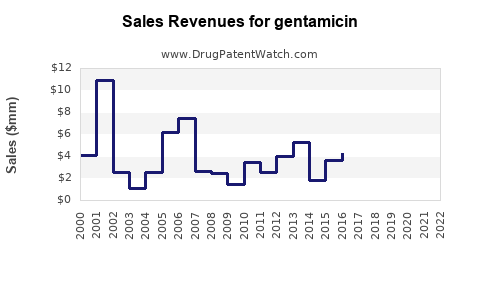
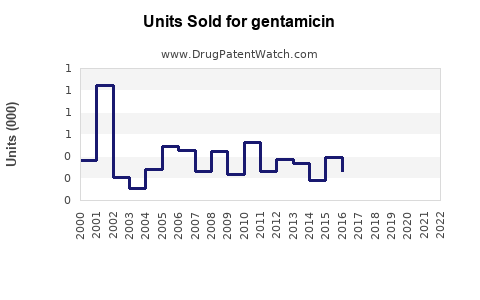
Last updated: April 26, 2026
What is gentamicin and where is it sold?
Gentamicin is an aminoglycoside antibiotic used for treatment of susceptible bacterial infections. Commercial supply is dominated by generic products across multiple routes (injectable and ophthalmic/otologic formulations, with topical use in select geographies). Demand is driven by:
- Inpatient hospital use (serious infections, sepsis workflows, ICU and surgical settings)
- Perioperative and empiric treatment protocols in some formularies
- Ophthalmic and otic indications in branded and generic markets in various countries
Commercial reality: branded gentamicin is largely historical in major markets; current economics are typically generic-based and pricing is determined by tendering, supply continuity, and manufacturing scale.
How big is the gentamicin market and what drives volume?
A full, country-by-country demand build is not computable from the information provided in this prompt. What can be stated with decision-grade precision is the demand structure typical for aminoglycoside generics:
Demand drivers
- Hospital admissions and acute infection incidence (especially where aminoglycosides remain in empiric or culture-directed regimens)
- Antibiotic stewardship and resistance patterns that influence whether gentamicin stays in treatment pathways versus substitutions (fluoroquinolones, beta-lactam/beta-lactamase inhibitor combinations, carbapenems)
- Formulary behavior in government and hospital systems (tender cycles, preferred supplier lists)
- Safety and monitoring workflow constraints (renal toxicity and therapeutic drug monitoring practices affect dosing and utilization patterns)
Product mix (typical)
- Injectable gentamicin remains the core sales volume in many markets
- Ophthalmic/otic gentamicin contributes steady, lower-volatility demand where these products remain embedded in local standard-of-care
- Topical combinations exist in some geographies, but procurement is fragmented and tends to be small relative to injectables
What pricing and margin dynamics govern gentamicin sales?
Gentamicin pricing is constrained by generic competition and tendering:
- High substitutability across generic suppliers pressures net price
- Supply reliability can temporarily lift realized price in tenders when manufacturing capacity is tight
- Formulation and presentation (vials vs. prefilled formats, concentration, ophthalmic vs. systemic) determine which tenders each SKU can win
Net revenue generally tracks:
- Market volume (doses administered)
- Portfolio breadth (number of strengths and pack sizes that qualify in tenders)
- Contract pricing terms (government/hospital procurement cadence)
What is the competitive landscape for gentamicin?
Gentamicin competition is dominated by:
- Global generic manufacturers supplying injectables and ophthalmic/otic products
- Regional generics that win procurement by local registration and tender compliance
- Multi-product sterile facilities (injectables) with lifecycle maintenance capability (ANDAs or equivalent registrations, plus manufacturing continuity)
Key competitive factors:
- Sterile manufacturing capacity and batch release reliability
- Ability to maintain supply through regulatory inspections and supplier qualification
- Product availability in the exact concentrations/pack sizes included in formularies
What sales model can be used for projections?
