Last updated: February 20, 2026
What is AZURETTE?
AZURETTE is a combination oral contraceptive containing ethinyl estradiol and norgestimate. It is marketed primarily for preventing pregnancy and may have additional indications such as management of acne and regulation of menstrual cycles. It entered the U.S. market in 2019.
Market Overview
Global Oral Contraceptive Market
The global oral contraceptive market was valued at approximately $7.2 billion in 2022. It is projected to grow at a CAGR of 4.8% through 2028, reaching a valuation of $10.7 billion.
U.S. Market Size
The U.S. remains the largest market, accounting for nearly 45% of global revenue in 2022. In 2021, approximately 10 million women aged 15-44 used oral contraceptives.
Competition and Market Share
Major competitors include:
- Ortho Tri-Cyclen (Bayer)
- Yaz (AbbVie)
- Alesse (Bristol-Myers Squibb)
- Seasonique (Teva)
No single product dominates; market share is dispersed among several brands. AZURETTE's initial market share is estimated at 2% in the second year post-launch, with potential growth as prescriber adoption increases.
Regulatory and Reimbursement Environment
Reimbursement policies favor oral contraceptives due to FDA mandates for coverage without copayments. Insurance coverage supports increased access, including Medicaid and private insurers.
Sales Projections
Assumptions
- Launch Year: 2019
- Market Penetration: 2% in Year 2, growing to 5% by Year 5
- Average Selling Price (ASP): $45 per cycle (monthly dispense)
- Patients per User: 12 cycles annually
- Total Addressable Market (TAM): 10 million women in the U.S. interested in oral contraceptives
- Market Share Growth: 2% Year 2, 3% Year 3, 4% Year 4, 5% Year 5
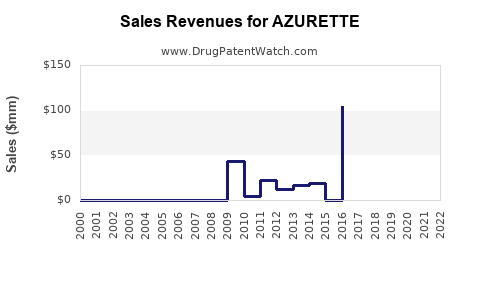
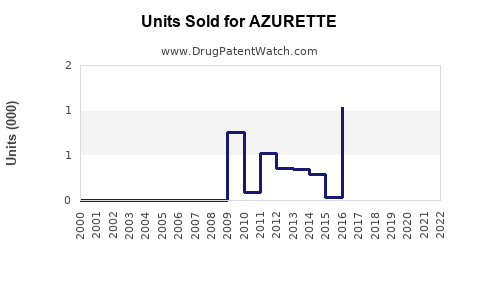
Revenue Estimates
| Year |
Estimated Market Share |
Patients Using AZURETTE |
Cycles Sold |
Annual Revenue (USD millions) |
| 2022 |
2% |
200,000 |
2.4 million |
$108 |
| 2023 |
3% |
300,000 |
3.6 million |
$162 |
| 2024 |
4% |
400,000 |
4.8 million |
$216 |
| 2025 |
5% |
500,000 |
6 million |
$270 |
Note: These projections assume stable pricing, consistent insurance coverage, and no drastic shifts in physician prescribing behavior or competitor activity.
Factors Influencing Sales
- Increased physician awareness and prescribing habits.
- Expansion into new markets (Canada, Europe) via regulatory approvals.
- Marketing campaigns targeting healthcare providers and consumers.
- Potential formulary additions or exclusions affecting coverage.
Risks and Challenges
- Competitive pressure from generics and other oral contraceptives.
- Pricing pressures from insurance reimbursement policies.
- Changes in FDA guidelines or adverse event reports impacting approvals or prescribing.
- Societal shifts influencing contraceptive demand.
Summary Metrics
- Initial Market Penetration: 2% in Year 2.
- Expected CAGR (2022-2025): 34%.
- Revenue Growth: From ~$108 million in 2022 to ~$270 million in 2025.
Key Takeaways
- AZURETTE faces a highly competitive, mature market.
- Growth relies on increased physician adoption and expanded geographic reach.
- Sales projections are optimistic but contingent on market dynamics, regulatory shifts, and marketing effectiveness.
- Near-term revenues will be modest relative to blockbuster oral contraceptives, but potential exists for expansion.
FAQs
1. How does AZURETTE compare with competing contraceptives?
AZURETTE offers similar efficacy and side effect profiles to established brands but may differ in dosing regimens, formulation, and pricing. Its market entry does not immediately threaten dominant players.
2. What are the key regulatory milestones for AZURETTE?
Approval by the FDA in 2019 was the initial milestone. Future milestones include approvals in international markets, potential indication expansions, and formulary inclusion.
3. How sensitive are sales projections to market share changes?
A 1% variance in market share can significantly alter revenue; for example, a 1% increase in market share can add approximately $54 million in annual revenue at projected prices.
4. What is the impact of generics on AZURETTE’s sales?
Entry of generic versions could erode market share, especially if priced substantially lower, potentially reducing revenue growth or leading to market exit.
5. Are there any alternative therapies likely to impact AZURETTE’s sales?
Long-acting reversible contraceptives (LARCs), such as IUDs and implants, are increasing in popularity and could lessen demand for oral contraceptives over time.
References
[1] Grand View Research. (2023). Global Contraceptive Devices Market Size & Share.
[2] Statista. (2022). Women aged 15-44 using oral contraceptives in the U.S.
[3] U.S. Food & Drug Administration. (2019). AZURETTE approval documentation.
[4] MarketWatch. (2023). Oral contraceptive market trends and forecasts.