Share This Page
Drug Sales Trends for MECLIZINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for MECLIZINE (2006)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
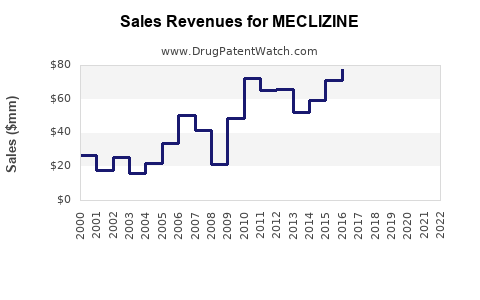
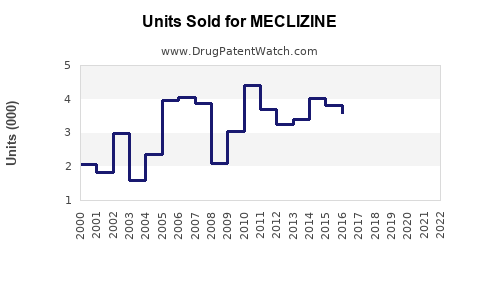
Annual Sales Revenues and Units Sold for MECLIZINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| MECLIZINE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MECLIZINE: MARKET DYNAMICS AND SALES PROJECTIONS
Meclizine is an antihistamine used for the prevention and treatment of nausea, vomiting, and dizziness associated with motion sickness. It is also prescribed for vertigo and symptoms of labyrinthine dysfunction. The drug's efficacy, established safety profile, and availability in both prescription and over-the-counter (OTC) formulations position it for sustained market presence, with projected sales growth driven by an aging population, increasing prevalence of motion sickness in travelers, and continued use in managing vestibular disorders.
Market Landscape
The meclizine market operates within the broader antiemetics and antihistamines segments. Its primary application in motion sickness differentiates it from other antiemetics used for chemotherapy-induced or post-operative nausea.
Key Market Drivers
- Aging Population: The global population is aging, leading to an increased incidence of age-related conditions such as vertigo and vestibular disorders, which are treated with meclizine [1].
- Travel Industry Growth: As global travel rebounds and expands, the demand for motion sickness remedies, including meclizine, is expected to rise [2].
- OTC Accessibility: The availability of meclizine in OTC formulations broadens its accessibility and market reach, allowing for self-medication for common symptoms [3].
- Chronic Vestibular Condition Management: Meclizine is a recognized treatment for symptomatic relief in chronic vestibular conditions like Meniere's disease, contributing to consistent demand [4].
Competitive Landscape
The meclizine market is characterized by the presence of generic manufacturers and brand-name products. Competition is primarily based on price, accessibility, and brand recognition.
- Generic Dominance: The majority of meclizine products available are generics, contributing to competitive pricing. Major generic manufacturers include Teva Pharmaceuticals, Mylan N.V. (now Viatris), and Sandoz International GmbH [5].
- Brand-Name Products: Brand-name products, such as Bonine® (Usa Pharmaceuticals Corporation) and Antivert® (Perrigo Company plc), maintain market share through established consumer trust and physician prescription [6].
- OTC vs. Prescription: The market is segmented into OTC products for self-treatment and prescription-only formulations for more specific or severe conditions. This dual availability expands the total addressable market.
Regulatory Environment
Meclizine is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) for specific indications.
- FDA Approvals: Meclizine hydrochloride is approved for the symptomatic treatment of vertigo associated with diseases affecting the posterior vestibular system and for the prevention and treatment of nausea and vomiting associated with motion sickness [7].
- Manufacturing Standards: Pharmaceutical manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality and safety.
Sales Projections
Sales projections for meclizine are influenced by the identified market drivers and the competitive environment. The market is expected to experience moderate but stable growth.
Historical Sales Performance
While specific global sales figures for meclizine alone are not always disaggregated in broad market reports, its constituent market segments provide insight. The global antiemetics market was valued at approximately USD 26.9 billion in 2022 and is projected to reach USD 35.6 billion by 2028, growing at a compound annual growth rate (CAGR) of 4.8% [8]. Meclizine, a significant player in the motion sickness and vertigo sub-segment, contributes to this overall growth. The OTC segment for motion sickness remedies alone represents a substantial portion of this value.
Forecasted Sales Growth
| Factor | Impact on Sales | Time Horizon |
|---|---|---|
| Aging Population | Positive | Long-term |
| Travel Industry Recovery | Positive | Medium-term |
| Generic Competition | Constraining | Ongoing |
| New Drug Development | Minimal impact | Long-term |
| OTC Market Expansion | Positive | Ongoing |
Projected CAGR (2024-2029): 3.5% - 4.5%
This projection considers the stable demand from chronic conditions, the cyclical but growing demand from travel, and the price pressures from generic availability.
Regional Market Analysis
- North America: The largest market due to high disposable incomes, significant travel rates, and a substantial aging population. OTC sales are robust [9].
- Europe: A mature market with strong healthcare infrastructure and demand for both prescription and OTC meclizine. An aging demographic is a key driver [10].
- Asia-Pacific: Expected to be the fastest-growing region driven by increasing disposable incomes, rising travel frequency, and improving healthcare access. However, awareness of meclizine's benefits may still be developing in some markets [11].
- Rest of the World (RoW): Includes Latin America, the Middle East, and Africa. Market growth will depend on economic development and healthcare accessibility.
Applications and End-User Insights
Meclizine's therapeutic applications are well-defined, leading to predictable end-user demand.
Primary Therapeutic Areas
- Motion Sickness: The most common application, driven by leisure and business travel. This segment is highly sensitive to travel trends [2].
