Last updated: February 15, 2026
What Is the Market Size and Current Sales of Acetaminophen (APAP)?
Acetaminophen, also known as paracetamol (APAP), remains one of the most widely used over-the-counter (OTC) analgesic and antipyretic drugs worldwide. In 2022, global sales of APAP products exceeded $10 billion, driven by high demand in North America, Europe, and parts of Asia. North America dominates the market, accounting for approximately 45% of total sales, with the United States leading consumption.
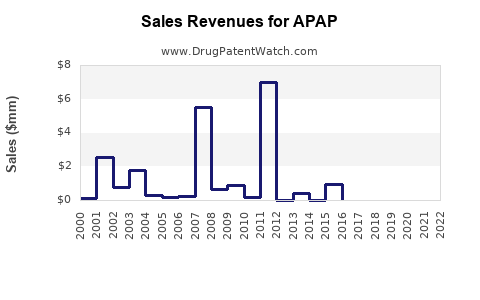
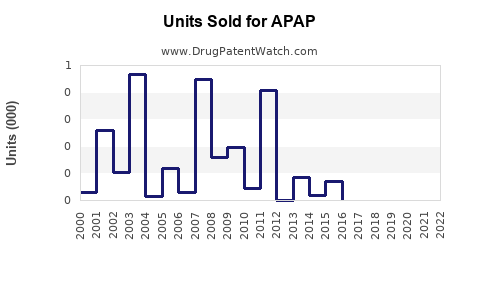
In the U.S., APAP products generate roughly $4.5 billion annually, with an average retail volume growth rate of 3% per year over the past five years. The U.S. market over the last decade reflects stable growth, with annual sales in the range of $4 billion to $4.7 billion, depending on flu season and healthcare trends.
Europe's market registers approximately $2.5 billion, with steady growth linked to OTC availability and increased awareness of overuse risks. Asian markets, particularly China and India, exhibit rapid growth at compounded annual growth rates (CAGR) of 8-10%, driven by expanding healthcare infrastructure and high prevalence of pain and fever management needs.
How Does the Competition Landscape Impact APAP Sales?
Major pharmaceutical firms dominate APAP sales through proprietary brands and generic products. Key players include Johnson & Johnson's Tylenol, Perrigo, and McNeil Consumer Healthcare. Generic versions constitute approximately 75% of market volume, with branded products capturing a premium for marketing and formulation variations.
Store brands from large retailers such as CVS, Walgreens, and Walmart account for nearly 20% of sales by volume, typically priced 10-15% lower than brand-name counterparts.
Despite high sales volume, regulatory concerns regarding APAP overdose, especially related to hepatotoxicity, influence product formulations and labeling. Regulations now emphasize maximum daily dose limits (generally 4 grams per day for adults) and warning labels, impacting marketing strategies.
What Are the Sales Projections Over the Next Five Years?
Forecasts anticipate a compound annual growth rate (CAGR) of 2-4% globally through 2028, reaching approximately $12 billion in annual sales. Key drivers include:
- Expanding middle-class populations in Asia and Latin America increasing demand.
- Growing aging populations worldwide, leading to higher analgesic use.
- Increased awareness of OTC options among consumers.
- Innovation in formulations such as combination products and improved delivery methods.
However, considerations such as regulatory restrictions on dosing, public health campaigns targeting overdose prevention, and the rising popularity of alternative pain relievers (e.g., NSAIDs, opioids with regulated access) could temper growth rates.
How Do Regulatory Policies Affect APAP Market Growth?
Regulations influence both product availability and consumer usage. The U.S. Food and Drug Administration (FDA) has issued guidance limiting single-ingredient APAP in OTC products to prevent overdose. Several countries impose restrictions or require warning labels, leading to product reformulations or reduced sales volumes in some regions.
In the European Union, stricter limits on pack sizes and dosage units aim to minimize overdose risks. These policies can slow growth by limiting product accessibility but also encourage innovation in safer formulations.
What Is the Impact of Safety Concerns and Public Health Campaigns?
Overdose-related hepatotoxicity has prompted public health efforts, including:
- Education campaigns warning against exceeding the maximum daily dose.
- Development of combination products with lower APAP content.
- Improved labeling and dosage instructions.
These measures generally lead to more cautious use but have the potential to slow sales growth if consumers reduce use or switch to alternative therapies.
What Are the Key Trends Shaping the Future Market?
- Product innovation: combination drugs with other analgesics or anti-inflammatory agents.
- Formulation improvements: sustained-release or pediatric-friendly preparations.
- Digital health integrations: apps and reminders to prevent overdose.
- Market entry of biosimilars or new generics: increasing price competition.
- Regulatory tightening: potential for further restrictions in high-risk markets.
Key Takeaways
- Global APAP sales exceed $10 billion annually.
- North America accounts for nearly half of total sales, followed by Europe and Asia.
- CAGR forecast of 2-4% through 2028, reaching around $12 billion.
- Major constraints include regulatory restrictions and safety concerns.
- Innovation and demographic shifts primarily drive future growth.
FAQs
-
What factors are most likely to affect APAP sales in the next five years?
Increased regulation, public health campaigns, demographic aging, and new formulation developments.
-
How do safety concerns impact consumer behavior?
They lead to more cautious use, product reformulation, and increased awareness, which can slow sales growth.
-
Are there significant regional differences in APAP demand?
Yes. North America and Europe have mature markets with stable growth; Asia exhibits high growth potential due to expanding healthcare access.
-
What role do generic manufacturers play?
They dominate volume sales, provide lower-cost options, and influence pricing and competition.
-
Could new formulations disrupt the market?
Potentially, especially those offering improved safety profiles, convenience, or combination therapy benefits.
References
[1] International Data Corporation, "Global Pain Management Market," 2022.
[2] U.S. Food and Drug Administration, "Safety of Acetaminophen," 2021.
[3] European Medicines Agency, "Regulations on OTC Pain Medications," 2022.
[4] IQVIA, "Pharmaceutical Market Analysis," 2022.
[5] Grand View Research, "Analgesics Market Size & Trends," 2022.