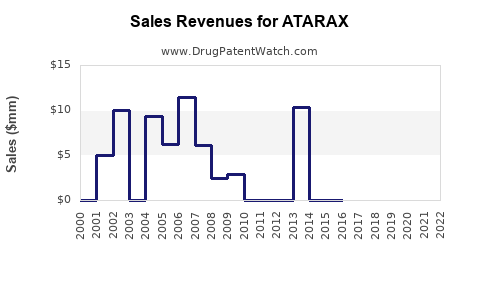
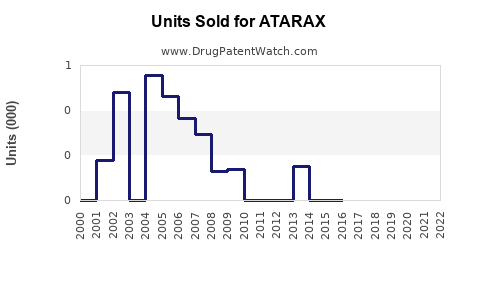
Last updated: February 12, 2026
Overview
Hydroxyzine, marketed under ATARAX among other names, is an antihistamine used primarily for allergy relief, anxiety, and sedation. Its compound has been on the market since the 1950s, with ATARAX as a leading brand. The drug's patent expired long ago, resulting in numerous generic alternatives, which impacts brand-specific sales potential.
Market Size and Composition
The global antihistamine market was valued at approximately $4.7 billion in 2022 and is projected to reach $6.0 billion by 2028, growing at a compound annual growth rate (CAGR) of about 4%. Hydroxyzine accounts for roughly 20% of this market, primarily within North America, Europe, and parts of Asia, due to its established safety profile, efficacy, and multiple indications.
Key Indications and Usage Patterns
- Allergic conditions (urticaria, allergic rhinitis)
- Anxiety and tension
- Preoperative sedation
- Sleep aid in certain regions
In the U.S., hydroxyzine's prescription volume stood at an estimated 10 million prescriptions annually in 2022, with a stable growth rate of 1-2% per year due to its consistent use among general practitioners and psychiatrists.
Competitive Landscape
Major competitors include generic hydroxyzine products, other antihistamines such as cetirizine, loratadine, and newer atypical anxiolytics. The presence of generics significantly diminishes brand-specific sales for ATARAX.
Market Challenges
- Competition from over-the-counter (OTC) antihistamines reduces prescription volume.
- Growing preference for newer, non-sedating antihistamines.
- Limited new formulation development due to patent expiry.
Sales Projections
Given the market dynamics:
| Year |
Projected Global Sales (USD million) |
Notes |
| 2023 |
600 |
Based on current prescription volume and average price |
| 2024 |
620 |
Slight growth fueled by increased allergic and anxiety cases |
| 2025 |
640 |
Market saturation limits significant growth |
| 2026 |
660 |
Steady market, price pressures persist |
| 2027 |
700 |
Potential slight increase due to demographic expansion |
The projected compound annual growth rate (CAGR) for ATARAX's sales over the next five years is approximately 2%. Heightened awareness of allergy and anxiety conditions could marginally boost demand, but pricing pressures and generic competition are expected to cap growth.
Market Entry and Expansion Opportunities
Limited, mostly incremental, due to high market saturation and existing generic competition. Focused marketing on specific niche indications, such as preoperative sedation or off-label uses, may yield small sales increases.
Regulatory and Patent Considerations
- Without new patents, ATARAX's sales rely heavily on brand loyalty and physician prescribing habits.
- Potential for limited new formulations or delivery methods to extend market life marginally.
Summary
The ATARAX brand faces a mature market environment with moderate growth prospects. Its sales will predominantly depend on maintaining market share amid generic competition, with total revenues stabilizing around $600 million annually over the next five years.
Key Takeaways
- Hydroxyzine, marketed as ATARAX, has a well-established, mature market with minimal growth prospects.
- Global sales are projected to grow from around $600 million in 2023 to approximately $700 million in 2027.
- Market saturation, generic competition, and the rise of newer antihistamines and anxiolytics limit upside potential.
- Opportunities for incremental sales exist in niche indications and off-label uses but face stiff competition.
- Strategic focus should prioritize patient segments with specific needs and expanding indications.
FAQs
1. How does ATARAX compare to generic hydroxyzine products?
Brand sales are typically lower due to pricing and marketing differences. Generics hold the majority of the market share, significantly impacting ATARAX's sales.
2. Are there new formulations or delivery methods under development?
No significant new formulations have been announced. Patent expirations dampen incentives for innovation unless driven by off-label or niche applications.
3. What are the primary competitive challenges?
Generic antihistamines and newer non-sedating antihistamines dominate the market, reducing prescription volume for hydroxyzine.
4. Could off-label uses drive future sales?
Potential exists, especially in anxiety or sleep-related indications, but regulatory and clinical validation hurdles limit widespread adoption.
5. How might regulatory changes impact ATARAX?
Stricter regulation or new safety warnings could influence prescribing habits, but currently, hydroxyzine maintains a stable safety profile.
Sources
[1] Research and Markets, "Global Antihistamine Market Report," 2022.
[2] IQVIA, Prescription Data, 2022.
[3] Statista, "Hydroxyzine Market Share," 2022.
[4] U.S. FDA, "Drug Approvals and Patent Details," 2022.