Share This Page
Drug Sales Trends for PENTASA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PENTASA (2006)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
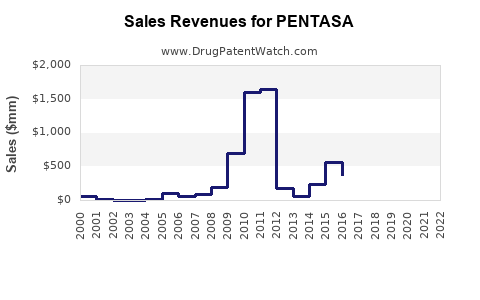
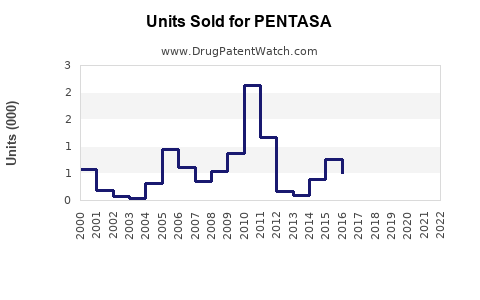
Annual Sales Revenues and Units Sold for PENTASA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PENTASA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PENTASA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PENTASA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PENTASA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PENTASA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PENTASA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PENTASA: Market Analysis and Sales Projections
PENTASA: Market Overview and Competitive Landscape
Pentasa (mesalamine), manufactured by Takeda Pharmaceutical Company, is a mesalamine-based drug used for the treatment of ulcerative colitis (UC). It is an extended-release formulation designed to deliver mesalamine to the entire colon, reducing inflammation in the gastrointestinal tract. The drug is available in various dosage forms, including capsules, tablets, and suppositories, catering to different patient needs and disease severities.
The global market for inflammatory bowel disease (IBD), which includes UC and Crohn's disease, is substantial and projected to grow. Factors driving this growth include increasing disease prevalence, advancements in diagnostic tools, and the development of novel therapeutic agents. Pentasa operates within this dynamic market, facing competition from other mesalamine formulations, biologics, and small molecule inhibitors.
What are the key indications for Pentasa?
Pentasa is primarily indicated for the treatment of mild to moderate ulcerative colitis. This includes both induction and maintenance of remission. The drug's mechanism of action targets inflammation within the colon wall. Clinical studies have demonstrated its efficacy in reducing symptoms such as bloody diarrhea, abdominal pain, and rectal bleeding. Off-label uses may exist but are not the primary market focus.
Who are Pentasa's main competitors?
The competitive landscape for Pentasa is segmented by both drug class and formulation.
- Other Mesalamine Formulations: Several other mesalamine drugs are available, including delayed-release formulations and topical preparations. Key competitors in this space include:
- Apriso (Allergan/FDA): An extended-release capsule.
- Lialda (Shire/Takeda): A multi-matrix system tablet.
- Asacol HD (Warner Chilcott/Allergan): A delayed-release tablet.
- Canasa (Salix Pharmaceuticals/Bausch Health): A delayed-release suppository and enema.
- Biologics: These are a significant class of drugs used for moderate to severe UC, often prescribed when mesalamine therapies are insufficient. Major biologic competitors include:
- Humira (adalimumab) (AbbVie): A TNF-alpha inhibitor.
- Remicade (infliximab) (Janssen Biotech): A TNF-alpha inhibitor.
- Entyvio (vedolizumab) (Takeda): An integrin receptor antagonist.
- Cimzia (certolizumab pegol) (UCB): A TNF-alpha inhibitor.
- Small Molecule Inhibitors: These oral medications offer an alternative mechanism of action.
- Xeljanz (tofacitinib) (Pfizer): A JAK inhibitor.
- Rinvoq (upadacitinib) (AbbVie): A JAK inhibitor.
The choice of therapy depends on disease severity, patient response, safety profiles, and cost. Pentasa generally positions itself as a first-line or early second-line therapy for mild to moderate UC.
What is the patent status of Pentasa?
Understanding the patent landscape is critical for assessing market exclusivity and the potential for generic competition. Pentasa's patent protection has evolved since its initial approval.
- Original Formulation Patents: Key patents covering the extended-release formulation of mesalamine have expired in major markets. For instance, in the United States, many of the foundational patents expired in the early to mid-2010s.
