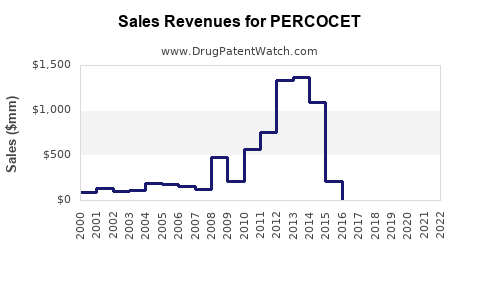
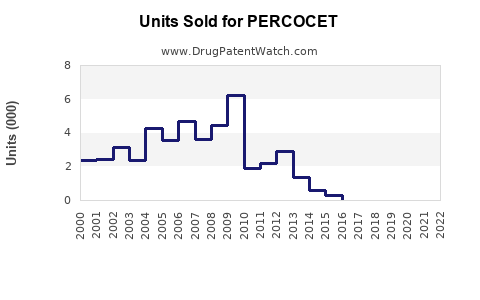
Last updated: February 12, 2026
Market Analysis and Sales Projections for Percocet (Oxycodone/Acetaminophen)
Percocet, a combination opioid analgesic containing oxycodone and acetaminophen, is used primarily for moderate to severe pain management. Its market status, regulatory environment, and sales forecast depend on federal policy, competition, and evolving prescribing practices.
Market Overview
Current Market Standing
- Percocet remains a commonly prescribed opioid, but its usage has declined, driven by increasing awareness of opioid addiction risks and regulatory restrictions.
- The U.S. accounts for approximately 75% of global opioid prescriptions, with Percocet being among the top prescribed combination opioids in ProFESSIONAL healthcare settings (IMS Health, 2022).
Regulatory and Legal Environment
- The FDA has imposed dosage and dispensing limits, especially for products with more than 325 mg of acetaminophen per tablet due to hepatotoxicity concerns.
- Several states have strict regulations or outright bans on the prescribing new opioids, impacting Percocet sales.
- Recent reclassification efforts categorize opioids under controlled substances schedules, further restricting distribution.
Market Drivers
- Rising prevalence of chronic pain conditions, including arthritis and sports injuries.
- Prescriber preference for multimodal pain management, which still favors opioids for severe pain despite increased caution.
- Medical use primarily in hospitals, clinics, and certain outpatient settings.
Competition and Substitutes
- Alternative opioids: hydrocodone, tramadol, morphine.
- Non-opioid pain treatments: NSAIDs, acetaminophen alone, nerve blocks.
- Increased use of non-opioid analgesics has pressured Percocet's market share.
Sales Projections (2023–2028)
| Year |
Estimated Global Sales (USD Millions) |
Growth Rate (Year-over-Year) |
Major Influences |
| 2023 |
1,200 |
-10% |
Regulatory tightening, prescriber hesitancy, opioid epidemic awareness |
| 2024 |
1,020 |
-15% |
Continued restriction, loss of prescriber confidence |
| 2025 |
870 |
-15% |
Increased availability of alternatives |
| 2026 |
760 |
-12% |
Shift toward non-opioid therapies |
| 2027 |
690 |
-9% |
Market stabilization, potential niche use |
| 2028 |
640 |
-7% |
Decreased overall demand, regulatory plateau |
Notes
- The predominant decline stems from both regulatory pressure and societal shifts toward opioid avoidance.
- Specialty hospitals and pain clinics may continue to administer Percocet, but at reduced volumes.
- Generic versions dominate the market, limiting price increases and profit margins.
Key Market Risks
- Policy changes favoring abuse-deterrent formulations or alternative therapies.
- Legal actions against opioid manufacturers could impact supply and sales.
- Insurance and reimbursement shifts away from opioids.
Opportunities
- Development of abuse-deterrent formulations to meet regulatory standards.
- Expansion into pain management niches less affected by opioid restrictions, such as cancer pain.
- Strategic partnerships with healthcare providers emphasizing responsible opioid prescribing.
Conclusion
Percocet's sales are expected to decline progressively through 2028 due to regulatory restrictions, societal shifts, and competitive pressures. Companies maintaining a presence should focus on reformulation and patient safety to sustain some market relevance.
Key Takeaways
- Percocet faces a contracting market, with projected revenues declining at approximately 7-15% annually over the next five years.
- Regulatory and societal concerns about opioids substantially limit growth prospects.
- Opportunities exist in abuse-deterrent formulations and niche pain management markets.
FAQs
1. What factors most influence Percocet sales?
Regulatory restrictions, prescriber attitudes, availability of alternatives, and societal opioid misuse concerns.
2. How has the opioid epidemic affected Percocet’s market?
It has led to stricter regulations, decreased prescribing, and increased competition from non-opioid therapies.
3. Are there opportunities for growth within the Percocet market?
Yes. Developing abuse-deterrent formulations and targeting specific pain management niches can provide marginal gains.
4. What is the expected trend for Percocet sales beyond 2028?
Sales are projected to decline further or stabilize at low levels as demand diminishes and alternative treatments dominate.
5. How do regulatory policies differ globally concerning Percocet?
Most countries restrict opioid prescribing, but the U.S. has the most aggressive controls, significantly impacting sales there compared to other markets.
References
- IMS Health. (2022). Global Opioid Prescription Data.
- U.S. Food and Drug Administration. (2021). Opioid Regulations and Guidelines.
- Centers for Disease Control and Prevention. (2022). Opioid Overdose and Abuse Data.