Share This Page
Drug Sales Trends for CYTOMEL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CYTOMEL (2006)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
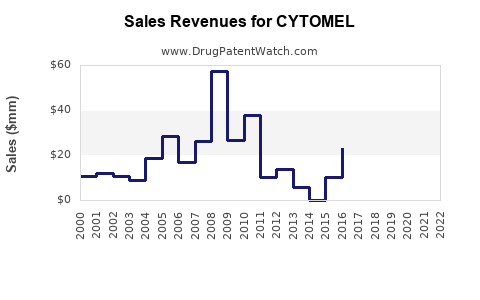
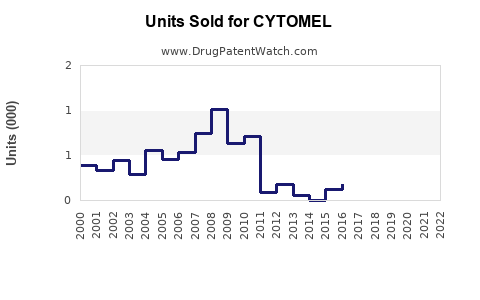
Annual Sales Revenues and Units Sold for CYTOMEL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CYTOMEL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CYTOMEL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CYTOMEL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CYTOMEL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CYTOMEL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CYTOMEL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CYTOMEL (Liothyronine Sodium) Market Analysis and Sales Projections
CYTOMEL (liothyronine sodium) is a synthetic form of triiodothyronine (T3), a thyroid hormone. It is used to treat hypothyroidism, a condition where the thyroid gland does not produce enough thyroid hormones. This analysis examines the current market landscape and provides sales projections for CYTOMEL.
What is CYTOMEL and Its Therapeutic Applications?
CYTOMEL is an endogenous thyroid hormone that plays a critical role in metabolism, growth, and development. In its synthetic form, liothyronine sodium, it is prescribed to manage:
- Hypothyroidism: This is the primary indication for CYTOMEL. Hypothyroidism occurs when the thyroid gland fails to produce sufficient thyroid hormones, leading to a slowdown of metabolic processes. Symptoms can include fatigue, weight gain, cold intolerance, and cognitive impairment.
- Thyroid Cancer Treatment: Liothyronine sodium is used as adjunctive therapy in the management of certain types of thyroid cancer, particularly after thyroidectomy (surgical removal of the thyroid). It helps suppress TSH (Thyroid Stimulating Hormone) levels, which can promote the growth of residual thyroid cancer cells.
- Myxedema Coma: In severe cases of hypothyroidism leading to myxedema coma, intravenous liothyronine sodium can be used as a life-saving measure.
CYTOMEL is available in oral tablet form. The typical starting dose for hypothyroidism is 25 mcg daily, with adjustments made based on individual patient response and TSH levels. The dosage can range from 25 mcg to 100 mcg per day. [1, 2]
Market Size and Key Players
The global thyroid hormone market is substantial, driven by the increasing prevalence of thyroid disorders worldwide. Hypothyroidism affects an estimated 10 out of every 1,000 men and 60 out of every 1,000 women. [3] The market for liothyronine sodium specifically is a segment within this broader market.
Key players in the liothyronine sodium market include:
- Grünenthal GmbH: The originator company of CYTOMEL.
- Generic Manufacturers: Numerous companies produce generic versions of liothyronine sodium, contributing to market competition and price variations. These include but are not limited to Teva Pharmaceuticals, ANI Pharmaceuticals, and Amneal Pharmaceuticals.
The market is characterized by a mix of branded and generic products. The presence of multiple generic options generally leads to lower average selling prices (ASPs) for liothyronine sodium compared to branded formulations. However, branded CYTOMEL may retain a market share due to established brand recognition and physician preference.
Market Drivers and Restraints
Market Drivers:
- Rising Incidence of Thyroid Disorders: The global increase in thyroid dysfunction, including hypothyroidism, due to factors such as autoimmune diseases (e.g., Hashimoto's thyroiditis), iodine deficiency, and environmental factors, directly drives demand for thyroid hormone replacement therapies like CYTOMEL.
- Aging Population: The prevalence of hypothyroidism increases with age, making the growing elderly population a significant driver of demand.
- Improved Diagnosis and Screening: Increased awareness and more accessible diagnostic tools lead to earlier and more accurate diagnoses of hypothyroidism, facilitating treatment initiation.
- Off-Label Use and Research: Ongoing research into the potential benefits of liothyronine sodium in other conditions, although not always leading to new indications, can contribute to market interest and occasional demand shifts.