- Dosage and Method of Use Patents: Takeda may hold or have held secondary patents related to specific dosage regimens, manufacturing processes, or novel methods of using Pentasa. However, the strength and longevity of these patents can vary significantly.
- Generic Entry: The expiration of primary patents has paved the way for generic versions of mesalamine to enter the market. Generic manufacturers often leverage process patents or formulation variations to secure their own intellectual property. The presence of generics puts downward pressure on pricing and market share for the branded product.
What are the regulatory approvals for Pentasa?
Pentasa has received regulatory approval from major health authorities worldwide.
- United States: Approved by the U.S. Food and Drug Administration (FDA) for the treatment of ulcerative colitis.
- European Union: Approved by the European Medicines Agency (EMA).
- Other Markets: Approvals are secured in other key pharmaceutical markets, including Japan and Canada.
Regulatory hurdles for new mesalamine formulations or indications are typically high, requiring robust clinical trial data demonstrating safety and efficacy.
Pentasa: Sales Performance and Market Projections
Assessing the sales performance and projecting future market demand for Pentasa requires an analysis of historical sales data, market trends, and the impact of competition and patent expirations.
What are Pentasa's historical sales figures?
Takeda's reporting of specific product sales can be segmented by region and therapeutic area. While exact global sales figures solely for Pentasa can be challenging to isolate from broader mesalamine portfolios or overall IBD drug sales, available data indicates a mature product with significant, albeit potentially declining, revenue streams.
- Global Sales Trends: For the fiscal year ending March 31, 2023, Takeda reported total sales for its Gastrointestinal products, which include Pentasa, as approximately ¥380 billion (USD $3.04 billion at an exchange rate of ¥125/USD). Within this segment, mesalamine products have historically contributed a substantial portion.
- Regional Performance: North America and Europe typically represent the largest markets for mesalamine products due to higher UC prevalence and established treatment protocols. Sales in these regions are influenced by formulary access, reimbursement policies, and the intensity of generic competition.
- Impact of Generics: The introduction of generic mesalamine products has undoubtedly impacted Pentasa's market share and pricing power. While branded Pentasa may retain a portion of the market due to physician and patient preference or specific formulation advantages, the overall revenue growth for the branded product is likely constrained by this factor.
What are the projected market growth rates for Pentasa?
Projecting specific growth rates for Pentasa as a branded product is complex due to several compounding factors.
- Mature Market Status: As a well-established drug with expired primary patents, Pentasa is considered a mature product. Mature products generally exhibit slower growth or even declines, particularly in markets with significant generic penetration.
- IBS Market Growth: The overall market for ulcerative colitis treatments is expected to grow, driven by increased diagnosis rates and the development of novel therapies. However, this growth is likely to be captured more by newer biologic and small molecule drugs rather than mature mesalamine formulations.
- Generic Erosion: The continued presence and potential expansion of generic mesalamine competition will exert downward pressure on Pentasa's sales volume and average selling price.
- Takeda's Strategic Focus: Takeda's strategic priorities may shift towards newer, higher-growth pipeline assets, potentially leading to reduced marketing and promotional efforts for mature products like Pentasa.
Based on these factors, it is unlikely that Pentasa will experience significant global sales growth as a branded product. A modest decline or flat sales trajectory in developed markets is a more probable scenario, with potential for some volume in emerging markets where generic penetration might be slower. Projections for the broader mesalamine market, however, will be influenced by the ongoing need for first-line UC therapies.
What is the estimated market size for mesalamine drugs globally?
The global market for mesalamine drugs, encompassing all formulations and brands, is substantial.
- Market Size Estimate: Industry reports estimate the global mesalamine market to be in the range of USD $1.5 billion to $2.5 billion annually. This figure fluctuates based on market research methodologies and the inclusion of various product types.
- Growth Drivers: The growth of this market is driven by the increasing incidence of ulcerative colitis worldwide and the established role of mesalamine as a foundational treatment. Advancements in drug delivery systems also contribute to market expansion by offering improved efficacy and patient compliance.
- Regional Distribution: North America and Europe are the largest contributors to the mesalamine market. Asia-Pacific is an emerging market with significant growth potential due to rising healthcare expenditure and increasing disease awareness.
Pentasa, as a key player within this market, contributes a significant portion to the overall mesalamine sales, but its share is subject to the competitive pressures outlined previously.
How do pricing strategies impact Pentasa's market position?
