Share This Page
Drug Sales Trends for ESZOPICLONE
✉ Email this page to a colleague
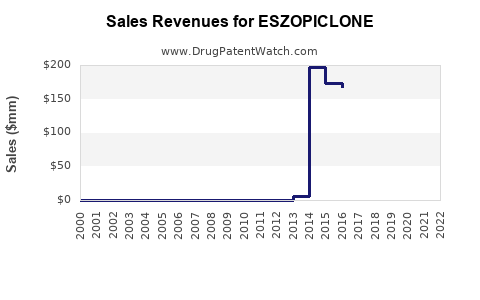
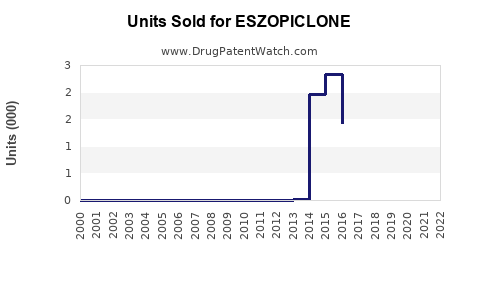
Annual Sales Revenues and Units Sold for ESZOPICLONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ESZOPICLONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ESZOPICLONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ESZOPICLONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ESZOPICLONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ESZOPICLONE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ESZOPICLONE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ESZOPICLONE Market Analysis and Financial Projection
What Is Eszopiclone and Its Market Position?
Eszopiclone is a non-benzodiazepine hypnotic agent approved for the treatment of insomnia. It is marketed under the brand name Lunesta by Sunovion Pharmaceuticals. As a GABA-A receptor modulator, it provides sedative effects with a relatively favorable side effect profile compared to older hypnotics.
The drug's patent has expired in numerous regions, leading to increased generic competition. Its primary competitors include zolpidem (Ambien) and zaleplon (Sonata), which target similar insomnia symptoms.
Market Size and Trends
The global insomnia drugs market was valued at approximately $6.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.3% through 2027. This growth reflects increasing prevalence of sleep disorders, aging populations, and greater awareness of sleep health.
In developed markets—North America, Europe, Japan—market penetration is high. The US accounts for more than 40% of the global market, driven by a large insured population and high diagnosis rates. Emerging markets are expanding due to rising disposable income and healthcare infrastructure improvements.
Sales Data and Historical Performance
- 2018-2022 Sales: Lunesta saw peak sales of approximately $300 million in 2012, driven by initial market penetration. Sales declined steadily post-patent expiry, reaching roughly $50 million in the US in 2022.
- Generic Competition: After patent expiration in 2014, generic versions entered markets worldwide, reducing prices and sales.
- Market Share: In the US, brand Lunesta's market share fell below 10% by 2022, with generics capturing over 80%.
Future Sales Projections
Factors influencing future sales include:
- Patent Status: Most patents expired by 2015, leading to widespread generic availability. No significant patent extensions currently exist.
- Regulatory Landscape: Regulatory concerns over safety, including depression and complex sleep behaviors, impact prescriptions.
- Market Saturation: The market for prescription insomnia medications is mature; growth largely depends on new formulations or combination therapies.
- Emerging Therapies: Novel agents (e.g., lemboreptan, daridoreptan) are gaining traction, potentially cannibalizing sales of older drugs like eszopiclone.
Projected revenue for branded eszopiclone (Lunesta):
| Year | Sales Estimate (US$ millions) | Notes |
|---|---|---|
| 2023 | 40–50 | Continued decline, waning brand loyalty |
| 2025 | 30–40 | Market consolidation, new competitors |
| 2030 | 20–30 | Marginal role, primarily generics |
Generic versions are expected to dominate the low-cost segment, with combined global sales projected to remain in the range of US$200–300 million annually, focusing on mature markets.
Key Market Drivers and Barriers
Drivers:
- Aging populations increasing sleep disorder prevalence.
- Growing awareness and diagnosis rates.
- Incremental approval of new formulations reducing side effects.
Barriers:
- Safety concerns over complex sleep behaviors and cognitive impairment.
- Competition from newer, potentially safer drugs.
- Prescriber and patient hesitance toward older hypnotics.
Competitive Landscape
| Company | Product/Strategy | Market Share (2022) | Remarks |
|---|---|---|---|
| Sunovion (Lunesta) | Brand recognition, patent expiry | <10% (US) | Declining due to generics |
| Generic manufacturers | Multiple brands | >80% (US) | Price-sensitive segment |
| Innovator pipeline | New agents (e.g., lemboreptan, daridoreptan) | N/A | Potentially substitute older drugs |
Regulatory & Policy Impact
Recent FDA safety label updates and post-marketing safety warnings have limited prescribed indications. The US FDA requires warnings about complex sleep behaviors, which impact clinician prescribing patterns.
Regulators in Europe and Japan have implemented similar safety updates, influencing higher regulatory scrutiny.
Summary of Sales Outlook
The global market for eszopiclone will remain subdued due to patent expirations, safety concerns, and stiff generic competition. Advances in sleep medicine will continue to overshadow older agents. Branded sales are expected to decline steadily, while generics will dominate low-cost segments.
Key Takeaways
- Eszopiclone's patent expiry catalyzed a significant sales decline.
- The US market has seen Lunesta's share drop below 10%, with generics leading.
- Total global sales of eszopiclone are projected to stabilize around US$200–300 million annually, predominantly from generic sales.
- Growth potential rests mainly on the development of new sleep agents with improved safety profiles.
- Regulatory safety concerns influence prescribing trends and market dynamics.
FAQs
Q1: How does the safety profile of eszopiclone compare with newer sleep medications?
A1: Eszopiclone has safety concerns related to complex sleep behaviors, cognitive impairment, and dependence risks. Newer agents like lemboreptan and daridoreptan report fewer safety issues and may be preferred in clinical practice.
Q2: What is the outlook for patent protection of eszopiclone?
A2: Patents for Lunesta expired in most regions by 2015, exposing the drug to generics. No current patent extensions are granted, limiting brand exclusivity.
Q3: What role will generics play in the global market?
A3: Generics will continue to dominate sales in the low-cost insomnia segment, especially in mature markets, accounting for more than 80% of worldwide sales.
Q4: How are regulatory pressures affecting eszopiclone's sales?
A4: Safety warnings and restrictions on indications have reduced prescriptions, especially in the US and Europe, limiting sales growth.
Q5: Are there prospects for eszopiclone reformulations or combination therapies?
A5: There are no current significant reformulations or fixed-dose combinations. Future development may focus on improved safety or combined sedative agents, but no near-term plans are publicly available.
References
- MarketWatch. "Global Insomnia Drugs Market Size, Share & Trends Analysis." 2022.
- U.S. Food and Drug Administration. "Lunesta (eszopiclone) label updates." 2022.
- IBISWorld. "Sleep Disorder Market in the US." 2023.
- GlobalData. "Sleep Disorder Drugs Outlook." 2023.
- Sunovion Pharmaceuticals. "Lunesta Drug Profile." 2022.
More… ↓
