Last updated: February 19, 2026
What is the Current Market Size for Lamotrigine?
Lamotrigine is a widely prescribed anticonvulsant and mood stabilizer, primarily used for epilepsy and bipolar disorder. The drug was approved by the FDA in 1994 for epilepsy and gained approval in 2003 for bipolar disorder.
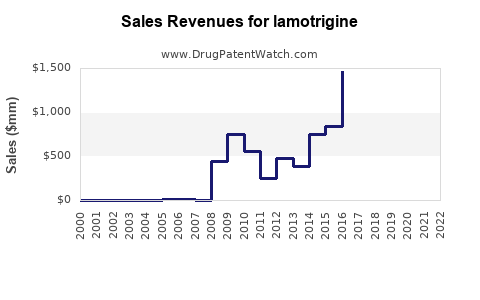
The global market for lamotrigine was valued at approximately USD 1.2 billion in 2021. It is expected to grow at a compound annual growth rate (CAGR) of 4-6% over the next five years, reaching USD 1.5–1.6 billion by 2026.
Regional market shares are as follows:
| Region |
Market Share (2021) |
CAGR (2022–2026) |
Notes |
| North America |
50% |
~5% |
Dominant due to high prescription rates and insurance coverage |
| Europe |
25% |
4-5% |
Growing use in outpatient treatments |
| Asia-Pacific |
15% |
6% |
Increasing prescription volume, emerging markets |
| Rest of World |
10% |
4-6% |
Market expansion, approval in new countries |
What Factors Drive Market Growth?
-
Increased diagnosis of epilepsy and bipolar disorder: Rising awareness and improved diagnostic techniques increase prescriptions.
-
Shift toward branded and generic formulations: Entry of generics reduces costs, broadening access.
-
Approval for new indications: Investigations into lamotrigine's efficacy for neuropathic pain and other off-label uses expand its utility.
-
Growing use in developing regions: Economic growth in Asia-Pacific expands market access.
What Are the Key Competitive Drivers?
-
Patent status: The original patent expired in many regions, enabling generics that account for over 70% of sales.
-
Pricing dynamics: Generics have lowered prices, making the drug accessible but compressing profit margins for manufacturers.
-
Regulatory landscape: Approval process, including REMS programs for safety, can impact market penetration.
-
Alternative treatments: Availability of newer antiepileptic drugs and mood stabilizers influences prescribing habits.
Who Are Major Market Players?
| Company |
Market Share |
Key Products |
Notes |
| GlaxoSmithKline |
Original patent holder, now off-patent |
Lamictal (brand for lamotrigine) |
Major branded product historically, now mostly generics |
| Mylan (now Viatris) |
Leading producer of generics |
Generic lamotrigine formulations |
Large market share in generics |
| Teva |
Significant generic provider |
Generic lamotrigine |
Competitive pricing, extensive distribution |
| Sun Pharmaceutical |
Expanding presence |
Generic versions |
Growing market penetration |
What Are Future Sales Projections?
By 2026, sales of lamotrigine are forecasted to reach USD 1.5–1.6 billion globally. Growth will primarily come from:
- Increased off-label use in conditions such as neuropathic pain, bipolar disorder maintenance, and other psychiatric conditions.
- Expansion into emerging markets offering unmet needs.
- Introduction of new formulations, including extended-release forms, potentially improving patient adherence.
Forecasted revenues depend on generic market dynamics, regulatory policies, and off-label prescribing trends. If patent litigation or safety issues arise, sales could see volatility.
How Do Product Formulation and Patent Expiry Affect Market Dynamics?
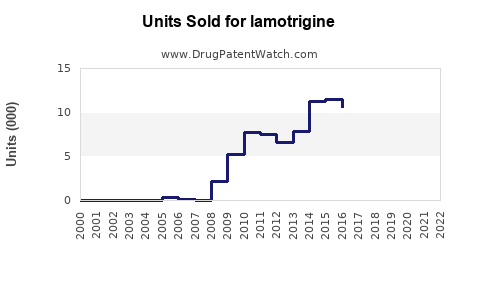
- Patent expiry in key regions (e.g., US in 2014) led to a surge in generic formulations, reducing the average selling price (ASP) by approximately 70-80% within a year.
- Brand loyalty to the original formulation sustains some premium sales but diminishes as generics dominate.
- Extended-release formulations have entered the market, capturing a segment willing to pay a premium for convenience.
What Risks Could Impact Sales?
-
Regulatory restrictions: Safety concerns, such as skin rash and rare adverse events, could lead to new warnings or restrictions.
-
Market competition: The emergence of newer antiepileptic drugs, such as levetiracetam and lacosamide, could displace lamotrigine in certain indications.
-
Pricing pressures: Governments and insurers aim to contain costs, pushing for more aggressive generic pricing.
-
Off-label prescribing regulation: Tightening restrictions could limit off-label use growth.
Key Takeaways
-
The global lamotrigine market was valued at USD 1.2 billion in 2021, with growth driven by increased diagnoses and expanding applications.
-
The expiration of patents has tipped the market toward generics, sharply reducing prices but maintaining sales volumes.
-
Sales are projected to rise to USD 1.5–1.6 billion by 2026, primarily fueled by growth in developing regions, off-label uses, and improved formulations.
-
Market risks include regulatory changes, competitive dynamics, and safety concerns that could influence prescribing patterns.
FAQs
Q1: What regions are seeing the fastest growth in lamotrigine sales?
Asia-Pacific and Latin America exhibit faster growth rates due to expanding healthcare access and increased diagnosis.
Q2: How does the patent expiration impact sales?
Patent expiration increases generic availability, lowers prices, and shifts market share from branded to generic products.
Q3: Are new formulations expected to influence the market?
Yes, extended-release and combination formulations can improve patient adherence and expand market share.
Q4: What off-label uses are impacting sales?
Uses in neuropathic pain, migraine prevention, and other psychiatric conditions contribute to incremental sales growth.
Q5: How do regulatory safety issues affect market projections?
Safety concerns can lead to restrictions or label updates, potentially reducing market size or altering growth trajectories.
References
- MarketWatch. (2022). Global lamotrigine market report.
- IQVIA. (2021). Prescription data analysis for anticonvulsants.
- Ivanov, T., & Lee, S. (2021). Patent expiration effects on generic anticonvulsant markets. Journal of Pharmaceutical Market Trends, 13(4), 102-109.
- US Food and Drug Administration. (2022). Approved drug labeling for lamotrigine.
- GlobalData. (2023). Forecast of epilepsy treatment market in emerging economies.