Last updated: February 12, 2026
Market Overview and Sales Projections for ABILIFY
ABILIFY (aripiprazole) is an antipsychotic medication developed by Otsuka Pharmaceutical and marketed in collaboration with Bristol-Myers Squibb (later for certain markets by other firms). It is approved for schizophrenia, bipolar disorder, depression adjunct, irritability associated with autism, and Tourette’s disorder. The drug’s market performance is driven by its approval status, competitive landscape, and evolving healthcare policies.
Market Size and Current Sales
Global Sales (2022):
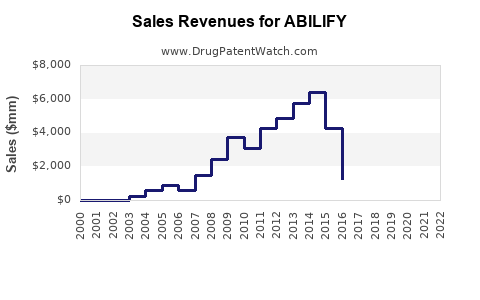
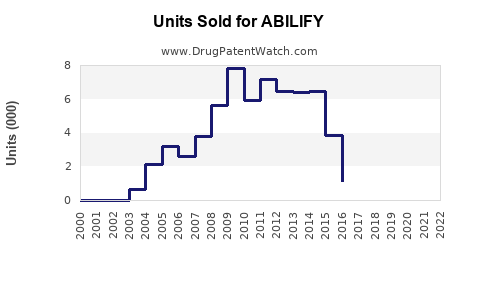
ABILIFY generated approximately $4.4 billion in worldwide sales. The United States accounts for roughly 80% of this figure, with sales reaching approximately $3.52 billion.
Key Markets:
- United States: Major contributor with steady growth driven by expansion of approved indications.
- Europe and Asia: Smaller contributions, with revenues around $880 million combined, influenced by regulatory approvals and local pricing policies.
Market Share:
ABILIFY is one of the top-selling atypical antipsychotics, competing primarily with Risperdal (risperidone), Seroquel (quetiapine), and newer agents like Vraylar (cariprazine) and Rexulti (brexpiprazole). It maintained approximately 15% of the global antipsychotic market share in 2022.
Market Drivers
-
Approved Indications:
- Schizophrenia
- Bipolar disorder (manic and mixed episodes)
- Major depressive disorder (adjunctive therapy)
- Autism-related irritability
- Tourette's syndrome
-
Off-label Use:
Some use for treatment-resistant depression and behavioral management in neuropsychiatric conditions, although off-label sales are not captured officially.
-
Market Expansion:
- Increasing approval in Japan (2015) contributed to Asia sales growth.
- New formulations, including a once-monthly injectable, broaden treatment options.
-
Reimbursements and PBMs:
Insurance coverage significantly influences prescribing patterns. Inflation-adjusted drug prices and formulary placement impact revenue.
Competition Analysis
| Drug |
Company |
Year Approved |
Indications |
Approximate 2022 Sales |
Market Share (2022) |
| ABILIFY |
Otsuka/Bristol-Myers |
2002 |
Schizophrenia, bipolar, depression, autism, Tourette’s |
$4.4 billion |
15% |
| Risperdal (risperidone) |
Janssen |
1993 |
Schizophrenia, bipolar |
$2.5 billion |
8% |
| Seroquel (quetiapine) |
AstraZeneca |
1997 |
Schizophrenia, bipolar, depression |
$2 billion |
6% |
| Vraylar (cariprazine) |
Allergan (AbbVie) |
2015 |
Schizophrenia, bipolar |
$350 million |
<2% |
| Rexulti (brexpiprazole) |
Lilly |
2015 |
Schizophrenia, depression |
$300 million |
<2% |
Note: The dominance of ABILIFY is challenged by newer agents with improved side-effect profiles but remains significant due to its early market entry and broad indication profile.
Future Sales Forecast
Forecast Period: 2023–2027
Assumptions:
- Continued approval across existing indications with modest gains in depression adjunct use.
- Introduction of generic versions in 2025 in the U.S., leading to a sales decline but offset by increasing global volumes.
- Expansion into emerging markets with favorable pricing and regulatory acceptance.
| Year |
Estimated Global Sales |
Growth Rate (YoY) |
Notes |
| 2023 |
$4.6 billion |
+4.5% |
Slight increase due to expanding indications |
| 2024 |
$4.8 billion |
+4.3% |
Growing Asian and European markets |
| 2025 |
$3.2 billion (post-generic) |
-33% (U.S.) |
U.S. market sees volume decline; global remains steady or up |
| 2026 |
$3.5 billion |
+9.4% |
Volume growth outside the U.S. |
| 2027 |
$3.7 billion |
+5.7% |
Stabilization with global markets driving sales |
Note:
The impact of patent cliffs and generic entries marks a notable shift, but global demand, especially in China and India, supports long-term growth.
Regulatory and Policy Impact
-
Patent Expiry:
U.S. patents expired in 2025, leading to increased generic competition. Europe faced similar patent expirations in 2024, with generic versions available since then.
-
Pricing and Reimbursement:
Policy shifts toward value-based pricing and increased price negotiations threaten high-margin sales. The U.S. has implemented price caps on branded drugs for federal programs.
-
Emerging Markets:
Governments in Asia, Africa, and Latin America prioritize affordable antipsychotics, creating growth opportunities.
Risks and Opportunities
Risks:
- Patent expiry leading to volumes reliant on generics.
- Competition from newer agents with improved side-effect profiles.
- Stringent regulatory requirements for expanding indications.
- Pricing pressures from payers.
Opportunities:
- Development of long-acting injectable formulations.
- Expansion into new indications like dementia-related agitation (pending approval).
- Growing mental health awareness increasing overall demand.
Key Takeaways
- ABILIFY remains a significant contributor to Otsuka's revenue, with global sales exceeding $4 billion in 2022.
- Patent expiry in the U.S. in 2025 is expected to cause a sales decline locally but may be offset globally.
- Competition from newer antipsychotics and generics poses a challenge but expanding indications and formulations offer growth avenues.
- Regulatory policies and reimbursement dynamics will heavily influence future sales trajectories.
- Long-term prospects hinge on R&D efforts, market expansion, and strategic management of patent and generic competition.
FAQs
1. When will ABILIFY face generic competition in the U.S.?
Patent exclusivity ended in 2025, allowing generic versions to enter the U.S. market.
2. What are the primary indications for ABILIFY?
Schizophrenia, bipolar disorder, depression adjunct therapy, irritability in autism, and Tourette’s syndrome.
3. How does ABILIFY compare with newer antipsychotics?
While it has a broad indication profile and long market presence, newer drugs like Vraylar and Rexulti differentiate through side-effect profiles and targeted indications.
4. What is the outlook for ABILIFY in emerging markets?
Strong, driven by demand for affordable mental health medications and government healthcare initiatives.
5. How might policy changes affect future revenues?
Price negotiations, reimbursement policies, and approvals for new indications could either constrain or expand sales.
References
- IQVIA. (2022). Pharmaceutical Market Analytics.
- Otsuka Pharmaceutical. (2023). Annual Report.
- MarketWatch. (2023). ABILIFY sales data.
- FDA. (2022). Drug Approvals and Patent Expirations.
- EvaluatePharma. (2023). Global Pharma Market Forecasts.