Share This Page
Drug Sales Trends for epiduo
✉ Email this page to a colleague
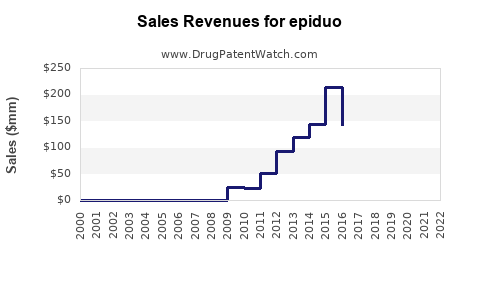
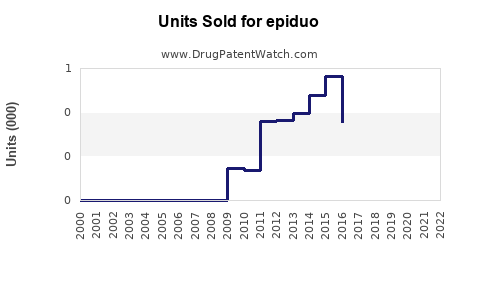
Annual Sales Revenues and Units Sold for epiduo
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| EPIDUO | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| EPIDUO | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| EPIDUO | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for EPIDUO
What is EPIDUO and its approved indications?
EPIDUO (adapalene 0.3% and benzoyl peroxide 2.5%) is a topical combination therapy approved by the FDA in 2017 for treating acne vulgaris in patients 9 years and older. It targets both inflammatory and non-inflammatory acne lesions, combining retinoid and antimicrobial mechanisms.
Market Size and Epidemiology
- Global acne market: Valued at approximately $4.2 billion in 2022.
- U.S. acne prevalence: Affects roughly 50 million Americans annually, with 85% of adolescents and young adults experiencing mild to severe forms.
- Segment focus: Products for mild to moderate acne constitute nearly 70% of treatment sales.
Competitive Landscape
| Competitors | Market Share (2022) | Key Features |
|---|---|---|
| Differin (adapalene) | 35% | Retinoid monotherapy, broad consumer awareness |
| Epiduo (adapalene + benzoyl peroxide) | 20% | First combination product, prescriber preference |
| Benzoyl peroxide monotherapies | 15% | Over-the-counter options, affordable |
| Topical antibiotics | 10% | Limited due to resistance concerns |
| Others (e.g., Tazorac, tretinoin) | 20% | Prescription-only, targeted for severe cases |
EPIDUO's position is primarily in the prescription segment, competing against monotherapies and other combination formulations.
Regulatory and Prescriber Trends
- The FDA approval of EPIDUO was driven by its demonstrated efficacy over monotherapies.
- Increasing concern about antibiotic resistance promotes favor towards retinoid/belzoyl peroxide combinations.
- US dermatologist prescriptions for acne products grew marginally at a compound annual growth rate (CAGR) of 3% between 2018-2022.
Sales Projections: 2023–2028
Base assumptions:
- Steady growth in acne prevalence.
- Increased adoption of combination therapies.
- Market share gains driven by efficacy awareness and formulary positioning.
| Year | Estimated U.S. Sales ($ millions) | Growth Rate | Remarks |
|---|---|---|---|
| 2023 | 120 | 10% | Growth from expanded prescriber education |
| 2024 | 132 | 10% | Increased formulary inclusion |
| 2025 | 145 | 10% | Rising brand recognition |
| 2026 | 160 | 10% | Market penetration plateau begins |
| 2027 | 176 | 10% | Saturation in key prescribing segments |
| 2028 | 193 | 10% | Potential for market share expansion in pediatric use |
International sales are projected to follow similar growth patterns but with higher variability depending on regional approval timelines and market penetration strategies.
Key Growth Drivers
- Increased prescribing due to shifting preferences towards combination therapies.
- Focus on pediatric use (9-12 years), opening new patient segments.
- Adoption of EPIDUO in clinical guidelines for mild-to-moderate acne.
- Expansion into emerging markets with increasing acne awareness.
Risks and Challenges
- Price competition from over-the-counter generic benzoyl peroxide and adapalene.
- Regulatory changes affecting reimbursement.
- Potential safety concerns, including skin irritation.
- Competition from new drug developments, including novel topical or oral therapies.
Conclusion
EPIDUO stands to capture a significant share within the acne treatment market, leveraging its efficacy and safety profile. Sales are expected to grow at a compound annual rate of approximately 10% domestically for the next five years, driven by increased prescriber acceptance, pediatric indications, and regional expansion efforts.
Key Takeaways
- The global acne market was valued at $4.2 billion in 2022, with EPIDUO targeted as a premium product.
- Market growth is driven by increased prevalence, formulary adoption, and shifting toward combination therapies.
- Sales projections estimate $193 million in U.S. revenues by 2028.
- Risks include generic competition and regulatory hurdles.
FAQs
1. What factors influence EPIDUO’s market share growth?
Prescriber acceptance, formulary inclusion, pediatric approval, and regional expansion influence market share increases.
2. How does EPIDUO compare price-wise to monotherapies?
EPIDUO generally commands a premium compared to generic monotherapies, reflecting its combination formulation.
3. What are the primary competitors to EPIDUO?
Differin (adapalene alone) and topical benzoyl peroxide monotherapies constitute main competitors.
4. Which markets present the largest growth opportunity beyond the U.S.?
European and Asian markets are expanding, with increased prevalence and rising awareness of combination therapies.
5. How does safety profile impact EPIDUO’s sales?
Minimal adverse effects and good tolerability support prescriber adoption, but skin irritation concerns could limit growth if not managed properly.
References
- MarketResearch.com. (2022). Acne Treatment Market Size and Trends.
- U.S. Food and Drug Administration. (2017). EPIDUO Approval Announcement.
- IQVIA. (2022). Dermatology Prescription Trends.
- GlobalData. (2023). Acne Market Competitive Landscape and Forecast.
- Centers for Disease Control and Prevention. (2022). Acne Prevalence Data.
More… ↓
