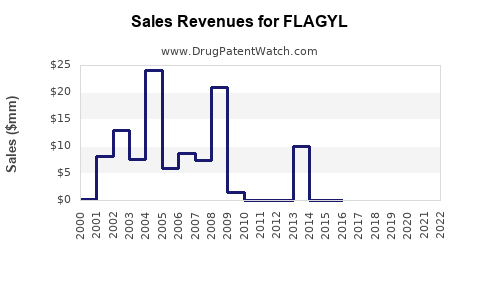
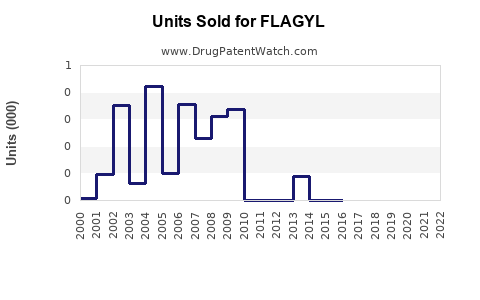
Last updated: February 15, 2026
Market Overview and Sales Projections for Flagyl (Metronidazole)
Flagyl, the brand name for metronidazole, is a widely prescribed antimicrobial agent. It targets anaerobic bacteria and protozoa, with applications spanning infectious diseases, gynecology, and gastrointestinal conditions. Market dynamics and future sales depend on evolving disease prevalence, resistance patterns, competition, and regulatory landscape.
Current Market Position
Flagyl remains a leading drug within the anti-infective segment. Its primary indications include bacterial vaginosis, anaerobic infections, and certain parasitic infections such as amebiasis. The drug is available in oral, injectable, and topical forms, enhancing its utility across treatment settings.
Global sales in 2022 are estimated to reach approximately $600 million, according to IQVIA data. The United States accounts for roughly 45% of sales, with Europe contributing 30%. The remaining sales are driven by markets in Asia-Pacific and Latin America.
Market Drivers
- Increasing Incidence of Infections: Rising cases of bacterial vaginosis, appendicitis, and parasitic diseases augment demand.
- Expanding Use in GI Disorders: Growing recognition of its efficacy against Clostridioides difficile infections.
- Rising Antimicrobial Use in Hospitals: Hospital-acquired infections boost use of broad-spectrum agents like Flagyl.
- Global Health Initiatives: Programs targeting parasitic diseases expand the market in developing countries.
Market Challenges
- Antibiotic Resistance: Emergence of resistant strains limits efficacy, prompting shifts to alternative therapies.
- Side-effect Profile: Gastrointestinal disturbances and metallic taste can affect adherence.
- Generic Competition: Patent expiration has released generic versions, reducing prices and profit margins.
Competitive Landscape
Generic metronidazole dominates sales, with multiple manufacturers supplying low-cost options. Brand-name Flagyl retains market share through clinician preference and formulation variations. Upcoming competition from new antibiotics targeting resistant strains could impact sales.
Future Sales Projections
2023-2027 Outlook
| Year |
Estimated Global Sales |
Key Drivers |
Potential Risks |
| 2023 |
$620 million |
Continued infection rates, generic sales volume |
Resistance development, market saturation |
| 2024 |
$640 million |
New indications, expanded use in GI disorders |
Market penetration hurdles |
| 2025 |
$660 million |
Increased focus on parasitic infections |
Competition from emergent agents |
| 2026 |
$680 million |
Adoption of combination therapies |
Regulatory delays |
| 2027 |
$700 million |
Global health initiatives targeting parasitic diseases |
Resistance, pricing pressures |
Projected compound annual growth rate (CAGR) approximates 2.5%. Growth is expected to slow as generic penetration stabilizes, but new indications and emerging markets can offset this slowdown.
Key Market Segments
- Hospital and Clinical Use: Accounts for 55% of sales due to broad-spectrum applications.
- Outpatient Prescriptions: Represents 35% with a focus on gynecological infections.
- Topical and Other Forms: Make up around 10%, used for skin infections and other localized conditions.
Regional Analysis
- United States: Largest market with stable growth driven by EUAs for resistant infections.
- Europe: Similar trends to the U.S.; regulatory developments impact prescribing patterns.
- Asia-Pacific: High growth potential due to parasitic disease prevalence, expanding healthcare infrastructure.
- Latin America: Growth driven by infectious disease burden, cost-effective generics.
Conclusion
Flagyl's market remains stable, with gradual growth driven by infection rates and expanding indications. Resistance and generic competition limit potential upside. Innovations in formulation and new therapeutic niches could influence future sales trajectories.
Key Takeaways
- Current global sales approximate $600 million, with slow growth projected.
- The drug maintains dominance due to broad-spectrum antimicrobial activity.
- Resistance and generic competition are primary challenges.
- Emerging markets and new applications offer growth opportunities.
- The drug’s market share hinges on efficacy against resistant strains and regulatory policies.
FAQs
1. How does resistance affect Flagyl sales?
Rising antimicrobial resistance reduces efficacy, leading clinicians to opt for alternative treatments, which can diminish Flagyl’s market share.
2. What new indications could expand Flagyl’s market?
Research into its utility against resistant bacterial infections and specific parasitic diseases may generate new prescribing opportunities.
3. How does generic competition influence pricing?
The introduction of multiple generic versions lowers prices, reducing revenue per unit but maintaining overall volume.
4. Which regions present the most growth potential?
Asia-Pacific and Latin America show high growth potential due to infectious disease prevalence and expanding healthcare access.
5. Are there any recent regulatory changes impacting Flagyl?
Regulatory agencies are monitoring resistance patterns, and approvals for new formulations or indications are considered case-specific.
References
[1] IQVIA, "Global Pharmaceutical Market Data," 2023.
[2] WHO, "Parasitic Disease Statistics," 2022.
[3] U.S. Food and Drug Administration, "Generic Drug Approvals," 2022.