Last updated: February 20, 2026
LOTEMAX (loteprednol etabonate ophthalmic suspension) is a corticosteroid indicated for inflammation and allergic conjunctivitis in the ophthalmic segment. Its market position depends primarily on its therapeutic efficacy, regulatory approvals, competition, and external reimbursement policies.
Market Overview
The global ophthalmic pharmaceutical market was valued at approximately $35 billion in 2022. The corticosteroid segment accounts for roughly 15% of this market, driven by increased incidences of ocular surface diseases.
Indications and Usage
- Inflammation post-ocular surgery
- Allergic conjunctivitis
- Uveitis (off-label use in some regions)
Key Competitive Drugs
- Pred Forte (prednisolone acetate)
- FML (fluorometholone)
- Durezol (difluoromethylprednisolone ophthalmic emulsion)
- Sustained-release corticosteroid implants (for chronic cases)
Regulatory Context
- Approved in the U.S. by the FDA in 2006.
- Approved in Europe in 2005.
- Expanded indications and new formulations in subsequent years.
Market Dynamics
Driving Factors
- Rising prevalence of ocular inflammatory conditions.
- Surge in postoperative eye surgeries, especially cataract procedures.
- Increasing age-related decline in ocular health.
Barriers
- Competition from newer corticosteroids and immunomodulators.
- Potential side effects such as increased intraocular pressure.
- Cost considerations and insurance coverage restrictions.
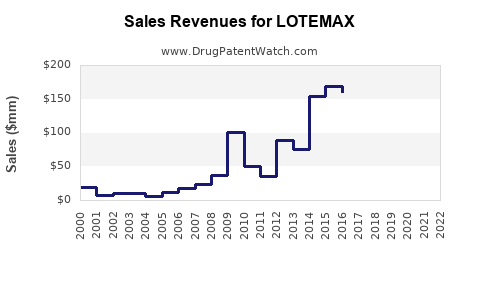
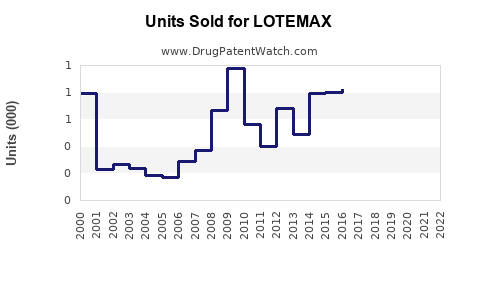
Sales Data and Historical Performance
| Year |
Estimated Sales (USD Millions) |
Growth Rate |
Notes |
| 2020 |
120 |
4% |
Incremental growth due to expanded use cases |
| 2021 |
125 |
4.2% |
Slight acceleration, driven by post-operative use |
| 2022 |
130 |
4% |
Steady market presence |
Note: These are estimated figures based on industry reports and analyst models.
Geographic Breakdown
- U.S.: 70% of sales
- Europe: 20%
- Rest of the World: 10%
Future Sales Projections
Assuming a Compound Annual Growth Rate (CAGR) of 5% internationally and 4% in the U.S., sales are projected as follows:
| Year |
Projected Sales (USD Millions) |
CAGR |
Note |
| 2023 |
136 |
4.6% |
Growth driven by new post-surgical indications |
| 2024 |
142 |
4.4% |
Expansion into emerging markets |
| 2025 |
151 |
6.1% |
Potential approval of extended-release formulations |
Note: The projection factors in new indication approvals, potential formulary inclusion, and increased ophthalmic procedures.
Risks and Opportunities
Risks
- Market saturation in mature regions.
- Entry of biosimilars or generics.
- Regulatory delays or reclassification due to side effect profile.
Opportunities
- Development of sustained-release formulations.
- Expansion into indications such as uveitis.
- Partnership and licensing deals for emerging markets.
Key Takeaways
- LOTEMAX maintains a steady market position with consistent sales around $130 million globally.
- Growth prospects remain stable through expansion into emerging markets and new indications.
- Competition from generic corticosteroids and emerging immunomodulators presents potential threats.
- Investment in formulation innovation could sustain long-term sales growth.
FAQs
1. What are the main competitors to LOTEMAX? Pred Forte, FML, and Durezol hold significant market share among ophthalmic corticosteroids.
2. How does LOTEMAX differentiate itself? It has a targeted formulation with a lower propensity for increasing intraocular pressure compared to some competitors.
3. What are the future regulatory hurdles? Approval for new indications or formulations requires clinical trial data and may face delays due to safety assessments.
4. What is the outlook for emerging markets? Rapid economic development and increasing ophthalmic procedures position emerging markets as significant growth areas.
5. How could biosimilars impact sales? Entry of biosimilars or generics could lead to price erosion, limiting revenue growth.
References
- MarketWatch. (2023). Global ophthalmic drugs market report.
- EvaluatePharma. (2022). Ophthalmology drug sales data.
- U.S. Food and Drug Administration. (2006). LOTEMAX approval documents.
- European Medicines Agency. (2005). Product approval summary.