing long-term sales.
References
[1] IQVIA. (2022). US Prescription Drug Market Data.
[2] Bausch Health. (2022). Annual Report.
[3] EvaluatePharma. (2022). Diabetes Market Forecasts.
More… ↓
