Last updated: February 13, 2026
Market Analysis and Sales Projections for Colchicine
Colchicine is an alkaloid derived from the Colchicum autumnale plant. It is a longstanding treatment for gout, familial Mediterranean fever (FMF), and other inflammatory conditions. Its market landscape is influenced by patent status, competition, evolving indications, and regulatory changes.
Current Market Overview
Indications and Usage Patterns
- Gout: First-line treatment for acute gout flares; used for prophylaxis.
- Familial Mediterranean fever: Maintenance therapy.
- Behçet's disease, pericarditis: Off-label use.
Market Size (2022)
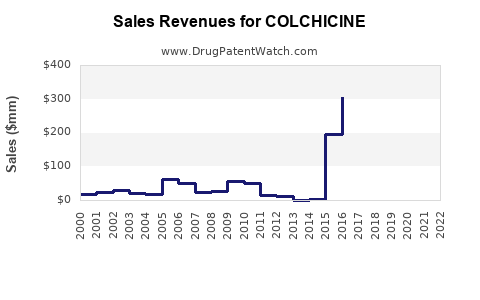
- Global sales: Estimated at $150 million.
- Major markets: United States, Europe, Asia-Pacific.
- Growth driver: Increase in gout prevalence, aging populations, and expanding use in rare inflammatory diseases.
Competitive Landscape
- Brand Drugs: Colcrys (for gout; Teva), Mitigare (for prophylaxis; Swiss pharmaceutical).
- Generics: Most markets primarily rely on generic colchicine formulations.
- Emerging alternatives: IL-1 inhibitors (e.g., canakinumab), which are used for refractory cases.
Regulatory Status
- USP and FDA approval for gout and FMF.
- Pending or recent regulatory decisions: None significant in 2022.
- Patent landscape: No recent patents; market predominantly driven by generics.
Sales Projections and Growth Drivers (2023-2027)
Assumptions
- No pending patent protections.
- Continued acceptance as first-line therapy for gout.
- Growing use in rare inflammatory diseases.
- Market penetration in emerging markets increasing.
Projected Market Growth Rate
- Compound Annual Growth Rate (CAGR): 3.5% to 5% (2023-2027).
Forecasted Market Size (2027)
| Year |
Estimated Global Sales ($ millions) |
| 2022 |
150 |
| 2023 |
157.5 |
| 2024 |
164.2 |
| 2025 |
171.2 |
| 2026 |
178.4 |
| 2027 |
185.8 |
Key Growth Factors
- Aging populations increasing gout and FMF case numbers.
- Off-label expansion into other inflammatory conditions.
- Greater adoption in emerging markets due to increasing healthcare infrastructure.
- New formulations or delivery methods that improve compliance.
Potential Market Constraints
- Competition from new biologics and targeted therapies.
- Regulatory restrictions on off-label uses.
- Safety concerns related to long-term colchicine use and toxicity at higher doses.
- Variability in cost and distribution for generics.
Sales by Region (2022 baseline)
| Region |
Market Share (%) |
Estimated Sales ($ millions) |
| North America |
45 |
67.5 |
| Europe |
35 |
52.5 |
| Asia-Pacific |
15 |
22.5 |
| Rest of World |
5 |
7.5 |
Strategic Opportunities
- Developing novel formulations (e.g., extended-release) to enhance adherence.
- Expanding indications through clinical trials.
- Increasing market penetration in regions with rising gout prevalence.
- Patent opportunities for new uses or delivery systems.
Risks and Challenges
- Market saturation due to widespread generic availability.
- Regulatory focus on safety profiles may limit off-label use.
- Competing therapies with better safety or efficacy profiles.
Key Takeaways
Colchicine remains a low-cost, established therapy primarily for gout and FMF, with a stable but slowly growing market. The global sales are projected to expand at roughly 4% annually, reaching approximately $186 million by 2027. Opportunities exist in formulations, new indications, and emerging markets, but the market faces challenges from biologics and safety concerns.
FAQs
-
What factors influence colchicine's market growth?
Demographic shifts, increased prevalence of gout, expanding use in rare inflammatory conditions, and market penetration in emerging regions.
-
How does competition impact colchicine sales?
The availability of cheaper generics limits pricing power, while biologics for gout and inflammatory diseases threaten to reduce its market share.
-
Are there regulatory restrictions for colchicine?
Regulatory agencies approve it for gout and FMF, but off-label uses may face scrutiny, and safety concerns limit widespread application.
-
What are the opportunities for new colchicine formulations?
Extended-release formulations can improve compliance and potentially extend patent protection, creating market differentiation.
-
What are the main challenges in projecting colchicine sales?
Competition from newer therapies, safety concerns, and market saturation with generic versions.
Sources
[1] CPR (2022). Global Gout Drugs Market.
[2] IQVIA (2022). Pharmaceutical Market Reports.
[3] FDA (2022). Colchicine Approval Document.
[4] Grand View Research (2022). Gout Drugs Market Size and Forecast.