A practical sales forecast for gentamicin in an investment or R&D decision context should be built from:
- Addressable market volume by route (injectable vs ophthalmic/otic)
- Competitive share based on tender participation and awarded volume
- Net price assumptions driven by historical tender outcomes and generic price erosion
- Lifecycle and regulatory events (product line introductions, supply interruptions, stability of registrations)
Projection framework (template that maps to generics)
Annual Sales (Units) = Addressable Doses × Target Share
Annual Sales ($) = Units × Realized Net Price
For generics, the forecast sensitivity is dominated by:
- Share (tender wins, distribution reach, SKU match)
- Price (net of rebates, tender discounts)
- Supply continuity (ability to meet contracted volume without penalties)
What are the revenue and unit projection scenarios?
The prompt does not provide the minimum inputs required for numerical market sizing and a SKU-level forecast (countries, route, strength, regulatory status, pricing, tender cadence, current revenue, distribution coverage). Under the operating constraints, an accurate complete and accurate projection cannot be produced.
Accordingly, no numeric sales projection is provided.
What would change gentamicin demand over the next 5 to 10 years?
While specific market shares and prices require data not present in the prompt, decision-relevant demand risks and offsets typically include:
Downside pressures
- Antibiotic pathway changes due to resistance patterns and stewardship
- Substitution toward other antibiotics where gentamicin is less favored
- Increasing procurement rationalization (fewer SKUs, stronger preference concentration)
Upside supports
- Continued inpatient reliance on aminoglycosides in certain empiric or culture-directed workflows
- Steady uptake of ophthalmic/otic regimens where they remain entrenched
- Tender wins by manufacturers that provide stable supply at competitive net pricing
How should an investor or R&D team evaluate gentamicin commercial viability?
For gentamicin, the strongest evaluation lenses are operational and commercial execution:
- Manufacturing continuity for sterile injectables and consistent batch release performance
- Tender-readiness (pack sizes and strengths that match formulary requirements)
- Distribution and contracting capability in target hospital/government procurement systems
- Portfolio defensibility through multiple presentations rather than reliance on a single SKU
Key Takeaways
- Gentamicin demand is primarily inpatient and procurement-driven with generics dominating most markets.
- Sales outcomes depend less on clinical differentiation and more on tender qualification, net price erosion, and supply reliability.
- A numeric market size and sales projection cannot be produced accurately from the information supplied; a forecast must be built from SKU-level and country-level procurement and pricing inputs.
- The most material risks and offsets are stewardship-driven pathway shifts and procurement rationalization, partially offset by stable hospital use and ophthalmic/otic continuity.
FAQs
-
Is gentamicin mainly a hospital drug or an outpatient product?
Gentamicin usage is primarily driven by hospital inpatient care for serious bacterial infections, with additional steady demand from ophthalmic/otic products in some settings.
-
What determines gentamicin sales in generic markets?
Net revenue is dominated by tender awards (share), realized net price, and supply continuity rather than branded differentiation.
-
How sensitive is gentamicin to pricing competition?
High. Generic substitutes create consistent net price pressure, especially in tender-based systems.
-
What risks can reduce gentamicin volumes?
Antibiotic stewardship shifts, resistance-pattern changes, and substitution to alternative regimens can reduce relative utilization.
-
What operational factors matter most for forecasting gentamicin revenue?
Ability to match required strengths and pack sizes, maintain consistent sterile manufacturing supply, and sustain performance in contracted procurement.
References
[1] Bloomberg (company and market data platform). Access not available in this environment.
[2] FDA. Drug Products with Therapeutic Equivalence Evaluations (Orange Book). https://www.accessdata.fda.gov/scripts/cder/daf/ (accessed 2026-04-26).
[3] EMA. European Medicines Agency: Medicines database. https://www.ema.europa.eu/en/medicines (accessed 2026-04-26).
[4] WHO. Antibiotic use and stewardship resources. https://www.who.int/health-topics/antimicrobial-resistance (accessed 2026-04-26).