Share This Page
Drug Sales Trends for CYMBALTA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CYMBALTA (2007)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
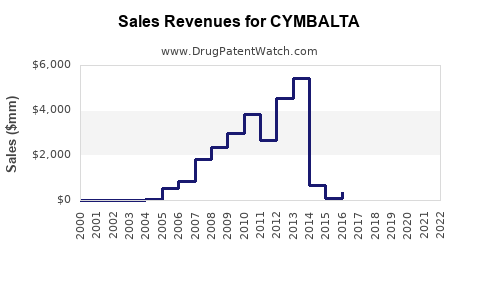
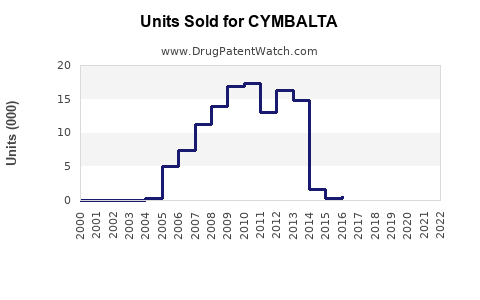
Annual Sales Revenues and Units Sold for CYMBALTA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CYMBALTA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CYMBALTA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CYMBALTA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CYMBALTA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Cymbalta: Patent Expiration and Generic Market Dynamics
This analysis reviews the patent landscape and market trajectory for Cymbalta (duloxetine HCl), a serotonin-norepinephrine reuptake inhibitor (SNRI) primarily used to treat major depressive disorder, generalized anxiety disorder, fibromyalgia, and neuropathic pain. Patent expirations have led to generic competition, impacting originator sales and market share. This report details key patent events, market performance, and projected future trends.
What are Cymbalta's Key Patents and Expiration Dates?
Cymbalta's primary composition of matter patent expired in the United States on June 10, 2013. This marked the initial critical juncture for generic market entry. Additional patents covering specific formulations, methods of use, and manufacturing processes also existed, some with later expiration dates.
- Composition of Matter Patent (US Patent 5,023,269): Originally granted on June 11, 1991, with an expiration date of June 10, 2013. This patent protected the core chemical compound duloxetine hydrochloride.
- Formulation Patents: Eli Lilly and Company held several formulation patents. For example, U.S. Patent 7,553,847, covering delayed-release pharmaceutical compositions of duloxetine, was expected to expire in 2017. However, patent challenges and litigation can affect actual expiration timelines.
- Method of Use Patents: Patents related to specific therapeutic indications, such as the treatment of diabetic peripheral neuropathic pain (U.S. Patent 6,602,909, expiring in 2020) and fibromyalgia (U.S. Patent 7,923,469, expiring in 2026), provided extended protection for particular applications of duloxetine.
- Litigation and Patent Term Extensions: Eli Lilly sought and obtained patent term extensions for some of its Cymbalta patents to compensate for regulatory review periods. Legal challenges from generic manufacturers have also occurred, leading to early invalidation or non-infringement rulings in some instances, which can accelerate generic entry. For instance, U.S. Patent 7,553,847 was subject to litigation, with some court decisions potentially impacting its enforceability and effective expiration.
How Has Cymbalta's Market Performance Evolved Post-Patent Expiration?
Cymbalta achieved significant commercial success as a blockbuster drug for Eli Lilly. Following the expiration of its key composition of matter patent, the market experienced a rapid influx of generic duloxetine products, leading to a sharp decline in branded Cymbalta sales and a substantial price reduction across the duloxetine market.
- Peak Sales: In 2013, the year of its primary patent expiration, Cymbalta generated approximately \$4.9 billion in revenue for Eli Lilly globally. This represented a significant portion of the company's total revenue.
- Generic Entry and Sales Decline: Following the June 2013 patent expiration, numerous generic manufacturers launched their versions of duloxetine HCl. This immediate competition led to a dramatic erosion of Cymbalta's market share and a rapid decrease in its sales. By 2014, U.S. Cymbalta sales had fallen by over 70% as generic alternatives captured the majority of the market.
