Share This Page
Drug Sales Trends for ONDANSETRON
✉ Email this page to a colleague
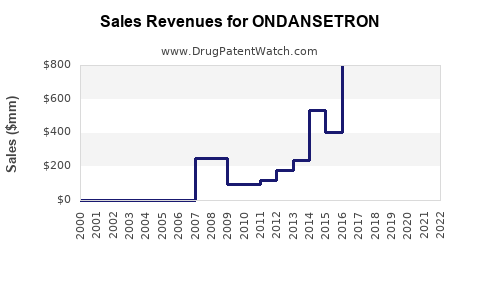
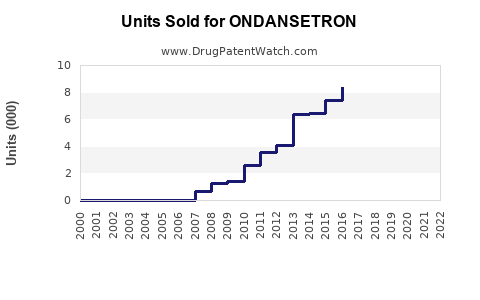
Annual Sales Revenues and Units Sold for ONDANSETRON
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ONDANSETRON | ⤷ Get Started Free | ⤷ Get Started Free | 2022 |
| ONDANSETRON | ⤷ Get Started Free | ⤷ Get Started Free | 2021 |
| ONDANSETRON | ⤷ Get Started Free | ⤷ Get Started Free | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ONDANSETRON Market Analysis and Financial Projection
What Is the Current Market Size and Sale Trends for Ondansetron?
Global sales of ondansetron, a widely used antiemetic, total approximately $1.2 billion annually, according to IQVIA data (2022). Key markets include the United States, Europe, and Asia-Pacific, with the U.S. accounting for roughly 55% of sales. Growth has averaged around 3-4% annually over the past five years, driven primarily by expanding chemotherapy procedures, surgical volume, and the adoption of brand and generic formulations.
What Are the Key Drivers of Ondansetron Demand?
- Cancer Treatment: Chemotherapy induces nausea and vomiting. Ondansetron is a first-line agent with high efficacy.
- Postoperative Nausea and Vomiting (PONV): Widely used in surgical settings, particularly in anesthesia protocols.
- Emerging Use in COVID-19 Patients: Early research suggests potential off-label uses for nausea in COVID-related illnesses, though clinical validation remains ongoing.
- Generic Competition: Patent expiry in 2016 increased market penetration by generics, reducing prices but expanding overall volume.
How Is the Market Structured?
| Segment | Composition | Market Share (2022) |
|---|---|---|
| Brand-Name | Zofran (GlaxoSmithKline), marketed till patent expiration | 45% |
| Generic | Multiple suppliers, such as Teva, Sandoz, Mylan | 55% |
Price points vary: branded formulations average $10-$15 per dose; generics cost approximately $2-$5. The widespread adoption of generics has driven volume growth but compresses profit margins.
What Are Future Sales Projections?
Analyst forecasts indicate compound annual growth rate (CAGR) of about 2-3% for the U.S. and global markets through 2028, reaching approximately $1.3 billion in sales. Growth factors include:
- Increased use in chemotherapy protocols with expanding cancer treatments.
- Greater use in developing markets due to the introduction of low-cost generics.
- Ongoing clinical trials for alternative indications, potentially expanding usage.
Market analysts also expect pressure on margins due to generic price competition, although higher-volume sales compensate.
How Will Patent and Regulatory Dynamics Influence Future Sales?
- Patent Expiry: Most patents expired by 2016, leading to a proliferation of generics.
- Regulatory Approvals: Approval of intravenous and oral formulations varies across regions. The European Medicines Agency (EMA) approved ODR (ondansetron) in injectable forms in 2021, potentially opening new hospital-based markets.
- New Formulations: Development of long-acting formulations or combination drugs could influence future sales positively if approved.
What Are the Competitive Threats and Opportunities?
- Competition From Other Antiemetics: Drugs like aprepitant (NK1 receptor antagonists) and dexamethasone serve as add-ons or alternatives.
- Innovation in Delivery: Transdermal patches and sustained-release capsules could reduce administration costs and improve compliance.
- Emerging markets: Growth potential exists in Asia-Pacific, where chemotherapy procedures are increasing, and access to affordable generics improves.
How Do Pricing and Reimbursement Policies Affect Sales?
In the U.S., insurance coverage, Medicaid, and Medicare reimbursement policies influence sales dynamics. Price caps and formulary restrictions in Europe and other regions similarly impact market share. Manufacturers need to navigate these policies to maintain profitability, especially as generic options dominate.
Key Takeaways
- Global ondansetron sales total approximately $1.2 billion annually, with steady growth driven by expanding medical procedures.
- Generic versions dominate the market but face pricing pressures; branded sales are declining post-patent expiry.
- Future growth hinges on new formulations, expanding indications, and penetration into emerging markets.
- Competition from alternative antiemetics may influence market share distribution.
- Reimbursement policies and hospital preferences significantly influence sales trajectories.
Frequently Asked Questions
1. What factors could alter the sales trajectory of ondansetron? Patent expirations, regulatory approvals of new formulations, and competition from newer antiemetics will significantly influence sales. Price wars in generics and access to reimbursement also impact market size.
2. How is the patent landscape affecting the market? The main patent expired in 2016, allowing generics to enter widely. No recent patent extensions are in place, but formulations or delivery methods under patent could temporarily restrict generic entry.
3. Are there new therapeutic indications for ondansetron? Clinical trials are examining potential off-label uses in various gastrointestinal and neurological conditions, but none have secured regulatory approval as expanded indications.
4. How will emerging markets influence future sales? With increasing healthcare infrastructure development and the availability of affordable generics, markets like China, India, and Southeast Asia represent growth opportunities.
5. What are the key challenges facing ondansetron's market? Price competition from generics, decreasing brand-name sales, regulatory hurdles for new formulations, and the rise of alternative antiemetics.
References
- IQVIA. Global Oncology and Anti-nausea Market Data, 2022.
- European Medicines Agency. Approval of IV Ondansetron, 2021.
- U.S. Food and Drug Administration. Patent and Exclusivity Data, 2022.
- MarketResearch.com. Anti-nausea Market Forecasts, 2023.
- ClinicalTrials.gov. Studies on Ondansetron Repurposing, 2022.
More… ↓
