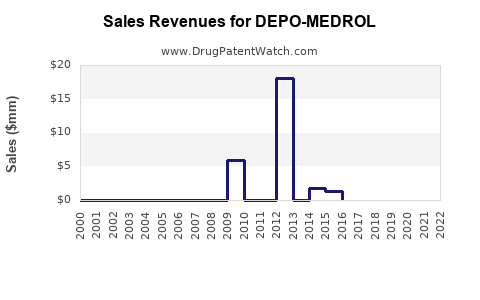
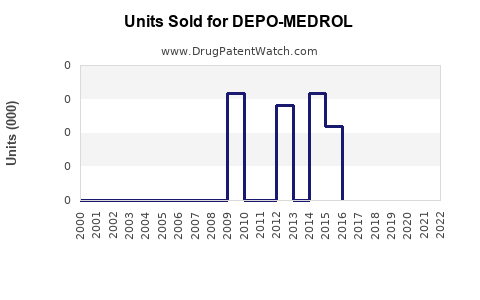
Last updated: February 19, 2026
What is DEPO-MEDROL?
DEPO-MEDROL (methylprednisolone acetate) is a corticosteroid used primarily for anti-inflammatory and immunosuppressive purposes. It is administered via intramuscular, intra-articular, or other injection routes for conditions like arthritis, allergic reactions, and skin diseases.
Market Overview
Current Market Size
The global corticosteroids market, including drugs like DEPO-MEDROL, was valued at approximately USD 12.4 billion in 2021. The segment dedicated to injectable corticosteroids, which includes methylprednisolone formulations, accounts for roughly 35% of this market, translating to USD 4.3 billion.
Key Market Drivers
- Rising prevalence of autoimmune and inflammatory conditions.
- Increasing use in orthopedic and rheumatology treatments.
- Growing adoption in hospital and outpatient settings.
- Patent expirations of competing corticosteroids leading to available biosimilars.
Geographic Distribution
| Region |
Market Share (2021) |
Growth Rate (CAGR 2022–2027) |
| North America |
40% |
4% |
| Europe |
25% |
3.5% |
| Asia-Pacific |
20% |
6% |
| Rest of World |
15% |
3.8% |
North America remains the largest market, driven by high healthcare spending and chronic disease prevalence.
Competitive Landscape
Major competitors include:
- Pfizer: Hydrocortisone and dexamethasone products.
- Teva Pharmaceuticals: Corticosteroid injectables.
- Sun Pharmaceutical: Generic methylprednisolone formulations.
- Mergers and licensing agreements are prominent to expand portfolio.
Sales Projections
Historical Sales Data
From 2018 to 2021, annual sales of methylprednisolone injectable formulations increased at a compound annual growth rate (CAGR) of approximately 3.2%.
| Year |
Sales (USD Billion) |
| 2018 |
3.9 |
| 2019 |
4.0 |
| 2020 |
4.2 |
| 2021 |
4.3 |
Short to Mid-term Projections (2022–2027)
Based on current trends, the corticosteroids segment is expected to grow at a CAGR of 3.5%, with injectable methylprednisolone contributing substantially due to increased adoption.
| Year |
Predicted Sales (USD Billion) |
| 2022 |
4.45 |
| 2023 |
4.60 |
| 2024 |
4.76 |
| 2025 |
4.91 |
| 2026 |
5.07 |
| 2027 |
5.23 |
Growth factors include expanding indications, greater hospital use, and biosimilar entry. Price erosion from generics could temper growth in mature markets.
Factors Influencing Sales
- Patent expirations: Several methylprednisolone formulations faced patent cliffs between 2020–2023, increasing generic competition.
- Regulatory approvals: Pending approvals for new indications or formulations could boost sales.
- Price sensitivity: Price decreases due to generic competition could limit revenue.
Opportunities and Risks
Opportunities
- Development of new formulations or delivery methods such as extended-release versions.
- Expansion into emerging markets with growing healthcare infrastructure.
- Combining corticosteroids with other agents for niche indications.
Risks
- Patent expirations reducing branded sales.
- Regulatory hurdles delaying new formulations.
- Market saturation in developed countries.
- Competition from biosimilars and generics.
Key Takeaways
- DEPO-MEDROL is part of a mature corticosteroid market with evolving dynamics.
- The injectable methylprednisolone segment is projected to grow steadily, driven by demand in inflammation and autoimmune territories.
- Patent expirations have increased generic presence, exerting downward pressure on prices.
- Growth will likely slow as the market matures, with significant gains coming from emerging markets and new formulations.
- Strategic focus on innovation and market expansion needed to sustain or enhance sales.
FAQs
1. What are the main therapeutic uses of DEPO-MEDROL?
Primarily used for inflammatory conditions, autoimmune diseases, allergic reactions, and in orthopedic procedures.
2. How does the patent status impact DEPO-MEDROL sales?
Patent expiration of some formulations has led to increased generic competition, reducing sales of branded versions.
3. Which regions hold the highest sales for methylprednisolone injectables?
North America leads, followed by Europe and Asia-Pacific, driven by healthcare infrastructure and disease prevalence.
4. What are the key factors influencing future sales?
Market saturation, biosimilar competition, regulatory approvals, and emerging markets expansion.
5. Are there new formulations or delivery methods expected for DEPO-MEDROL?
Potential exists for extended-release formulations or combination therapies, which could open new market segments.
References
[1] Markets and Markets. (2022). Corticosteroids Market by Type, Route of Administration, Application, End User – Global Forecast to 2027.
[2] Statista. (2022). Global corticosteroids market size and forecast.
[3] EvaluatePharma. (2022). Top branded corticosteroids and biosimilars sales analysis.
[4] FDA. (2019). Patent expirations and implications for corticosteroids in the U.S.
[5] IMS Health. (2021). Market data for injectable corticosteroids, including methylprednisolone formulations.