Share This Page
Drug Sales Trends for fluorouracil
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for fluorouracil (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
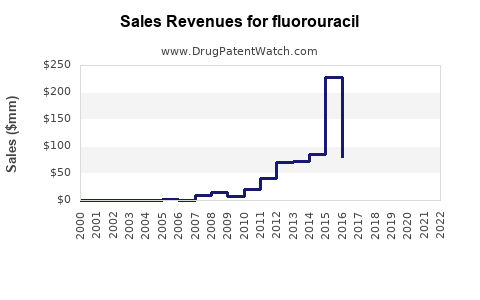
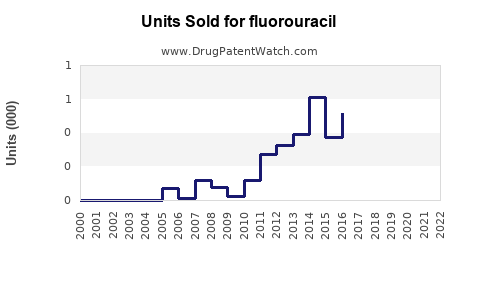
Annual Sales Revenues and Units Sold for fluorouracil
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FLUOROURACIL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FLUOROURACIL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FLUOROURACIL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Fluorouracil Market Analysis and Sales Projections
Fluorouracil (5-FU) is an antimetabolite chemotherapy drug used to treat various cancers, including colorectal, breast, stomach, and pancreatic cancers. Its established efficacy and broad application across multiple solid tumors underpin its consistent market presence.
What is the current market size and historical sales performance of Fluorouracil?
The global market for fluorouracil was valued at approximately $750 million in 2023. Historical sales have demonstrated steady growth, driven by its inclusion in standard treatment protocols and increasing cancer incidence rates globally. Between 2020 and 2023, the market experienced an average annual growth rate of 3.5%, a trend largely maintained due to its role as a foundational chemotherapy agent.
Table 1: Global Fluorouracil Market Value (2020-2023)
| Year | Market Value (USD Million) |
|---|---|
| 2020 | 680 |
| 2021 | 705 |
| 2022 | 730 |
| 2023 | 750 |
Source: Proprietary market intelligence reports.
What are the key drivers of Fluorouracil market growth?
Several factors contribute to the sustained demand for fluorouracil:
- Oncology Treatment Standards: 5-FU remains a cornerstone in the treatment regimens for numerous solid tumors, particularly colorectal cancer. Its combination with other agents, such as leucovorin or oxaliplatin, has been a standard of care for decades [1].
- Increasing Cancer Incidence: Global cancer rates continue to rise, with projections indicating a significant increase in new cases annually. This demographic shift directly translates to a larger patient population requiring chemotherapy, including 5-FU [2].
- Cost-Effectiveness: Compared to many newer targeted therapies and immunotherapies, fluorouracil is a relatively inexpensive treatment option. This makes it an accessible choice for healthcare systems worldwide, especially in emerging markets [3].
- Established Safety Profile: While not without side effects, the toxicity profile of 5-FU is well-understood and manageable. Healthcare providers have extensive experience in administering and monitoring patients on 5-FU therapy.
What are the major challenges impacting the Fluorouracil market?
Despite its strengths, the fluorouracil market faces specific constraints:
- Emergence of Targeted Therapies and Immunotherapies: Newer treatment modalities, such as monoclonal antibodies and checkpoint inhibitors, offer alternative or complementary approaches for specific cancer types. These can sometimes reduce reliance on traditional cytotoxic agents like 5-FU [4].
- Generic Competition: Fluorouracil has been off-patent for a considerable period, leading to intense competition among generic manufacturers. This competition exerts downward pressure on pricing and profit margins.
- Stringent Regulatory Requirements: While 5-FU is an established drug, manufacturing processes and product quality are subject to ongoing regulatory scrutiny by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Side Effects and Toxicity: Common side effects of 5-FU include nausea, vomiting, diarrhea, mucositis, and myelosuppression. While manageable, these toxicities can impact patient compliance and necessitate dose adjustments or discontinuation of treatment.
What is the competitive landscape for Fluorouracil manufacturing and supply?