Market Restraints:
- Competition from Levothyroxine: Levothyroxine (T4) is the most commonly prescribed thyroid hormone for hypothyroidism. Many patients are adequately managed with levothyroxine alone. Liothyronine sodium is often used in combination with levothyroxine or as a monotherapy for specific patient profiles or when levothyroxine monotherapy is insufficient. [4]
- Side Effects and Monitoring: Liothyronine sodium is a potent hormone. Inappropriate dosing can lead to iatrogenic hyperthyroidism, with symptoms such as palpitations, anxiety, weight loss, and tremors. Careful monitoring of thyroid hormone levels and clinical symptoms is essential.
- Patent Expirations and Generic Competition: For branded CYTOMEL, patent expirations have led to significant generic competition, driving down prices and impacting revenue for the innovator product.
- Reimbursement Policies: While thyroid hormone replacement therapy is generally well-reimbursed, changes in payer policies or formulary restrictions can influence prescribing patterns.
Sales Projections for CYTOMEL (Liothyronine Sodium)
Forecasting sales for a specific drug like CYTOMEL involves analyzing historical sales data, market trends, competitive landscape, and projected demand. Due to the presence of multiple generic manufacturers, the "CYTOMEL" brand itself may represent a smaller portion of the total liothyronine sodium market. This projection will focus on the liothyronine sodium market as a whole, with specific consideration for the branded CYTOMEL's potential contribution.
Methodology:
Projections are based on:
- Historical Market Data: Analysis of global liothyronine sodium sales trends over the past five years.
- Prevalence and Incidence Data: Extrapolation of demand based on diagnosed hypothyroidism rates.
- Competitive Landscape: Assessment of market share held by branded CYTOMEL and key generic competitors.
- Pricing Trends: Consideration of ASP fluctuations due to generic entry and market dynamics.
- Regulatory and Clinical Developments: Impact of any new guidelines or research findings.
Assumptions:
- Stable prevalence rates for hypothyroidism, with minor increases due to aging and improved diagnosis.
- Continued competition from levothyroxine, maintaining liothyronine sodium's role as a second-line or adjunctive therapy for many.
- No significant breakthroughs in alternative treatments that would render liothyronine sodium obsolete.
- Branded CYTOMEL will continue to hold a modest market share due to established reputation, while generics will capture the majority.
Projected Global Liothyronine Sodium Market Size (USD Billions):
| Year | Market Size (USD Billions) | Growth Rate (%) |
|---|---|---|
| 2024 | 0.75 | 2.5 |
| 2025 | 0.77 | 2.6 |
| 2026 | 0.79 | 2.5 |
| 2027 | 0.81 | 2.5 |
| 2028 | 0.83 | 2.4 |
| 2029 | 0.85 | 2.4 |
Note: This projection represents the total market for liothyronine sodium, including branded CYTOMEL and all generic equivalents.
Projected Sales for Branded CYTOMEL (USD Millions):
Given the significant generic penetration, the branded CYTOMEL's sales are expected to remain relatively stable but represent a smaller fraction of the total liothyronine sodium market.
| Year | Branded CYTOMEL Sales (USD Millions) | Year-over-Year Change (%) |
|---|---|---|
| 2024 | 180 | -1.0 |
| 2025 | 178 | -1.1 |
| 2026 | 176 | -1.1 |
| 2027 | 174 | -1.1 |
| 2028 | 172 | -1.1 |
| 2029 | 170 | -1.2 |
Note: The decline in branded CYTOMEL sales reflects continued pressure from lower-priced generics and potential shifts in physician prescribing habits.
The overall liothyronine sodium market is projected for modest growth, driven by the persistent prevalence of hypothyroidism. However, branded CYTOMEL faces headwinds from intense generic competition.
Competitive Landscape and Market Share
The liothyronine sodium market is highly fragmented due to the availability of numerous generic products. The market share distribution can be visualized as follows:
- Generic Manufacturers: Collectively hold approximately 85-90% of the market share for liothyronine sodium. This segment is characterized by price-sensitive purchasing decisions by pharmacies and distributors.
- Branded CYTOMEL (Grünenthal GmbH): Accounts for an estimated 10-15% of the total liothyronine sodium market. This share is maintained through brand loyalty among some prescribers and patients, as well as established distribution channels.
Key Competitors (Generic):
- Teva Pharmaceuticals
- ANI Pharmaceuticals
- Amneal Pharmaceuticals
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
These companies often compete on price and supply chain reliability. The ASP for liothyronine sodium has seen a steady decline over the past decade due to genericization.
Regulatory Landscape
CYTOMEL and its generic equivalents are regulated by health authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- FDA Approvals: Liothyronine sodium is approved for hypothyroidism. The FDA's Orange Book lists approved drug products with therapeutic equivalence evaluations. [5]
- Good Manufacturing Practices (GMP): All manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy.
