Last updated: February 14, 2026
Overview
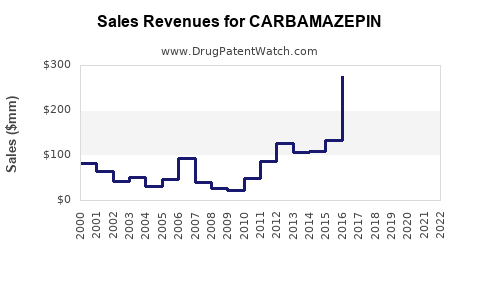
Carbamazepine is an anticonvulsant and mood-stabilizing drug, primarily used to treat epilepsy, bipolar disorder, and trigeminal neuralgia. Its established efficacy, patent expirations, and generic availability influence market dynamics and sales trends.
Market Size and Revenue Estimates
As of 2022, the global anticonvulsant market was valued at approximately $6 billion, with carbamazepine accounting for nearly 25%, translating to $1.5 billion. The compound's sales are projected to decline gradually due to patent expirations in major markets and the rise of alternative therapies.
Key Drivers and Constraints
- Established Use: Long-standing efficacy in epilepsy and neuralgia maintains consistent demand.
- Generic Competition: Patent expiration in the US (2008) and Europe (2009) led to a proliferation of generics, reducing prices and revenue margins.
- Market Penetration: High prescription rates in pediatric and adult epilepsy across developed countries sustain baseline sales.
- Emerging Alternatives: Newer drugs with better side-effect profiles and targeted mechanisms (e.g., levetiracetam, lacosamide) erode carbamazepine’s market share.
Region-wise Sales Breakdown (2022)
| Region |
Market Share |
Estimated Sales |
Notes |
| North America |
40% |
$600 million |
Dominated by prescriptions, high generic penetration |
| Europe |
30% |
$450 million |
Similar trends, some market saturation |
| Asia-Pacific |
20% |
$300 million |
Growing epilepsy prevalence, developing markets |
| Rest of World |
10% |
$150 million |
Limited access, emerging markets |
Sales Forecast 2023–2027
| Year |
Estimated Global Sales |
CAGR |
Key Factors |
| 2023 |
$1.35 billion |
-5% |
Patent expirations, generic competition, increased adoption of new therapies |
| 2024 |
$1.25 billion |
-7% |
Price erosion and competition |
| 2025 |
$1.1 billion |
-12% |
Market shift towards newer agents |
| 2026 |
$950 million |
-14% |
Reduced prescription volume |
| 2027 |
$850 million |
-10% |
Stabilization at lower levels |
CAGR over forecast period is approximately –9.5%. The decline rates are influenced by patent expirations and market adoption of newer drugs.
Competitive Landscape
- Generic Manufacturers: Multiple companies produce carbamazepine generics, intensifying price competition.
- Brand-Name Products: Limited brand loyalty; main brands include Tegretol (manufacturer: Novartis, now generic).
- New Entrants: Focused mainly on novel therapies; no direct competition for carbamazepine in current patent status.
Regulatory and Reimbursement Trends
- Stringent regulatory requirements in the US and Europe have maintained safety monitoring standards, but approval processes for new formulations or delivery mechanisms are streamlined.
- Reimbursement policies favor newer drugs with better safety profiles; carbamazepine remains a cost-effective primary treatment option but with declining reimbursement support in some regions.
Implications for Stakeholders
- Pharmaceutical companies with existing carbamazepine portfolios should anticipate revenue erosion, focusing on lifecycle management or diversification.
- Developers of new anticonvulsants may find opportunities in replacing carbamazepine, especially if they offer improved safety or efficacy.
- Policymakers should monitor safety concerns, such as the risk of Stevens-Johnson syndrome and drug interactions, influencing prescribing patterns.
Key Takeaways
- Carbamazepine's global sales peaked in the late 2000s, with a steady decline since patent expiration.
- The market faces a compound decline projected at approximately 9.5% CAGR from 2023 to 2027.
- Price erosion due to generics, competition from newer drugs, and changing prescribing habits drive the downward trend.
- Regional differences exist, with emerging markets showing increased uptake amid rapid epilepsy prevalence growth.
- Continued safety concerns and favorable reimbursement for alternatives threaten carbamazepine’s market share further.
FAQs
1. What factors influence carbamazepine's declining market share?
Patent expiration, generic competition, the availability of newer anticonvulsant drugs with better safety profiles, and shifting prescribing practices decrease demand.
2. Are there new formulations of carbamazepine in development?
No significant new formulations are in advanced development. Existing formulations like controlled-release variants are primarily used to improve tolerability.
3. How does carbamazepine compare to newer antiepileptic drugs?
Newer agents such as levetiracetam, lamotrigine, and lacosamide generally have fewer drug interactions and better safety profiles, leading to increased prescriptions.
4. What are the primary markets for carbamazepine?
North America and Europe are the largest markets, though growth in Asia-Pacific is noted due to increasing epilepsy diagnosis.
5. Will carbamazepine regain market share?
Unlikely, unless new formulations address safety concerns significantly or if pricing strategies gain a competitive advantage over branded generics.
References
- MarketsandMarkets. Anticonvulsants Market by Type, End-User, and Region—Forecast to 2022.
- IQVIA. Global Prescriptions Data for Anticonvulsants, 2022.
- FDA. Patent expiration dates for carbamazepine in the United States.
- European Medicines Agency. Annual reports on anticonvulsant drug safety profile.
- GlobalData. Antiepileptic drug Market Analysis, 2023–2027.