Share This Page
Drug Sales Trends for hydrocortisone
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for hydrocortisone (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
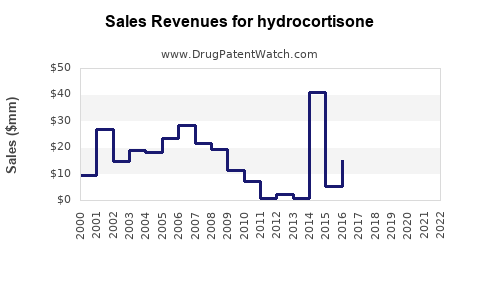
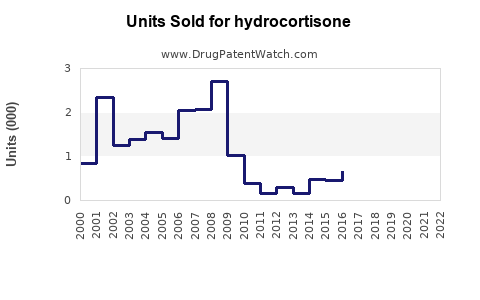
Annual Sales Revenues and Units Sold for hydrocortisone
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HYDROCORTISONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HYDROCORTISONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HYDROCORTISONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HYDROCORTISONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HYDROCORTISONE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| HYDROCORTISONE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Hydrocortisone Market Analysis and Sales Projections
Hydrocortisone, a corticosteroid, is a critical therapeutic agent used to treat a wide range of inflammatory and allergic conditions. Its market performance is influenced by the prevalence of these conditions, global healthcare expenditure, and the competitive landscape of topical and systemic treatments. This analysis projects market growth based on current trends and anticipated future developments.
What is the Current Global Market Size for Hydrocortisone?
The global hydrocortisone market was valued at approximately $1.1 billion in 2023. The market is segmented by product type (prescription and over-the-counter [OTC]), route of administration (topical, oral, injectable, rectal), and end-user (hospitals, clinics, pharmacies, homecare). The topical segment accounts for the largest share due to its widespread use in dermatology.
Key Market Data (2023):
- Total Market Value: $1.1 billion
- Projected Compound Annual Growth Rate (CAGR) (2024-2030): 4.2%
- Largest Segment by Product Type: Prescription
- Largest Segment by Route of Administration: Topical
- Key Therapeutic Areas: Dermatology, Respiratory Diseases, Rheumatology, Gastroenterology
What are the Primary Drivers of Hydrocortisone Market Growth?
The increasing incidence of inflammatory and allergic diseases, such as eczema, psoriasis, asthma, and inflammatory bowel disease (IBD), is a primary driver for hydrocortisone demand. Growing awareness of these conditions and improved diagnostic capabilities contribute to higher prescription rates.
Furthermore, the expansion of healthcare infrastructure in emerging economies and increased healthcare spending facilitate broader access to medications like hydrocortisone. The availability of hydrocortisone as an OTC product for milder skin conditions also supports market volume.
Market Drivers:
- Rising prevalence of inflammatory and allergic conditions:
- Eczema (Atopic Dermatitis): Global prevalence estimated between 10-20% in children and 1-3% in adults.
- Psoriasis: Affects approximately 3% of the global population.
- Asthma: Over 300 million people worldwide suffer from asthma.
- Inflammatory Bowel Disease (IBD): Affects over 1.5 million Americans and millions more globally.
- Increasing healthcare expenditure: Global healthcare spending reached an estimated $10 trillion in 2022.
- Growing demand for dermatological treatments: The global dermatology market is projected to grow significantly.
- Availability of OTC formulations: Facilitates self-treatment for common skin ailments.
- Aging global population: Older individuals are more susceptible to certain chronic inflammatory conditions.
What are the Key Restraints and Challenges in the Hydrocortisone Market?
Despite its therapeutic benefits, the hydrocortisone market faces challenges related to the side effects associated with long-term or high-dose corticosteroid use, including adrenal suppression, Cushing's syndrome, and immunosuppression. This drives research into alternative treatments and the judicious use of corticosteroids.
The patent expiry of branded hydrocortisone products has led to increased generic competition, putting downward pressure on prices for certain formulations. Additionally, stringent regulatory approvals for new drug delivery systems or formulations can impact market entry timelines.
Market Restraints and Challenges:
- Adverse side effects of corticosteroids:
- Systemic effects (long-term use): Osteoporosis, hypertension, diabetes mellitus, growth retardation in children.
- Topical effects: Skin thinning, striae, telangiectasias.
- Development of alternative therapies: Biologics and other immunomodulatory drugs offer new treatment avenues for chronic inflammatory diseases, potentially reducing reliance on corticosteroids.
- Price erosion due to generic competition: Over 80% of hydrocortisone products are available as generics.
- Regulatory hurdles for novel formulations: The approval process for new drug delivery systems can be lengthy and costly.
- Environmental concerns related to pharmaceutical waste: Growing awareness and regulations regarding pharmaceutical disposal.
What is the Competitive Landscape for Hydrocortisone Products?
The hydrocortisone market is characterized by a fragmented competitive landscape, with numerous global and regional players. Major pharmaceutical companies and generic manufacturers compete on product quality, pricing, and distribution networks. The market includes both originator brands and a substantial number of generic alternatives.
Major Market Players (Examples):
- Bayer AG: Offers a range of topical hydrocortisone products.