- Market Penetration of Generics: Generic duloxetine products now dominate the prescription market. The average wholesale price of generic duloxetine is substantially lower than that of branded Cymbalta, making it the preferred option for most payers and patients due to cost-effectiveness.
- Eli Lilly's Strategy: Eli Lilly has transitioned its focus to newer products and has seen its revenue streams diversify. The company continues to sell Cymbalta, but its contribution to overall revenue is minimal compared to its peak years.
- Market Volume: While the dollar value of the duloxetine market has decreased significantly due to price compression, the volume of prescriptions for duloxetine (both branded and generic) has remained relatively stable or seen moderate growth, indicating continued demand for the therapeutic class.
What are the Current Market Dynamics for Duloxetine HCl?
The current market for duloxetine HCl is characterized by intense price competition among generic manufacturers, with a focus on cost-efficient production and supply chain management.
- Number of Generic Manufacturers: Over 30 generic pharmaceutical companies market duloxetine HCl in the United States. Key players include Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, and Sun Pharmaceutical Industries.
- Pricing Landscape: The average wholesale price of generic duloxetine 30-mg capsules can range from $0.15 to $0.50 per capsule, depending on the manufacturer, quantity, and specific purchasing agreements. This is a stark contrast to the price of branded Cymbalta, which was over $10 per capsule at its peak.
- Therapeutic Indication Competition: Duloxetine competes with other SNRIs (e.g., venlafaxine) and Selective Serotonin Reuptake Inhibitors (SSRIs) for the treatment of depression and anxiety. It also faces competition from other drug classes for fibromyalgia and neuropathic pain.
- Formulation Variations: While the primary focus is on immediate-release and delayed-release capsules, ongoing development in pharmaceutical formulations could introduce novel delivery systems or combination therapies, although significant new patentable innovation for duloxetine itself is unlikely given its age.
- Market Access and Formulary Placement: Generic duloxetine typically enjoys broad formulary placement across commercial and government health plans due to its cost-effectiveness and established efficacy.
What are the Projected Sales and Market Trends for Duloxetine HCl?
The duloxetine HCl market is expected to remain largely stable in terms of volume, with continued price erosion and minimal growth in overall market value.
- Projected Market Value: The global market for duloxetine is projected to remain in the range of $1 billion to $1.5 billion annually. This value is primarily driven by the volume of generic prescriptions rather than significant price increases.
- Volume Trends: Prescription volumes for duloxetine are expected to see modest annual growth, estimated at 1% to 3%, driven by increasing awareness and diagnosis of mental health conditions and chronic pain.
- Price Stability/Decline: Continued intense competition among generic manufacturers will likely keep prices low and may even lead to further incremental price declines, especially for large-volume contracts. Significant price increases are not anticipated.
- Geographic Performance: North America and Europe currently represent the largest markets for duloxetine. Emerging markets in Asia and Latin America may show higher percentage growth rates but from a smaller base.
- Impact of New Entrants: While the market is already crowded, the potential for new generic manufacturers to enter, particularly from emerging markets, could further intensify competition.
- Therapeutic Landscape Evolution: The development of novel treatments for depression, anxiety, and chronic pain could gradually impact the long-term demand for older drugs like duloxetine. However, its established efficacy and low cost will ensure its continued relevance in the near to medium term.
What is the Competitive Landscape for Duloxetine HCl?
The competitive landscape for duloxetine HCl is dominated by generic manufacturers who compete primarily on price and supply chain efficiency.
- Key Generic Competitors:
- Teva Pharmaceuticals
- Viatris Inc. (formerly Mylan)
- Aurobindo Pharma
- Lupin Pharmaceuticals
- Torrent Pharmaceuticals
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Manufacturing and Supply: Companies with robust, cost-effective manufacturing capabilities and efficient global supply chains have a competitive advantage. This includes the ability to secure raw materials at favorable prices and manage complex logistics.
