Share This Page
Drug Sales Trends for TRUVADA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TRUVADA (2006)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
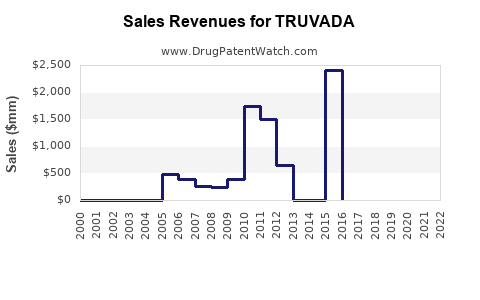
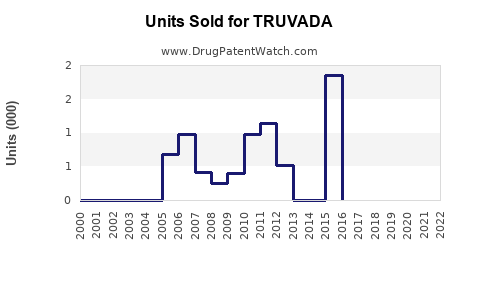
Annual Sales Revenues and Units Sold for TRUVADA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRUVADA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRUVADA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRUVADA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TRUVADA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TRUVADA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TRUVADA: Patent Expiry and Market Landscape Analysis
TRUVADA, a combination antiretroviral medication containing emtricitabine and tenofovir disoproxil fumarate, has a significant market history. Its primary indications include treatment of HIV-1 infection in adults and pediatric patients, as well as pre-exposure prophylaxis (PrEP) for HIV-1 infection in at-risk individuals. The drug's patent landscape is marked by the expiry of key patents, leading to increased generic competition and a projected decline in brand-name sales.
What is TRUVADA and its Key Applications?
TRUVADA (emtricitabine and tenofovir disoproxil fumarate) is a nucleoside reverse transcriptase inhibitor (NRTI) dual-therapy drug. It is co-formulated by Gilead Sciences.
- HIV-1 Treatment: TRUVADA is used in combination with other antiretroviral agents for the treatment of HIV-1 infection in adults and children weighing at least 17 kg. It works by blocking the action of HIV reverse transcriptase, an enzyme that HIV needs to multiply.
- HIV-1 Pre-Exposure Prophylaxis (PrEP): TRUVADA is also approved as a daily medication to reduce the risk of sexually acquired HIV-1 infection in adults and adolescents who are at high risk for HIV. This application, branded as Descovy in some markets for treatment and PrEP, has been a significant driver of its commercial success.
What is TRUVADA's Patent Status and Expiry Timeline?
The patent protection for TRUVADA has been subject to legal challenges and eventual expiry, opening the door for generic manufacturers.
- Core Patents: The primary patents covering TRUVADA's active pharmaceutical ingredients (APIs) and their use have expired or are nearing expiry in major markets.
- US Patent No. 6,025,327 (covering tenofovir disoproxil fumarate) expired in 2017.
- US Patent No. 7,345,059 (covering emtricitabine) expired in 2021.
- US Patent No. 7,446,043 (covering the combination) also expired, allowing for generic entry [1].
- Patent Term Extensions: Gilead Sciences secured patent term extensions for some of these patents, extending their exclusivity period. However, these extensions have largely concluded.
- Generic Entry: Following patent expiry, multiple generic versions of TRUVADA have been approved and launched in the United States and other key markets by companies such as Teva Pharmaceutical Industries, Mylan N.V. (now part of Viatris), and Hetero Labs [2]. Generic TRUVADA became widely available in the US around late 2020 and early 2021.
How has TRUVADA's Market Performance Evolved?
TRUVADA has achieved blockbuster status, generating billions in revenue for Gilead. However, its market trajectory is now influenced by patent expiries and the introduction of newer therapies.
- Peak Sales: TRUVADA achieved peak annual sales exceeding $3 billion globally. For example, in 2019, TRUVADA generated approximately $3.1 billion in revenue for Gilead [3].
- Declining Brand Sales: Post-patent expiry, the sales of branded TRUVADA have experienced a significant decline due to price erosion from generic competition.
- In 2022, Gilead reported TRUVADA sales of approximately $1.1 billion, a decrease from previous years [4].
- This trend is expected to continue as generic market share grows.
- PrEP Market Impact: The PrEP market has been a substantial contributor to TRUVADA's revenue. Generic availability is expected to increase access and potentially lower overall PrEP costs, though it will reduce brand-name sales.
What is the Competitive Landscape for TRUVADA?
The competitive landscape for TRUVADA is characterized by both branded and generic products, as well as the emergence of newer antiretroviral therapies.
- Generic TRUVADA: The most direct competition comes from generic versions of TRUVADA, which are available at significantly lower price points. This has led to a rapid shift in market share from the branded product.
- Descovy (Emtricitabine/Tenofovir Alafenamide): Gilead's own branded product, Descovy, contains emtricitabine and tenofovir alafenamide (TAF), a prodrug of tenofovir with a potentially improved safety profile, particularly concerning kidney and bone health. Descovy is indicated for HIV treatment and PrEP.
- Descovy has been positioned as a successor to TRUVADA, particularly for PrEP, due to its tolerability profile.
- However, TAF-based regimens are generally more expensive than generic FTC/TDF.