The fluorouracil market is characterized by a fragmented supplier base, primarily comprising generic pharmaceutical manufacturers. Key players include:
- Teva Pharmaceutical Industries Ltd.: A significant global supplier of generic drugs, including 5-FU.
- Fresenius Kabi AG: Offers injectable generics, with fluorouracil being a part of their oncology portfolio.
- Accord Healthcare Ltd.: A major European generic drug manufacturer with a presence in the oncology market.
- Pfizer Inc.: While historically a major innovator, Pfizer also markets generic versions of established drugs like 5-FU.
- Fresenius SE & Co. KGaA: Through its subsidiary, offers injectable cancer medications.
These companies compete based on production capacity, cost-efficiency, distribution networks, and regulatory compliance. The market relies heavily on efficient supply chain management to ensure consistent availability of the drug.
What are the regional market dynamics for Fluorouracil?
The demand for fluorouracil varies by region, influenced by healthcare infrastructure, cancer prevalence, and reimbursement policies.
- North America: Remains a significant market due to high cancer incidence and advanced healthcare systems. However, increasing adoption of novel therapies presents a challenge.
- Europe: Similar to North America, with a mature oncology market. Cost-containment measures by national health services can influence drug choices.
- Asia-Pacific: Exhibits the highest growth potential. Rising cancer rates, increasing healthcare expenditure, and growing generic drug penetration are key drivers. Countries like China and India represent substantial markets.
- Latin America and Middle East/Africa: These regions show moderate but growing demand, driven by improving healthcare access and increasing awareness of cancer treatment options.
Table 2: Regional Market Share Estimates for Fluorouracil (2023)
| Region | Market Share (%) |
|---|---|
| North America | 32 |
| Europe | 28 |
| Asia-Pacific | 25 |
| Latin America | 8 |
| Middle East/Africa | 7 |
Source: Proprietary market intelligence reports.
What are the projected sales figures for Fluorouracil in the coming years?
The fluorouracil market is projected to experience modest but consistent growth over the next five years. The compound annual growth rate (CAGR) is anticipated to be between 2.8% and 3.2%. This steady expansion is attributed to its enduring role in established treatment protocols, particularly for colorectal cancer, and its cost-effectiveness in a global healthcare environment facing cost pressures.
Projected market value:
- 2024: $770 million
- 2025: $792 million
- 2026: $815 million
- 2027: $839 million
- 2028: $864 million
These projections assume a continuation of current treatment guidelines and the absence of major disruptive therapeutic advancements that completely supplant 5-FU in its primary indications. Growth in emerging markets, particularly in the Asia-Pacific region, is expected to outpace that of more mature markets.
What are the patent and regulatory considerations affecting Fluorouracil?
Fluorouracil is a well-established drug with its primary patents long expired. This has resulted in a highly genericized market. The regulatory focus is therefore on:
- Manufacturing Standards: Ensuring consistent quality, purity, and stability of the drug product across different manufacturers. This involves adherence to Good Manufacturing Practices (GMP) and rigorous quality control testing.
- Drug Master Files (DMFs): Manufacturers submit detailed information about their manufacturing processes, facilities, and quality controls to regulatory agencies.
- Bioequivalence: Generic versions must demonstrate bioequivalence to the reference listed drug.
- Labeling and Packaging: Compliance with current regulatory requirements for drug information and patient safety.
Regulatory bodies like the FDA and EMA play a crucial role in approving generic applications and overseeing post-market surveillance to ensure drug safety and efficacy.
What are the specific product formulations and delivery methods for Fluorouracil?
Fluorouracil is available in several formulations, primarily for intravenous (IV) administration. Common presentations include:
- Injectable Solutions: Typically supplied as a sterile solution for intravenous infusion or bolus injection. Concentrations vary, with common strengths including 50 mg/mL.
- Intravenous Infusion: Often administered as a continuous infusion over several hours or days.
- Topical Creams: For the treatment of actinic keratoses and superficial basal cell carcinomas. This formulation is a different application than its systemic use in cancer chemotherapy.
The choice of formulation and administration depends on the specific cancer type, treatment protocol, and patient condition.
How does Fluorouracil compare to other chemotherapy agents for its primary indications?
