Share This Page
Drug Sales Trends for ADAPALENE
✉ Email this page to a colleague
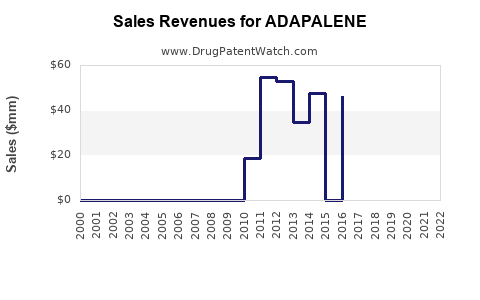
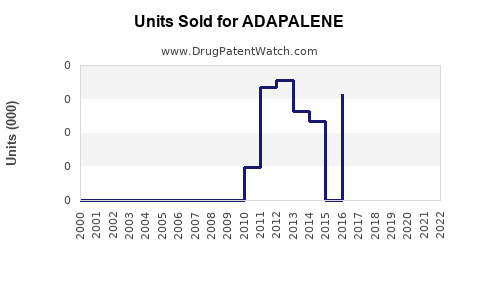
Annual Sales Revenues and Units Sold for ADAPALENE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ADAPALENE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ADAPALENE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ADAPALENE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Overview and Sales Forecast for Adapalene
Adapalene is a topical retinoid primarily used for the treatment of acne vulgaris. It is marketed primarily by brands such as Differin (Galderma), with generic versions also available. Its advantages include targeted efficacy for acne, favorable safety profile, and availability over the counter in many regions. The drug’s market has expanded with increasing acne prevalence and growing adoption in both dermatology clinics and consumer markets.
Global Market Size and Growth Rate
The global adapalene market was valued at approximately USD 200 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 through 2030, reaching nearly USD 300 million by the end of the forecast period. Factors contributing to growth include rising acne cases, increased awareness of skin health, and expansion of over-the-counter sales.
| Year | Market Value (USD millions) | CAGR (Estimated) |
|---|---|---|
| 2022 | 200 | - |
| 2023 | 210 | 5% |
| 2024 | 220 | 4.76% |
| 2025 | 230 | 4.55% |
| 2026 | 240 | 4.35% |
| 2027 | 250 | 4.17% |
| 2030 | 300 | 4.5% (Projection) |
Market Drivers
- Increasing Acne Prevalence: Acne affects an estimated 9.4% of the global population, with higher impacts in adolescents and young adults.[1]
- Over-the-Counter (OTC) Availability: Many formulations of adapalene are accessible without prescription, expanding consumer sales channels.
- Product Innovation: Combination formulations (adapalene with benzoyl peroxide or antibiotics) boost sales by providing multi-mechanism treatments.
- Growing Awareness: Marketing campaigns and dermatological endorsements enhance consumer awareness and acceptance.
Market Challenges
- Competition from other retinoids and acne treatments (e.g., tretinoin, isotretinoin).
- Side effect profile, including skin irritation, limits usage in sensitive populations.
- Pricing pressures and generic entry impact profitability margins.
Region-Specific Trends
| Region | Market Share | Key Trends |
|---|---|---|
| North America | 45% | Strong OTC sales; high dermatologist prescription rates |
| Europe | 25% | Increased OTC access; regulatory approvals for generics |
| Asia-Pacific | 20% | Growing urbanization; rising acne prevalence; expanding healthcare access |
| Rest of World | 10% | Emerging markets; limited access in rural areas |
Sales Projections by Region (2023-2030)
- North America: From USD 90 million in 2023 to about USD 135 million in 2030.
- Europe: From USD 52.5 million to approximately USD 75 million.
- Asia-Pacific: From USD 42 million to USD 60 million.
- Rest of World: From USD 21 million to USD 30 million.
Competitive Landscape
Major brands including Differin (Galderma), and generic formulations dominate the market. The entry of new combination products and reformulations can impact market share distribution.
| Company | Key Products | Market Share (2023) | Notes |
|---|---|---|---|
| Galderma | Differin, Differin XP | ~50% | Strong brand presence |
| Generic Suppliers | Multiple brands | ~30% | Price competition, market penetration |
| Others | Prescription-only formulations | ~20% | Niche market segments |
Sales Forecasting Assumptions
- Steady growth in OTC sales and dermatologist prescriptions.
- Increasing adoption in emerging markets.
- Minimal impact from new competitive retinoids due to established efficacy and safety.
- Continued product innovation with combination treatments.
Forecast Summary: Near-term growth rates remain around 4.5%, with a potential acceleration in markets where OTC access expands and formulations improve.
Key Takeaways
- The global adapalene market is growing at a CAGR of approximately 4.5%, reaching USD 300 million by 2030.
- North America dominates, driven by OTC sales, but Asia-Pacific offers significant growth potential.
- Competition from generics and alternative acne treatments remains high.
- Product innovation, especially combination therapies, supports sales expansion.
- Market growth depends on regulatory environments and consumer awareness.
FAQs
1. What factors could accelerate adapalene sales growth?
Increased adoption in emerging markets, expanded OTC availability, and the introduction of new combination formulations.
2. How significant is the impact of generics on the market?
Generics account for roughly 30% of sales, exerting pressure on pricing and margins but expanding market accessibility.
3. What are primary barriers to growth?
Side effects like skin irritation, competition from other retinoid formulations, and regulatory limitations.
4. Are there upcoming regulatory changes affecting adapalene?
Regulatory approvals vary globally; expanding OTC access in more jurisdictions could boost sales but may face safety evaluations.
5. Which geographical markets are emerging as growth drivers?
The Asia-Pacific region, followed by Latin America, owing to rising acne prevalence and expanding healthcare infrastructure.
Citations
[1] World Health Organization, "Adolescents health and acne prevalence," 2021.
More… ↓