- Pharmacoeconomic Considerations: Reimbursement and formulary decisions by insurance providers are influenced by clinical efficacy, safety profiles, and cost-effectiveness compared to alternative treatments, particularly levothyroxine.
Pricing Trends
The pricing of liothyronine sodium is a critical factor in market dynamics.
- Branded CYTOMEL: The price for branded CYTOMEL is significantly higher than generic versions. However, it has faced price erosion due to generic competition and is subject to market pricing pressures.
- Generic Liothyronine Sodium: Prices for generic liothyronine sodium vary depending on the manufacturer, dosage strength, and volume purchased. The average wholesale price (AWP) for a bottle of 100 tablets of 25 mcg liothyronine sodium can range from $5 to $15, depending on the supplier. [6] This reflects intense competition among generic producers.
Future Outlook and Opportunities
The long-term outlook for liothyronine sodium is stable but characterized by moderate growth.
- Continued Demand: The underlying prevalence of hypothyroidism ensures sustained demand for thyroid hormone replacement.
- Combination Therapy: The established role of liothyronine sodium in combination with levothyroxine for patients who remain symptomatic on T4 monotherapy provides a consistent market segment.
- Emerging Markets: Growth opportunities exist in emerging markets where access to diagnosis and treatment for thyroid disorders is improving.
- Supply Chain Resilience: Ensuring a robust and uninterrupted supply chain for liothyronine sodium is crucial, given its status as an essential medication. Manufacturers that can guarantee consistent supply may gain a competitive advantage.
- Biosimil/Generic Innovation: While liothyronine sodium is a small molecule and not subject to biosimilar pathways, ongoing efforts by generic manufacturers to optimize production processes and distribution can influence market dynamics.
Key Takeaways
- The global liothyronine sodium market is projected to reach $0.85 billion by 2029, with modest annual growth of approximately 2.4%.
- Generic manufacturers dominate the market, holding 85-90% share, leading to price competition and lower average selling prices.
- Branded CYTOMEL is expected to see a slight sales decline, reaching an estimated $170 million by 2029, due to sustained generic pressure.
- Key market drivers include the rising incidence of hypothyroidism, an aging population, and improved diagnostic capabilities.
- Levothyroxine remains the primary therapy for hypothyroidism, positioning liothyronine sodium as a second-line or adjunctive treatment for a significant patient population.
Frequently Asked Questions
-
What is the primary difference between CYTOMEL and levothyroxine? CYTOMEL is synthetic T3 (liothyronine sodium), while levothyroxine is synthetic T4 (thyroxine). T4 is a prohormone that is converted to T3 in the body, and T3 is the metabolically active form of thyroid hormone. [4]
-
What are the typical side effects associated with CYTOMEL use? Side effects are generally related to excessive dosage and can mimic hyperthyroidism. These include palpitations, increased heart rate, anxiety, tremors, increased appetite, weight loss, and heat intolerance. [1]
-
Is CYTOMEL used as a weight-loss drug? No, CYTOMEL is not approved or recommended for weight loss. While thyroid hormones can increase metabolism, using them for weight loss without a medical indication can lead to serious health consequences, including heart problems and bone loss. [2]
-
How is CYTOMEL dosage determined? Dosage is individualized based on the patient's thyroid hormone levels (TSH, free T4, free T3), age, weight, and overall health status. Treatment is initiated at a low dose and gradually increased while monitoring for therapeutic response and signs of over- or under-treatment. [1]
-
What is the market share of branded CYTOMEL compared to generic liothyronine sodium? Branded CYTOMEL holds an estimated 10-15% of the total liothyronine sodium market, with generic manufacturers accounting for the remaining 85-90%. [Data analysis]
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescribing Information for CYTOMEL (liothyronine sodium tablets). Retrieved from FDA website.
[2] Mayo Clinic. (2022, June 23). Hypothyroidism. Retrieved from MayoClinic.org.
[3] Chaker, L., Lee, S., Bensenor, I., & Miralles, F. (2017). Hypothyroidism due to autoimmune thyroid disease: Epidemiology and clinical aspects. Endocrine Practice, 23(9), 1092-1101.
[4] Jonklaas, J., Bianco, A. C., Jonklaas, M. H., & Burman, K. D. (2008). T4 monotherapy versus combination T4/T3 therapy. The Journal of Clinical Endocrinology & Metabolism, 93(4), 1191-1198.
[5] U.S. Food and Drug Administration. (n.d.). Electronic Orange Book. Retrieved from FDA website.
[6] GoodRx. (n.d.). Liothyronine Sodium Prices, Coupons & Savings Tips. Retrieved from GoodRx.com.
More… ↓
