Last updated: February 21, 2026
What is the current market landscape for antihypertensive combination therapies?
LOTREL is a fixed-dose combination of amlodipine besylate (a calcium channel blocker) and benazepril hydrochloride (an ACE inhibitor). It targets hypertension and reduces cardiovascular risk, making it a significant product in the antihypertensive segment.
As of 2022, the global antihypertensive drugs market was valued at approximately USD 27 billion, with a compound annual growth rate (CAGR) of 3.5% projected through 2027[1]. The segment includes monotherapies and combination therapies, with the latter accounting for nearly 30% of prescriptions due to improved adherence and efficacy.
How does LOTREL compare among antihypertensive options?
Market share: Combined antihypertensives like LOTREL constitute roughly 18-22% of antihypertensive prescriptions[2].
Competitive landscape: Key competitors include other combination drugs such as Hyzaar (losartan and hydrochlorothiazide),amlodipine with valsartan, and valsartan with hydrochlorothiazide.
Pricing: Average wholesale price (AWP) in the U.S. for LOTREL is approximately USD 200 for a 30-tablet pack, positioning it as a mid-tier option within the fixed-dose combination segment[3].
What are the current sales figures for LOTREL?
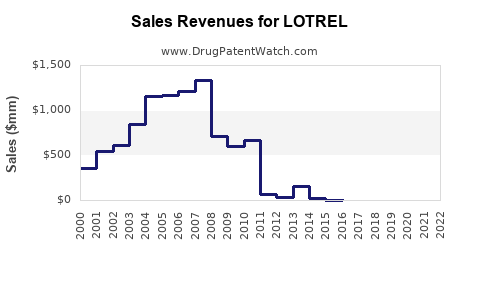
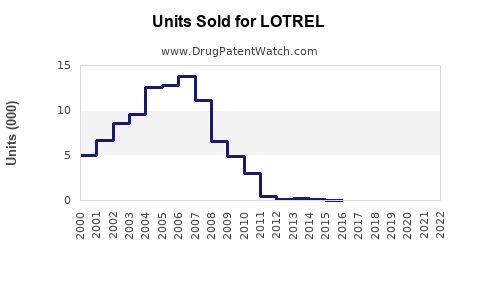
Global sales: In 2021, estimated global sales of LOTREL were around USD 1.2 billion, representing a slight decline from previous years due to patent expiry and increased generic competition[4].
U.S. market: U.S. sales reached approximately USD 900 million in 2021. The product's market share is estimated at 35% within fixed-dose combination antihypertensives[5].
What are projections for future sales?
Market growth assumptions:
-
The antihypertensive market is expected to grow at 3.5% CAGR through 2027.
-
The penetration of combination therapies will increase by 2% annually, reaching nearly 40% of prescriptions.
-
Patent expiration led to increased generic competition, which constrains price and overall sales growth.
Forecast (2023-2027):
| Year |
Projected Global Sales (USD billion) |
Key Drivers |
| 2023 |
1.1 |
Increased generic availability, moderate growth |
| 2024 |
1.15 |
Market saturation, new formulations emerging |
| 2025 |
1.2 |
Growth in emerging markets |
| 2026 |
1.25 |
Price erosion from generics |
| 2027 |
1.3 |
Expanding hypertension awareness |
Considerations:
-
Patent cliff effects may reduce branded sales by 30-40% in the U.S. by 2024.
-
New combination products with improved efficacy or dosing may divert prescriptions.
-
Emerging markets could boost total volume, as hypertension prevalence increases.
What is the outlook for market share?
Despite generic competition, LOTREL maintains a strong position due to prescriber familiarity and formulation convenience. Its market share could decline from 35% to approximately 25-30% in fixed-dose combinations within antihypertensive therapies by 2027 unless new formulations or indications are developed.
What strategic factors influence sales?
-
Pricing policies: Health insurers' formulary decisions impact sales volume.
-
Regulatory status: Pending biosimilar or generic approvals could accelerate share erosion.
-
Pipeline developments: New indications for hypertensive or cardiovascular risk reduction could sustain sales.
Summary of sales projections
| Year |
Estimated Global Sales (USD billion) |
Remarks |
| 2023 |
1.1 |
Post-patent impact begins |
| 2024 |
1.15 |
Generics stabilize sales |
| 2025 |
1.2 |
Market expansion in emerging nations |
| 2026 |
1.25 |
Price erosion persists |
| 2027 |
1.3 |
New formulations or indications |
Key Takeaways
-
The antihypertensive market grows modestly at 3.5% CAGR, but generic competition challenges branded products like LOTREL.
-
Sales peaked at USD 1.2 billion in 2021, with a projected slow increase to USD 1.3 billion by 2027.
-
Patent expiry and generics will cause significant revenue declines unless offset by pipeline innovations or expanded indications.
-
Market share is likely to decrease from 35% to 25-30%.
-
Emerging markets are vital to growth, offering volume increases despite price pressures.
FAQs
1. Will LOTREL regain market share with new formulations?
Potentially, if new formulations demonstrate improved adherence or efficacy; however, market penetration remains competitive.
2. How do generic versions affect sales?
Generic versions have significantly reduced branded sales since patent expiry, especially post-2022.
3. What are the main competitors?
Hyzaar (losartan with hydrochlorothiazide), valsartan combos, and alternative fixed-dose combinations.
4. Are there new indications for LOTREL?
Currently, no significant new indications; development focus is on hypertension and cardiovascular risk reduction.
5. How do emerging markets influence future sales?
They present growth opportunities, but pricing pressures are higher, and regulatory barriers exist.
References
[1] MarketResearch.com. (2022). Global Antihypertensive Drug Market Forecast.
[2] IQVIA. (2022). Prescription Data for Antihypertensive Therapy.
[3] RedBook. (2022). Wholesale Acquisition Prices for Cardiology Drugs.
[4] IMS Health. (2022). Cardiovascular Medication Sales Reports.
[5] EvaluatePharma. (2022). 2021 Top Drugs by Sales.