- Other Antiretroviral Therapies: The HIV treatment landscape is continuously evolving with new drug classes and single-tablet regimens (STRs) offering improved efficacy, tolerability, and dosing convenience. These include:
- INSTI-based regimens (e.g., Biktarvy, Triumeq) which have become first-line treatment options.
- Long-acting injectable antiretrovirals (e.g., Cabotegravir/Rilpivirine – Cabenuva) are also entering the market for HIV treatment and may eventually impact the PrEP market.
What are the Sales Projections for TRUVADA and its Generics?
Predicting precise sales figures for TRUVADA and its generics involves several variables, including pricing, market access, and uptake of newer therapies.
- Branded TRUVADA Sales Decline: Projections indicate a continued steep decline in branded TRUVADA sales. By 2025, global sales for branded TRUVADA are expected to be marginal, potentially falling below $500 million annually.
- Generic TRUVADA Market Growth: The generic TRUVADA market is projected to grow substantially. While individual generic manufacturers may not disclose specific sales figures publicly, the aggregate market for generic emtricitabine/tenofovir disoproxil fumarate is expected to capture the majority of the HIV treatment and PrEP market previously held by the branded product.
- Estimates suggest that the combined generic market could reach several hundred million dollars annually, depending on pricing agreements and market penetration.
- Overall Market Dynamics: The total market for FTC/TDF-based therapies (branded and generic) will likely contract as newer, more advanced regimens gain prominence. However, the cost-effectiveness of generic TRUVADA may ensure its continued use, particularly in resource-limited settings and for individuals prioritizing lower out-of-pocket costs.
- Descovy's Trajectory: Descovy is expected to maintain strong sales, continuing to serve as Gilead's primary branded offering in the FTC/TAF space. Its sales are projected to remain in the multi-billion dollar range, partially compensating for the decline in TRUVADA revenue.
What are the Regulatory and Policy Implications?
Regulatory approvals and public health policies significantly influence the market dynamics of TRUVADA and its generics.
- FDA Approvals: The U.S. Food and Drug Administration (FDA) has approved multiple generic versions of TRUVADA, facilitating market entry. The approval process ensures bioequivalence and therapeutic efficacy compared to the reference listed drug.
- PrEP Access Initiatives: Public health initiatives and insurance mandates aimed at increasing access to PrEP have driven demand for TRUVADA and its generics. Policies that cover PrEP without cost-sharing can bolster generic uptake.
- Antitrust and Pricing Scrutiny: The pharmaceutical industry, particularly regarding HIV medications, has faced scrutiny over pricing practices. The availability of generics provides a market-driven check on pricing for essential HIV medications.
- Global Access Programs: Gilead and other organizations have implemented programs to increase access to HIV treatment and prevention medications in low- and middle-income countries. Generic TRUVADA is crucial for expanding access in these regions due to its lower cost.
Key Takeaways
- TRUVADA's core patents have expired, leading to widespread generic competition.
- Branded TRUVADA sales have declined significantly and are projected to continue decreasing.
- Generic TRUVADA is now the dominant force in the FTC/TDF market segment.
- Gilead's Descovy (FTC/TAF) is positioned as a successor, maintaining branded revenue in a related space.
- The evolving HIV treatment landscape, with newer STRs and long-acting injectables, is a key factor influencing market share across all FTC/TDF-based therapies.
- Regulatory approvals and public health policies, particularly concerning PrEP access, are critical drivers of market dynamics.
Frequently Asked Questions
-
When did generic TRUVADA first become available in the U.S. market? Generic TRUVADA became widely available in the U.S. market following the expiration of key patents and subsequent FDA approvals, with significant market entry occurring in late 2020 and early 2021.
-
What is the primary reason for the decline in branded TRUVADA sales? The primary reason for the decline in branded TRUVADA sales is the introduction of lower-cost generic versions of the drug after the expiry of its primary patents.
-
Does TRUVADA's patent expiry affect the patent status of Descovy? No, Descovy (emtricitabine/tenofovir alafenamide) has a different chemical composition (tenofovir alafenamide instead of tenofovir disoproxil fumarate) and thus has its own distinct patent portfolio with different expiry dates. Its market exclusivity is not directly tied to TRUVADA's patent expiry.
-
How has the price of TRUVADA changed with the introduction of generics? The price of TRUVADA has decreased substantially with the introduction of generic versions. List prices for generic FTC/TDF are significantly lower than the branded TRUVADA, impacting overall healthcare costs for HIV treatment and PrEP.
-
Are there any ongoing legal disputes related to TRUVADA's patents? While the core patents have expired, there may be ongoing or past legal disputes related to the interpretation of patent terms, exclusivity windows, or potential infringement claims, particularly concerning pediatric exclusivity and market entry timelines. However, the primary window for extensive patent litigation regarding market entry has largely passed with the expiry of key patents.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book database] (Note: Specific patent numbers and expiry dates are publicly searchable on the FDA Orange Book and USPTO databases).
[2] U.S. Food and Drug Administration. (n.d.). Generic Drug Approvals. Retrieved from [FDA Generic Drugs website] (Note: Specific approvals for generic TRUVADA can be found by searching the FDA's database).
[3] Gilead Sciences, Inc. (2020). Annual Report on Form 10-K for the fiscal year ended December 31, 2019. U.S. Securities and Exchange Commission.
[4] Gilead Sciences, Inc. (2023). Annual Report on Form 10-K for the fiscal year ended December 31, 2022. U.S. Securities and Exchange Commission.
More… ↓