Pricing is a critical determinant of market share and profitability, especially for a mature drug like Pentasa facing generic competition.
- Branded Pricing: Takeda sets the price for branded Pentasa, taking into account manufacturing costs, R&D recoupment, marketing expenses, and perceived therapeutic value. The pricing also considers the competitive landscape, particularly the price points of other branded mesalamine products and biologics.
- Generic Pricing: Generic versions of mesalamine are typically priced significantly lower than the branded product, often at 20% to 50% of the branded price. This aggressive pricing by generic manufacturers is a major driver of market share displacement for branded drugs.
- Reimbursement and Formulary Access: Pharmaceutical benefit managers (PBMs) and payers negotiate drug prices to control healthcare costs. Branded Pentasa's inclusion on formularies and its co-payment structure for patients are heavily influenced by its price relative to generics and other UC therapies. Lower price points or higher co-pays for branded products can steer patients towards generics.
- Value-Based Pricing: In some markets, there is a move towards value-based pricing, where drug prices are linked to clinical outcomes. For a mature drug like Pentasa, demonstrating ongoing value and cost-effectiveness compared to newer, more expensive therapies is crucial for maintaining favorable pricing and market access.
Takeda's pricing strategy for Pentasa likely involves a balance between maintaining profitability from its established patient base and acknowledging the market realities of generic competition. This might involve differentiated pricing across regions or specific contract negotiations with payers.
Key Takeaways
Pentasa is an established mesalamine-based treatment for mild to moderate ulcerative colitis. Its market position is defined by a mature product lifecycle, significant generic competition following patent expirations, and a dynamic therapeutic landscape for inflammatory bowel disease. While the overall mesalamine market is substantial and driven by increasing UC prevalence, branded Pentasa's future sales trajectory is likely constrained by pricing pressures and market share erosion from lower-cost generic alternatives. Takeda's strategic decisions regarding pricing, lifecycle management, and market support will be critical in determining the long-term performance of Pentasa. The continued demand for effective first-line therapies for UC suggests an ongoing, albeit potentially declining, role for Pentasa and other mesalamine products in the global pharmaceutical market.
Frequently Asked Questions
1. How does Pentasa's extended-release formulation differ from other mesalamine delivery methods?
Pentasa utilizes an ethylcellulose-coated matrix to provide extended release of mesalamine throughout the colon. This contrasts with delayed-release formulations that release medication in the small intestine or at specific pH levels, and topical preparations (suppositories, enemas) that act locally in the rectum and distal colon. The extended-release design aims to ensure mesalamine reaches inflammatory sites along the entire length of the colon.
2. What is the impact of biologic therapies on Pentasa's market share?
Biologic therapies, such as TNF-alpha inhibitors and integrin receptor antagonists, are typically reserved for moderate to severe ulcerative colitis patients who have not responded to or cannot tolerate conventional therapies like mesalamine. While biologics are not direct competitors for mild to moderate UC where Pentasa is indicated, they represent the next tier of treatment, and a portion of patients who initially fail mesalamine may transition to biologics, indirectly impacting the overall volume of mesalamine use.
3. Are there any new indications or formulations for Pentasa under development?
As a mature product, significant investment in novel indications or formulations for Pentasa is unlikely. The focus for Takeda and other manufacturers is typically on lifecycle management for established drugs. The development of new mesalamine-based treatments has largely shifted towards entirely new chemical entities or advanced delivery systems to circumvent existing patents and offer distinct advantages.
4. What is the typical patient profile for Pentasa treatment?
Pentasa is generally prescribed for adult patients diagnosed with mild to moderate ulcerative colitis. It is often a first-line therapy for many patients newly diagnosed with this condition. The choice to use Pentasa can also depend on the extent of the disease, with its colonic coverage being advantageous for pancolitis or left-sided colitis.
5. How does the cost of Pentasa compare to generic mesalamine alternatives?
Branded Pentasa is significantly more expensive than generic mesalamine formulations. The price differential can be as high as 50% or more. This cost difference is a primary driver for patients and payers to opt for generic versions once they become available, impacting branded Pentasa's market share.
Citations
[1] Takeda Pharmaceutical Company Limited. (2023). Integrated Report 2023. Takeda. [2] Various pharmaceutical market research reports (e.g., Grand View Research, Fortune Business Insights) on the IBD and mesalamine markets. (Specific report details not publicly available for direct citation without subscription).
More… ↓
