Share This Page
Drug Sales Trends for acetaminophen
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for acetaminophen (2006)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
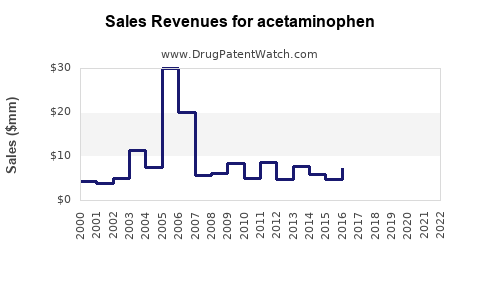
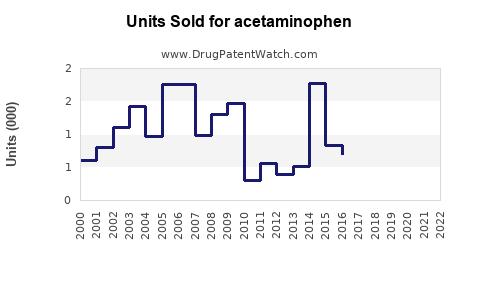
Annual Sales Revenues and Units Sold for acetaminophen
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ACETAMINOPHEN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ACETAMINOPHEN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ACETAMINOPHEN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ACETAMINOPHEN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Acetaminophen Market: Patent Landscape and Sales Projections
Acetaminophen, a widely available over-the-counter (OTC) analgesic and antipyretic, demonstrates a mature market with stable, predictable sales driven by broad consumer use and generic availability. While the patent landscape for the molecule itself expired decades ago, innovation continues in formulation, delivery mechanisms, and combination therapies. Sales are projected to remain consistent, with minor growth influenced by demographic shifts and increased awareness of pain management options.
What is the current market size and projected growth for acetaminophen?
The global acetaminophen market is substantial, with sales driven by its ubiquitous presence in households and healthcare systems. In 2022, the global acetaminophen market was valued at approximately USD 4.5 billion. Projections indicate a compound annual growth rate (CAGR) of 2.5% to 3.5% over the next five years. This growth is primarily attributed to factors such as increasing prevalence of chronic pain conditions, a growing elderly population, and its accessibility as an affordable pain relief option.
Global Acetaminophen Market Value (USD Billion)
| Year | Market Value |
|---|---|
| 2022 | 4.5 |
| 2023 | 4.6 |
| 2024 | 4.7 |
| 2025 | 4.8 |
| 2026 | 5.0 |
| 2027 | 5.1 |
Source: Market research reports and industry analysis.
What are the key patent trends impacting acetaminophen?
The foundational patent for acetaminophen, originally developed by Bayer in the late 19th century, has long since expired. Consequently, the market is dominated by generic manufacturers. However, ongoing patent activity focuses on:
- Novel Formulations: Patents are filed for extended-release formulations designed to provide longer-lasting pain relief and reduce dosing frequency. This includes technologies like osmotic pump systems and microencapsulation.
- Combination Therapies: New patents often cover formulations combining acetaminophen with other active pharmaceutical ingredients (APIs) for synergistic effects or to target multiple pain pathways. Examples include combinations with opioids (for prescription use), caffeine, or other OTC pain relievers.
- Delivery Systems: Innovation extends to improved delivery methods, such as rapidly dissolving tablets, oral suspensions with enhanced palatability, and even transdermal patches.
- Manufacturing Processes: While less common for established molecules, process patents for more efficient, cost-effective, or environmentally friendly manufacturing of acetaminophen or its precursors can emerge.
- New Indications/Therapeutic Uses: Research into new or underappreciated therapeutic applications of acetaminophen, even in low doses, could lead to new patent filings if novel mechanisms of action or efficacy are demonstrated.
Key Patent Filing Areas (Last 5 Years)
- Extended-release formulations
- Combination products with analgesics and adjuvants
- Taste-masking and improved oral delivery systems
- Specific polymorphic forms with improved stability or bioavailability
Source: Patent databases and analysis of recent filings.
What are the primary drivers of acetaminophen sales?
Acetaminophen's market success is underpinned by several key drivers:
- Broad Efficacy: It is effective for a wide range of mild to moderate pain conditions, including headaches, muscle aches, backaches, and fever.
- Safety Profile (When Used Appropriately): Compared to NSAIDs, acetaminophen generally has a lower risk of gastrointestinal side effects, making it a preferred choice for individuals with sensitive stomachs or certain medical conditions. Its safety profile in pregnancy is also a significant factor.
- Affordability and Accessibility: As a widely available generic drug, acetaminophen is highly affordable and accessible without a prescription, making it a cornerstone of self-care for pain management.
- Pediatric Use: It is a common and recommended first-line treatment for fever and pain in infants and children.
- Combination with Other Drugs: The availability of combination products, particularly those including caffeine or other analgesics, expands its utility for specific pain types.
- Demographic Trends: An aging global population, which often experiences higher rates of chronic pain, contributes to sustained demand.
Key Sales Drivers:
- Headache relief
- Fever reduction
- Musculoskeletal pain
- Post-operative pain (mild)
- Pediatric pain and fever
- Low risk of GI side effects
What are the regulatory considerations and potential challenges for acetaminophen?
Despite its established position, acetaminophen faces regulatory scrutiny and potential challenges:
- Hepatotoxicity Concerns: The primary safety concern is liver damage from overdose. Regulatory bodies continuously monitor adverse event reports and have implemented measures to limit maximum daily dosages and promote consumer education. The FDA has issued public safety warnings and engaged with manufacturers regarding labeling and packaging to reduce overdose risk.