- GlaxoSmithKline plc: Holds significant market share in respiratory and dermatological indications.
- Pfizer Inc.: Provides various formulations for systemic and topical use.
- Sanofi S.A.: Active in the treatment of allergic and inflammatory conditions.
- Teva Pharmaceutical Industries Ltd.: A leading producer of generic hydrocortisone.
- Viatris Inc.: Offers a broad portfolio of generic pharmaceuticals.
Competitive Strategies:
- Product Differentiation: Development of novel delivery systems (e.g., extended-release formulations, advanced topical carriers).
- Geographic Expansion: Targeting emerging markets with increasing healthcare access.
- Cost Leadership: Focus on efficient manufacturing and supply chain management for generic products.
- Strategic Partnerships and Acquisitions: Expanding product portfolios and market reach.
What are the Projected Sales for Hydrocortisone in Key Regions?
North America currently dominates the hydrocortisone market, driven by high healthcare spending, advanced medical infrastructure, and a high prevalence of chronic inflammatory diseases. The Asia-Pacific region is expected to exhibit the fastest growth due to increasing disposable incomes, improving healthcare access, and a large patient population. Europe remains a significant market, with a well-established healthcare system and a strong demand for dermatological and respiratory treatments.
Regional Market Share (2023 Estimate):
- North America: 35%
- Europe: 28%
- Asia-Pacific: 20%
- Latin America: 10%
- Middle East & Africa: 7%
Projected Regional Growth (CAGR 2024-2030):
- Asia-Pacific: 5.5%
- North America: 3.8%
- Europe: 3.9%
- Latin America: 4.5%
- Middle East & Africa: 4.8%
What is the Future Outlook and Sales Projection for the Hydrocortisone Market?
The global hydrocortisone market is projected to reach approximately $1.4 billion by 2030, growing at a CAGR of 4.2% from 2024. Growth will be sustained by the persistent need for effective anti-inflammatory treatments, particularly in dermatology and respiratory care. Innovation in drug delivery systems and formulations, along with expanding access to healthcare in developing regions, will further contribute to market expansion.
The increasing focus on personalized medicine and the development of targeted therapies for specific inflammatory pathways may present competition, but the established efficacy, affordability, and broad spectrum of activity of hydrocortisone ensure its continued relevance. The market will likely see continued growth in topical OTC applications and a steady demand for systemic formulations in chronic disease management.
Sales Projection:
- 2024 Estimated Market Value: $1.14 billion
- 2030 Projected Market Value: $1.45 billion
- CAGR (2024-2030): 4.2%
Factors Influencing Future Growth:
- Technological advancements in drug delivery:
- Nanoparticle formulations for improved topical penetration.
- Controlled-release injectable formulations for extended systemic effects.
- Expanding indications for use: Research into hydrocortisone's role in less common inflammatory conditions.
- Demographic shifts: Aging populations with higher rates of chronic diseases.
- Global economic recovery: Increased healthcare spending capacity in various regions.
Key Takeaways
The hydrocortisone market is a mature yet growing sector driven by the persistent prevalence of inflammatory and allergic diseases. While facing competition from newer therapies and challenges related to side effects, its affordability, broad efficacy, and accessibility as both prescription and OTC products ensure continued demand. North America leads the market, with Asia-Pacific poised for the fastest growth. Projections indicate a steady CAGR of 4.2%, reaching approximately $1.45 billion by 2030.
Frequently Asked Questions
-
What is the primary indication driving current hydrocortisone sales? Dermatological conditions, such as eczema and psoriasis, represent the largest segment of hydrocortisone sales due to its topical application.
-
How will the rise of biologic therapies impact the hydrocortisone market? Biologics offer targeted treatments for specific inflammatory pathways. While they may reduce hydrocortisone use in severe cases of conditions like rheumatoid arthritis or IBD, hydrocortisone remains a cost-effective first-line or adjunctive therapy for many inflammatory and allergic conditions.
-
Are there significant patent expirations anticipated for major hydrocortisone products in the near future? Most major hydrocortisone products have long since gone off-patent, leading to a highly genericized market. New patent activity would likely focus on novel formulations or delivery systems rather than the active pharmaceutical ingredient itself.
-
What is the expected growth rate for over-the-counter (OTC) hydrocortisone products compared to prescription products? OTC hydrocortisone is expected to grow at a slightly higher CAGR than prescription hydrocortisone, driven by increased self-treatment of minor skin irritations and broader retail accessibility.
-
Which emerging markets are projected to show the most significant growth in hydrocortisone consumption? The Asia-Pacific region, particularly China and India, is projected to show the most significant growth due to expanding healthcare infrastructure, rising disposable incomes, and a large population base with a high burden of inflammatory diseases.
Citations
[1] Global Market Insights. (2023). Corticosteroids Market Size, Share & Trends Analysis Report. [2] National Eczema Association. (n.d.). Eczema Statistics. [3] National Psoriasis Foundation. (n.d.). Psoriasis Statistics. [4] World Health Organization. (2022). Global asthma report. [5] Crohn's & Colitis Foundation. (n.d.). About IBD. [6] World Health Organization. (2023). Global health expenditure: Fact sheet. [7] U.S. Food and Drug Administration. (2021). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). [8] Various pharmaceutical company annual reports and investor presentations (2022-2023).
More… ↓
