Last updated: February 14, 2026
What Is the Current Market for Amphetamine?
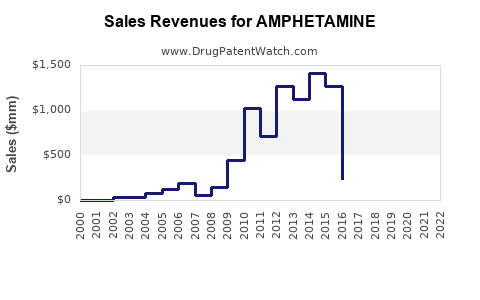
Amphetamine, primarily used for ADHD and narcolepsy treatment, has a substantial global market. The drug is classified as a Schedule II controlled substance in the United States due to its high potential for abuse. The global prescription stimulants market, including amphetamine-based drugs, was valued at approximately $10 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4% to 5% through 2030, driven by increased diagnoses of ADHD, rising awareness, and expanding approval of related medications [1].
How Is the Amphetamine Market Positioned Globally?
North America
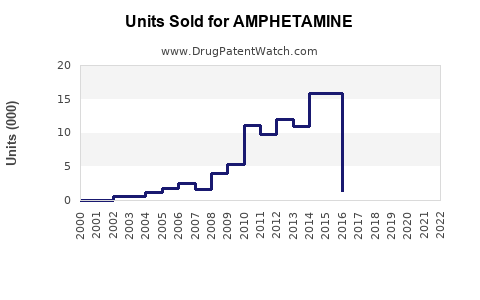
North America, especially the U.S., dominates the market due to high ADHD diagnosis rates. In 2022, estimated prescriptions of amphetamine and related stimulants in the U.S. reached 40 million units, with annual sales exceeding $4 billion. The U.S. accounts for approximately 80% of global sales for prescription stimulants.
Europe
Europe shows steady growth, with an estimated 15% of total prescriptions for stimulants. Countries like the UK, Germany, and France have growing awareness and regulatory approval. The European stimulants market stood at around $2 billion in 2022, with a CAGR of approximately 3% to 4%.
Asia-Pacific
Market expansion is driven by increasing ADHD diagnoses and pharmaceutical regulation reforms. The region's market was valued at around $1 billion in 2022, expected to grow rapidly at 6% to 8% CAGR, upon regulatory approval and increased healthcare infrastructure.
Other Regions
Markets in Latin America, the Middle East, and Africa remain limited due to regulatory restrictions and lower diagnosis rates. However, these regions exhibit potential growth segments over the next decade.
What Are Sales Projections for Amphetamine?
Short-Term (2023-2025)
Sales are expected to increase modestly, with the global market reaching approximately $11 billion by 2025. North America will account for the majority of this growth, driven by ongoing prescription rate increases.
Medium-Term (2026-2030)
Projected global sales could approach $13 billion to $14 billion, with CAGR stabilizing around 4% to 5%. Expansion into emerging markets and potential generic formulations may play roles in growth.
Factors Influencing Sales
- Regulatory changes legalization in some countries that favor medical use.
- Enhanced diagnosis and awareness of ADHD and narcolepsy.
- Development of new formulations and delivery methods to improve compliance.
- Concerns over abuse potential and resulting restrictions could suppress growth.
What Does Competition Look Like?
Amphetamine faces competition from other stimulant drugs, including methylphenidate (e.g., Ritalin), mixed amphetamine salts (Adderall), and non-stimulant medications like atomoxetine. Patent expirations and generic competition are increasing availability and lowering prices.
Key Players
- Teva Pharmaceuticals: Major producer of generic amphetamine products.
- Eli Lilly, Shire (now part of Takeda): Marketed branded stimulants.
- Adequate regulatory frameworks: Can limit or promote entry of new formulations or synthetic alternatives.
What Regulatory and Ethical Considerations Affect Sales?
Amphetamine's controlled status affects prescribing practices and distribution. Regulations aimed at curbing abuse, including prescription monitoring programs and tighter prescribing limits, can impede rapid market growth.
Emerging policies in various countries aim to balance access for legitimate medical uses with measures to prevent misuse. Legalizing or relaxing restrictions in specific regions could expand access, whereas stricter controls will restrict sales.
Key Market Drivers and Challenges
| Drivers |
Challenges |
| Increased recognition of ADHD |
Stringent regulations to prevent misuse |
| Growing global diagnostic rates |
Negative perceptions linked to abuse |
| Development of extended-release formulations |
Competition from non-stimulants |
| Microdosing and alternative delivery methods |
Patent expirations leading to generic growth |
Final Outlook
The amphetamine market remains robust within regulated medical settings. Sustained growth depends on regulatory environments, addressing abuse concerns, and developing novel formulations. Market entrants or investors should monitor policy shifts, emerging competition, and advances in neuropharmacology.
Key Takeaways
- The global prescription amphetamine market was valued at approximately $10 billion in 2022.
- North America accounts for around 80% of sales, primarily driven by high ADHD diagnosis rates.
- Market growth is projected at 4% to 5% CAGR through 2030.
- Emerging markets and new formulations could expand the market but face regulatory risks.
- Competition from generics and alternative treatments is intensifying.
FAQs
1. How do regulatory restrictions impact amphetamine sales?
Stringent controls on prescribing and distribution reduce availability, slow market expansion, and increase compliance costs for manufacturers.
2. What is the potential for generic amphetamine products?
Patent expirations have led to an increase in generic options, reducing prices and expanding access in mature markets like the U.S. and Europe.
3. Are non-stimulant alternatives affecting amphetamine sales?
Yes. Medications such as atomoxetine and guanfacine offer alternatives with lower abuse potential, slightly restraining stimulant market growth.
4. What are the main health concerns associated with amphetamine?
Risks include dependency, cardiovascular issues, and abuse potential, prompting regulatory restrictions and influencing prescriber habits.
5. How might future regulations shape the market?
Potential policy shifts favoring access for medical use or tighter controls to prevent misuse will directly influence sales and market growth.
Sources
[1] MarketsandMarkets, "Prescription Stimulants Market," 2022.