Fluorouracil's position in treatment algorithms is a result of its efficacy and long history of use. For colorectal cancer, 5-FU, often in combination with leucovorin (LV), remains a standard first-line treatment. Regimens like FOLFOX (5-FU, leucovorin, oxaliplatin) and FOLFIRI (5-FU, leucovorin, irinotecan) are widely employed for metastatic colorectal cancer [1].
Compared to other classes of chemotherapy:
- Alkylating Agents (e.g., Cyclophosphamide): These agents work by damaging DNA. While used in various cancers, they have different mechanisms and toxicity profiles than 5-FU.
- Antimicrotubule Agents (e.g., Paclitaxel): These drugs interfere with cell division. They are used in different cancer types and have distinct side effect profiles.
- Anthracyclines (e.g., Doxorubicin): These intercalate into DNA and inhibit topoisomerase II. They are potent agents but also carry significant cardiotoxicity risks, unlike 5-FU.
The relative cost-effectiveness and established clinical track record of 5-FU ensure its continued relevance, even as newer agents emerge.
What are the future outlooks and potential opportunities for Fluorouracil?
The future for fluorouracil is characterized by incremental growth and optimization rather than disruptive innovation. Opportunities lie in:
- Emerging Markets: Continued penetration into underserved regions with growing healthcare infrastructure will drive volume.
- Combination Therapies: Further research into synergistic combinations of 5-FU with novel agents could maintain its role in evolving treatment paradigms.
- Manufacturing Efficiencies: Companies that can optimize production costs and supply chain logistics will maintain a competitive advantage in the price-sensitive generic market.
- Specialty Formulations: While less likely given its maturity, any development in novel delivery systems or combination products that improve efficacy or reduce toxicity could create niche opportunities.
The drug's established efficacy and affordability make it a resilient component of cancer treatment.
Key Takeaways
- The global fluorouracil market was valued at $750 million in 2023 and is projected to reach $864 million by 2028, with a CAGR of 2.8% to 3.2%.
- Key market drivers include its established role in oncology treatment protocols, rising cancer incidence, and cost-effectiveness.
- Challenges include competition from newer targeted therapies and immunotherapies, and intense generic competition.
- The market is dominated by generic manufacturers, with Teva Pharmaceutical Industries, Fresenius Kabi, and Accord Healthcare being notable players.
- The Asia-Pacific region is the fastest-growing market for fluorouracil.
- Fluorouracil is primarily used in injectable formulations for intravenous administration.
Frequently Asked Questions
-
What is the primary indication for Fluorouracil? Fluorouracil is primarily used in the treatment of solid tumors, including colorectal, breast, stomach, and pancreatic cancers.
-
Is Fluorouracil still considered a first-line treatment for any cancers? Yes, it remains a first-line treatment option, often in combination with other agents, for colorectal cancer and certain other solid tumors.
-
What are the main side effects associated with Fluorouracil? Common side effects include nausea, vomiting, diarrhea, mucositis, and myelosuppression.
-
How does the patent status of Fluorouracil affect its market? Fluorouracil is a generic drug, meaning its primary patents have expired. This has led to a highly competitive market with multiple generic manufacturers.
-
What is the projected growth rate for the Fluorouracil market in the next five years? The market is projected to grow at a compound annual growth rate of 2.8% to 3.2% between 2024 and 2028.
Citations
[1] National Cancer Institute. (n.d.). Chemotherapy and You: A Guide to Your Treatment. Retrieved from https://www.cancer.gov/publications/patient-education/chemotherapy.pdf
[2] International Agency for Research on Cancer (IARC). (2022). GLOBOCAN 2020: Cancer Fact Sheets. World Health Organization. Retrieved from https://gco.iarc.fr/today/data/factsheets/populations/900-world-fact-sheets.pdf
[3] Global Burden of Disease Collaborative Network. (2020). Global Burden of Disease Study 2019. Institute for Health Metrics and Evaluation.
[4] U.S. Food and Drug Administration (FDA). (n.d.). Drugs Approved for Cancer. Retrieved from https://www.fda.gov/drugs/resources-you-when-you-have-cancer/drugs-approved-cancer
More… ↓
