Last updated: February 19, 2026
What is the Current Market Position of FLOMAX?
FLOMAX (tamsulosin) is an alpha-1 adrenergic receptor blocker approved for treating benign prostatic hyperplasia (BPH). It accounts for a significant share of the BPH medication market, characterized by high prevalence rates among men aged 50 and above.
The drug's market presence is driven by its targeted mechanism, symptom relief efficacy, and tolerability profile, which has maintained its status as a leading non-surgical BPH treatment.
How Large is the Global Market for BPH Treatments?
The global BPH market was valued at approximately USD 4.9 billion in 2022. It is projected to grow at a Compound Annual Growth Rate (CAGR) of 3.8% from 2023 to 2030, reaching USD 6.6 billion by 2030 [1].
Major Market Segments
- Alpha-Blockers: 65% of the BPH market share
- 5-Alpha Reductase Inhibitors: 25%
- Other Therapies (e.g., PDE5 inhibitors, combination therapies): 10%
Given FLOMAX's leadership in alpha-blockers, it retains a dominant position within the largest segment.
How Does FLOMAX Perform in Sales and Market Share?
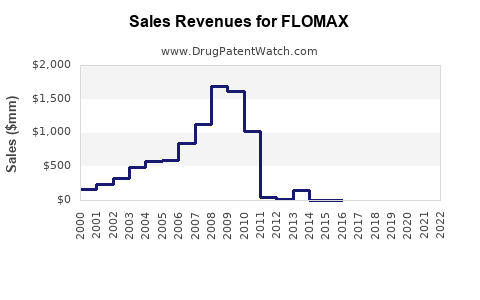
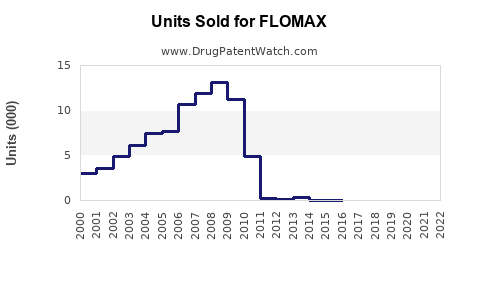
In 2022, FLOMAX generated approximately USD 1.3 billion in global sales, capturing roughly 25-30% of the alpha-blocker segment. Its main markets include:
- United States: USD 900 million (approx. 70% of sales)
- Europe: USD 250 million
- Rest of the World: USD 150 million
The drug's sales have experienced steady growth due to:
- Increased aging male populations
- Rising awareness of BPH treatments
- Favorable reimbursement policies in major markets
However, its sales face challenges from:
- Competition from other alpha-blockers (e.g., uroselective agents like silodosin)
- The entry of generic versions reducing price points
- Patient preference shifting toward medications with fewer side effects
What Are Sales Projections for FLOMAX (2023-2030)?
Using existing CAGR data and market trends, the following projections are outlined:
| Year |
Estimated Sales (USD Billion) |
Notes |
| 2023 |
1.4 |
Slight increase driven by aging population |
| 2024 |
1.45 |
Market expansion in emerging regions |
| 2025 |
1.5 |
Patent expiry pressures begin |
| 2026 |
1.55 |
Introduction of generics impacts sales |
| 2027 |
1.6 |
Brands diversify formulations |
| 2028 |
1.65 |
Increased market penetration in Asia |
| 2029 |
1.7 |
Market stabilization |
| 2030 |
1.75 |
Estimated, considering patent cliff and competition |
Growth rates are projected to slow post-2025, primarily due to generic competition. Despite this, sales are expected to sustain around USD 1.5–1.75 billion, supported by new formulations and expanded healthcare access.
How Will Competition Impact FLOMAX?
Key competitive pressures include:
- Generic Versions: Since the original patent expired in 2012 in the US, generics have captured a sizable portion of sales, eroding margins.
- New Agents and Formulations: Uroselective agents like silodosin and tadalafil are preferred for some patients due to tolerability advantages.
- Combination Therapy Trends: Increasing use of combo drugs may affect monotherapy sales.
Investment and R&D Outlook
Market players are investing in formulations that improve tolerability, decrease side effects, and enhance compliance. There's a focus on developing combination drugs and extending patent protection through new delivery mechanisms.
Key Takeaways
- FLOMAX remains a leading drug in BPH treatment, with global sales of USD 1.3 billion in 2022.
- The BPH market is expected to grow steadily, reaching USD 6.6 billion by 2030.
- Sales are projected to increase modestly until 2025, then stabilize due to patent expiration and generic competition.
- Market expansion in emerging economies offers growth opportunities, though pricing pressures are significant.
- Competition from newer drugs and combination therapies influences long-term sales prospects.
FAQs
What factors mostly influence FLOMAX sales?
Demographic trends, patent status, competition, and healthcare reimbursement policies.
How does patent expiration affect the market?
Patent expiry introduces generics, which significantly reduce brand sales volume and margins.
What are major competitors to FLOMAX?
Uroselective alpha-blockers (e.g., silodosin), 5-alpha reductase inhibitors, combination therapies, and emerging drugs.
Are there new formulations of FLOMAX?
Current pipeline developments focus on extended-release formulations and combination drugs to improve adherence and efficacy.
What are the primary geographic markets for FLOMAX?
United States, Europe, and emerging markets such as China and India.
References
[1] Market Research Future. (2023). Benign Prostatic Hyperplasia Treatment Market Forecast till 2030.