- Labeling Requirements: Stringent labeling requirements are in place to inform consumers about maximum daily doses, potential for liver damage, and the importance of checking for acetaminophen in other medications to avoid accidental overdose.
- Manufacturing Standards: Like all pharmaceuticals, acetaminophen manufacturing must adhere to Good Manufacturing Practices (GMP) to ensure product quality, purity, and consistency.
- Competition from NSAIDs and Newer Analgesics: While acetaminophen holds a strong position, it competes with NSAIDs (like ibuprofen and naproxen) which offer anti-inflammatory benefits, and with newer prescription pain relief options, particularly for moderate to severe pain.
- Supply Chain Vulnerabilities: Like many generic APIs, the supply chain for acetaminophen can be vulnerable to disruptions, impacting availability and price. Reliance on a few key manufacturing regions for API production presents a potential risk.
Regulatory and Market Challenges:
- Liver toxicity risk management
- Adherence to evolving labeling guidelines
- Competition from NSAIDs and novel analgesics
- Global API supply chain stability
- Consumer education on safe usage
What are the competitive landscape and market segmentation for acetaminophen?
The acetaminophen market is highly competitive and segmented primarily by:
- Formulation:
- Tablets (Immediate Release)
- Extended-Release Tablets
- Liquids/Suspensions
- Suppositories
- Rapidly Dissolving Tablets
- Combination Products (e.g., with caffeine, opioids - prescription)
- Distribution Channel:
- Retail Pharmacies (OTC)
- Supermarkets and Mass Merchandisers
- Hospitals and Clinics (both OTC and prescription combinations)
- Online Retailers
- Brand vs. Generic: While branded versions (e.g., Tylenol) maintain strong brand recognition, generic alternatives capture the majority of the market volume due to significant price advantages.
Major Market Players (Examples):
- Johnson & Johnson (Tylenol brand)
- GSK plc (Panadol brand)
- Perrigo Company plc
- Teza-Perrigo
- Sanofi S.A.
- Various large generic manufacturers globally.
The market is characterized by price-sensitive consumers and a focus on volume sales. Brand loyalty exists, particularly for established brands, but is often overcome by cost considerations for generic options.
What are the future sales projections and growth factors?
Future sales projections for acetaminophen remain positive, albeit with moderate growth rates. Key factors influencing these projections include:
- Aging Global Population: The increasing number of individuals aged 65 and older is a significant driver, as this demographic typically experiences higher incidence of pain and fever.
- Rising Chronic Pain Prevalence: An increase in conditions leading to chronic pain, such as arthritis and lower back pain, will sustain demand.
- Emerging Markets: Growing access to healthcare and increased consumer spending on health and wellness products in emerging economies will contribute to market expansion.
- Continued Innovation in Formulations: Development of more convenient or effective delivery systems could capture new market segments or increase patient compliance. For example, combination products targeting specific pain types (e.g., migraine formulations) can create niche growth.
- Public Health Initiatives: Continued emphasis on non-opioid pain management strategies by healthcare systems and governments will benefit acetaminophen, positioning it as a preferred first-line treatment.
Factors Driving Future Growth:
- Geriatric population expansion
- Increased chronic pain incidence
- Healthcare access in developing economies
- Development of advanced drug delivery systems
- Emphasis on opioid-sparing pain management
The market will likely see continued volume growth, with value growth potentially augmented by the introduction of more sophisticated, higher-priced formulations or combination products.
Key Takeaways
Acetaminophen holds a stable, mature market position driven by its efficacy, safety profile, affordability, and broad accessibility. The patent landscape is characterized by expired foundational patents, with innovation focused on formulations, delivery systems, and combination therapies. Future sales are projected to grow modestly, supported by demographic trends, increasing chronic pain prevalence, and emerging markets. Regulatory oversight remains focused on managing hepatotoxicity risks and ensuring product safety and quality.
FAQs
-
What is the primary patent expiry date for acetaminophen? The original composition of matter patent for acetaminophen expired in the early to mid-20th century. Specific formulation patents may have later expiry dates, but the active ingredient itself has been off-patent for decades.
-
Are there any significant new chemical entities (NCEs) for acetaminophen? No, there are no new chemical entities based on the acetaminophen molecule itself. Innovation lies in how it is formulated, combined, or delivered.
-
How does acetaminophen's safety profile compare to NSAIDs in the long term? Acetaminophen generally has a better gastrointestinal safety profile than NSAIDs, with a lower risk of ulcers and bleeding. However, acetaminophen carries a significant risk of liver damage if taken in excess, which is a primary safety concern and distinct from the long-term risks associated with chronic NSAID use.
-
What is the market share of generic acetaminophen versus branded versions? Generic acetaminophen commands a dominant market share, estimated to be over 80% of the total market volume globally, due to its significantly lower price point.
-
Can new patents be issued for acetaminophen if novel therapeutic uses are discovered? Yes, if a novel, non-obvious therapeutic use for acetaminophen is discovered and validated, it could potentially be patented as a method of treatment, provided it meets patentability criteria. This would not be a patent on the molecule itself, but on its specific application.
Citations
[1] Market Research Report. (2023). Global Acetaminophen Market Analysis. [Publisher Name].
[2] Industry Analysis. (2023). OTC Analgesics Market Trends. [Publisher Name].
[3] U.S. Food and Drug Administration. (Ongoing). Drug Safety Communications. Retrieved from [FDA Website URL] (Specific date of access not applicable for general retrieval).
[4] Patent Database Search. (2023). Recent patents related to acetaminophen formulations and delivery systems. [Patent Database Name].
More… ↓
