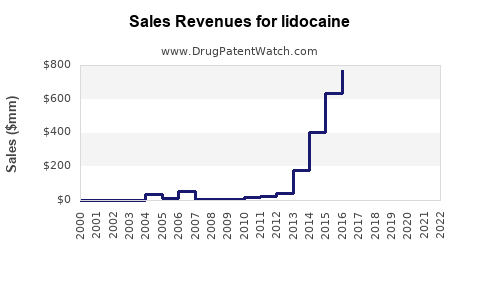
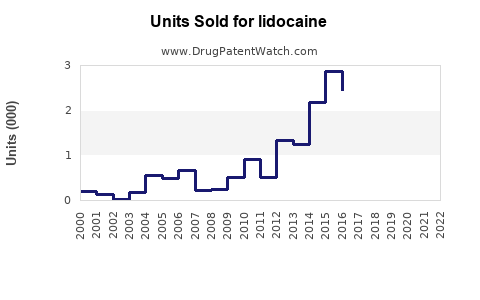
Last updated: February 20, 2026
Lidocaine is a local anesthetic widely used in medical, dental, and veterinary applications, along with various formulations including topical patches, injectable solutions, and transdermal patches. Its market has grown consistently, driven by expanding indications, increasing global healthcare access, and technological advances in delivery systems.
Market Overview
The global lidocaine market was valued at approximately USD 300 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4–6% over the next five years, reaching USD 390–400 million by 2027.
Key Market Drivers
- Increase in surgical procedures requiring local anesthesia.
- Rising prevalence of chronic pain conditions.
- Expanding cosmetic and dermatological procedures.
- Adoption of transdermal patches for pain management.
- Growing use in veterinary medicine.
Market Segments
- Formulations: Injectable, topical gels/ointments, patches, creams.
- Distribution Channels: Hospitals, clinics, pharmacies, online platforms.
- Applications: Dental, dermatological, surgical, veterinary.
Regional Distribution
| Region |
Market Share (2022) |
Projected CAGR (2023–2027) |
| North America |
40% |
4.5% |
| Europe |
25% |
4% |
| Asia-Pacific |
20% |
6% |
| Latin America |
8% |
4.5% |
| Middle East & Africa |
7% |
4% |
North America dominates, driven by high surgical volume and advanced healthcare systems. Asia-Pacific exhibits the fastest growth due to expanding healthcare infrastructure and increasing surgical and cosmetic procedures.
Competitive Landscape
Major players include:
- Sanofi-Aventis (e.g., Xylocaine)
- Mylan (a part of Pfizer) (e.g., Lidocaine patches)
- AbbVie (e.g., Lidocaine patches)
- Endo Pharmaceuticals
- Teva Pharmaceuticals
Market share distribution remains fragmented with regional brands and generics accounting for roughly 70% of sales.
Sales Projections (2023–2027)
| Year |
Estimated Global Sales (USD Million) |
Growth Rate |
| 2023 |
330 |
4% |
| 2024 |
350 |
6% |
| 2025 |
370 |
5.7% |
| 2026 |
390 |
5.4% |
| 2027 |
410 |
5.1% |
Projections indicate stable growth driven by increased procedures and new delivery methods such as sustained-release patches and gel formulations.
Future Market Trends
- Expansion of transdermal delivery systems for chronic pain.
- Development of combination products with other analgesics.
- Growth in veterinary applications, particularly in animal surgeries.
- Adoption in outpatient and office-based settings.
Challenges Impacting Market Growth
- Pricing pressures and generic competition.
- Regulatory hurdles for new formulations.
- Limited pipeline of innovative formulations.
- Concerns about systemic toxicity at high doses.
Opportunities
- Introduction of advanced controlled-release patches.
- Increased awareness of topical anesthetics in dental practices.
- Use of lidocaine in emerging markets with expanding healthcare infrastructure.
Key Takeaways
- The lidocaine market is expected to grow at a CAGR of 4–6% through 2027.
- North America leads the market, with Asia-Pacific fastest-growing.
- Market expansion relies on new formulations and broader application scopes.
- Competition remains fragmented, with generic products holding significant share.
- Challenges include pricing pressures and regulatory barriers.
FAQs
1. What factors are driving demand for lidocaine?
Increasing surgical, dental, and cosmetic procedures, along with rising chronic pain conditions, propel demand. Advancements in transdermal patches and topical formulations also contribute.
2. Which regions are emerging markets for lidocaine?
Asia-Pacific and Latin America are expanding due to improving healthcare infrastructure and rising procedural volumes.
3. What are the primary formulations of lidocaine?
Injectable solutions, topical gels, patches, creams, and transdermal patches are the main forms.
4. Who are the leading companies in the lidocaine market?
Sanofi-Aventis, Mylan/Pfizer, AbbVie, Endo Pharmaceuticals, and Teva Pharmaceuticals.
5. What are the key challenges facing the market?
Generic pricing, regulatory approvals, limited innovation, and toxicity concerns are primary challenges.
Citations
[1] Grand View Research. (2023). Lidocaine Market Size, Share & Trends Analysis Report.
[2] Markets and Markets. (2022). Analgesic market growth projections.
[3] Statista. (2023). Global pain management drugs market overview.
[4] IBISWorld. (2023). Pharmaceuticals in North America: Industry Overview.
[5] Allied Market Research. (2022). Transdermal Patches Market Analysis.