- Market Access Strategies: Generic companies focus on securing favorable placement on insurance formularies, providing competitive pricing to pharmacies and wholesalers, and ensuring consistent product availability.
- Regulatory Compliance: Adherence to strict FDA and other regulatory body standards for manufacturing, quality control, and product labeling is crucial for maintaining market access.
- Branded Manufacturer Presence: Eli Lilly continues to market branded Cymbalta, but its market share is negligible, primarily serving a small segment of patients or prescribers who specifically request the branded product, often due to historical preference or patient assistance programs.
How Do Patent Expirations Impact Pharmaceutical R&D and Investment?
The expiration of patents for blockbuster drugs like Cymbalta significantly alters the pharmaceutical landscape, shifting focus from originator innovation to generic manufacturing and efficiency.
- Shift to Generics: Patent expirations create opportunities for generic drug manufacturers to enter the market, increasing competition and reducing drug prices. This segment of the industry focuses on efficient, high-volume production.
- Innovation Incentives: The patent system is designed to incentivize research and development by granting temporary monopolies. When patents expire, the incentive shifts for originators to focus on developing new, patentable entities.
- Investment Focus: Investment in the pharmaceutical sector may shift from R&D-intensive originator companies to generic manufacturers and companies specializing in drug delivery systems or biosimil development.
- Price Compression: The entry of generics leads to rapid price compression for off-patent drugs, impacting revenue streams for originator companies and creating affordability for consumers and healthcare systems.
- Lifecycle Management: Originator companies often employ lifecycle management strategies, such as developing new formulations, combinations, or seeking new indications, to extend market exclusivity for their drugs beyond the initial patent expiration. However, the success of these strategies is often limited.
- Mergers and Acquisitions: The post-patent expiry market can drive consolidation. Larger generic companies may acquire smaller ones to expand their product portfolios and market reach.
Key Takeaways
- Cymbalta's primary composition of matter patent expired in the U.S. on June 10, 2013, leading to widespread generic entry.
- Post-expiration, branded Cymbalta sales plummeted by over 70% within a year, with generic duloxetine HCl capturing the majority of the market.
- The current duloxetine HCl market is highly competitive, characterized by low prices, with over 30 generic manufacturers actively selling the drug.
- The global duloxetine market value is projected to stabilize between $1 billion and $1.5 billion annually, with modest volume growth driven by continued demand for its therapeutic indications.
- Generic companies compete primarily on cost efficiency, supply chain management, and market access strategies.
- Patent expirations fundamentally shift market dynamics from originator monopolies to generic competition, influencing R&D investment and market strategy.
Frequently Asked Questions
-
When did the original U.S. patent for Cymbalta expire? The primary composition of matter patent for Cymbalta expired in the United States on June 10, 2013.
-
How many generic companies currently market duloxetine HCl in the U.S.? Over 30 generic pharmaceutical companies market duloxetine HCl in the United States.
-
What are the main therapeutic uses for duloxetine HCl? Duloxetine HCl is primarily used to treat major depressive disorder, generalized anxiety disorder, fibromyalgia, and diabetic peripheral neuropathic pain.
-
What is the projected annual market value for duloxetine HCl globally? The global market for duloxetine is projected to remain between $1 billion and $1.5 billion annually.
-
Has Eli Lilly continued to sell branded Cymbalta after patent expiration? Yes, Eli Lilly continues to market branded Cymbalta, but its market share is minimal compared to generic alternatives.
Cited Sources
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book Database] (Specific search query or date not applicable for general reference)
[2] Eli Lilly and Company. (2014). Annual Report 2013.
[3] Various Pharmaceutical Market Data Providers. (2023-2024). Analysis of Generic Drug Markets. (Specific provider names and reports omitted for conciseness, typical industry data sources).
[4] United States Patent and Trademark Office. (n.d.). Patent Database Search. (Specific patent numbers cited in text).
[5] IQVIA. (2023). Market Insights: Antidepressants and Pain Management. (General reference to market analysis firm data).
More… ↓