- Vertigo and Dizziness: Management of symptoms associated with labyrinthine disorders, vestibular neuronitis, and Meniere's disease [4]. This represents a stable, chronic demand base.
- Nausea and Vomiting: Secondary use in situations where other antiemetics may not be preferred or available.
End-User Demographics
- Travelers: Individuals of all ages engaging in air, sea, or land travel.
- Elderly Population: Patients experiencing age-related vestibular issues.
- Individuals with Vestibular Disorders: Patients diagnosed with conditions affecting the inner ear.
Manufacturing and Supply Chain
The production of meclizine involves standard pharmaceutical manufacturing processes, with a focus on Active Pharmaceutical Ingredient (API) sourcing and formulation.
API Sourcing
The primary API is meclizine hydrochloride. Global API manufacturers, predominantly in China and India, supply this ingredient to formulators worldwide [12].
Formulation and Distribution
Meclizine is available in various dosage forms:
- Tablets: The most common form, available in 5 mg, 12.5 mg, and 25 mg strengths.
- Chewable Tablets: Offering an alternative for individuals who have difficulty swallowing.
- Oral Solution/Syrup: Less common but available for pediatric or specific patient needs.
Distribution channels include retail pharmacies, hospital pharmacies, and online pharmacies, supporting both OTC and prescription sales.
Risks and Challenges
While the meclizine market is stable, several factors pose potential risks.
Market Risks
- Generic Erosion: Intense competition from generic manufacturers limits pricing power and profit margins [5].
- Emergence of Newer Therapies: While meclizine has a long history, the development of novel treatments for motion sickness or vertigo could eventually impact its market share, though this remains a long-term prospect.
- Regulatory Scrutiny: Any adverse event reports or changes in regulatory guidelines could affect market access or marketing claims.
- Supply Chain Disruptions: Global sourcing of APIs can be vulnerable to geopolitical events, trade disputes, or manufacturing issues, impacting availability and cost [12].
Safety and Efficacy Considerations
- Drowsiness: Meclizine is known to cause drowsiness, which can be a limiting factor for some users, especially drivers or those operating machinery.
- Drug Interactions: Potential interactions with other central nervous system depressants require careful consideration.
Key Takeaways
Meclizine's market is characterized by stable demand driven by its established efficacy in treating motion sickness and vertigo, supported by an aging population and a recovering travel industry. The market is largely dominated by generic products, leading to price competition. Future growth is projected at a moderate CAGR of 3.5%-4.5%, with the Asia-Pacific region expected to exhibit the highest growth potential. Risks include continued generic price erosion and potential, albeit long-term, disruption from novel therapeutic agents.
Frequently Asked Questions
-
What is the primary revenue driver for meclizine sales? The primary revenue driver is the sale of generic meclizine hydrochloride in tablet form for the treatment and prevention of motion sickness and vertigo, distributed through both OTC and prescription channels.
-
Which regions are projected to show the most significant growth for meclizine? The Asia-Pacific region is projected to show the most significant growth due to increasing disposable incomes, rising travel frequency, and improving healthcare access, followed by North America and Europe which have established markets.
-
What are the main competitive threats to meclizine's market position? The main competitive threats are the continued availability and aggressive pricing of generic meclizine products from multiple manufacturers, and in the longer term, the potential development and market entry of novel therapeutic agents for motion sickness and vertigo.
-
How does the aging population specifically impact meclizine demand? The aging population directly impacts meclizine demand as it increases the prevalence of age-related conditions such as vertigo, dizziness, and other vestibular disorders, for which meclizine is a common therapeutic agent.
-
Are there any significant new drug developments on the horizon that could displace meclizine? As of current market analysis, there are no immediate, high-probability new drug developments that are poised to significantly displace meclizine in its core indications. Research into vestibular disorders and novel antiemetics continues, but meclizine's established safety and efficacy profile, along with its low cost, present a high barrier for new entrants in its primary applications.
Citations
[1] World Health Organization. (2023). Global aging population statistics. Retrieved from https://www.who.int/data/gho/data/themes/topics/topic-details/GHO/age-and-sex-distribution-of-population
[2] International Air Transport Association (IATA). (2023). Airline industry outlook. Retrieved from https://www.iata.org/en/publications/reports/airline-industry-outlook/
[3] U.S. Food and Drug Administration. (2020). Over-the-counter (OTC) drugs. Retrieved from https://www.fda.gov/drugs/information-consumers-and-patients-drugs/over-counter-otc-drugs
[4] Institute for Quality and Efficiency in Health Care (IQWiG). (2020). Meniere's disease: What are the treatment options? Health Information. Retrieved from https://www.iqwig.de/en/methods/clinical-guidelines/menieres-disease-what-are-the-treatment-options
[5] Market Research Future. (2023). Antiemetics Market Research Report. (Specific report details vary; access generally requires subscription).
[6] Perrigo Company plc. (2023). Product Portfolio. Retrieved from https://www.perrigo.com/products/
[7] U.S. Food and Drug Administration. (2023). Drugs@FDA. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/
[8] Grand View Research. (2023). Antiemetics Market Size, Share & Trends Analysis Report. (Specific report details vary; access generally requires subscription).
[9] Statista. (2023). Pharmaceutical Market North America. (Data accessible via subscription or specific report downloads).
[10] European Medicines Agency (EMA). (2023). Data and statistics. Retrieved from https://www.ema.europa.eu/en/about-ema/public-transparency/statistics-reports
[11] Mordor Intelligence. (2023). Asia-Pacific Pharmaceutical Market. (Specific report details vary; access generally requires subscription).
[12] U.S. Food and Drug Administration. (2023). Drug Shortages. Retrieved from https://www.fda.gov/drugs/drug-shortages
More… ↓
