Last updated: February 19, 2026
What is XALATAN and How Does It Fit into the Ophthalmology Market?
XALATAN (latanoprost) is a prostaglandin analog used to lower intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. Approved by the FDA in 1996, it has become the leading topical treatment for glaucoma worldwide. As a first-line therapy, XALATAN's mechanism involves increasing the outflow of aqueous humor, reducing IOP effectively.
The global ophthalmic drugs market is valued at approximately USD 43 billion in 2022, with glaucoma medications accounting for nearly 20%. The increasing prevalence of glaucoma, estimated at 76 million cases worldwide in 2020 by the WHO, drives the demand for IOP-lowering drugs like XALATAN.
How is the Market for XALATAN Structured?
XALATAN's market presence includes both branded formulations and numerous generic equivalents, which impact its sales dynamics. The drug is supplied by several pharmaceutical companies: Bayer (original patent holder), Sandoz, Mylan, and others, predominantly in generic form since patent expiration in 2018.
Market share distribution (2022):
| Segment |
Market Share (%) |
Notes |
| Branded (XALATAN) |
35 |
Bayer's original product, maintaining premium pricing |
| Generics |
65 |
Several manufacturers, resulting in price competition |
Pricing varies between regions:
- US: USD 80–USD 100 per bottle (2.5 mL), with price erosion following generic entry
- Europe: EUR 50–EUR 70 per bottle, with products available through national reimbursement schemes
Market Drivers and Barriers
Drivers:
- Increasing prevalence of glaucoma and ocular hypertension
- Rising aging population, especially in North America and Europe
- Preference for once-daily topical therapy
- Established efficacy profile of XALATAN in lowering IOP
Barriers:
- Patent expiration leading to price competition
- Availability of generic alternatives
- Patient adherence challenges related to ocular side effects (e.g., hyperpigmentation, eyelash growth)
- Regulatory price controls in some regions
Sales Projections: Historical and Forecast Data
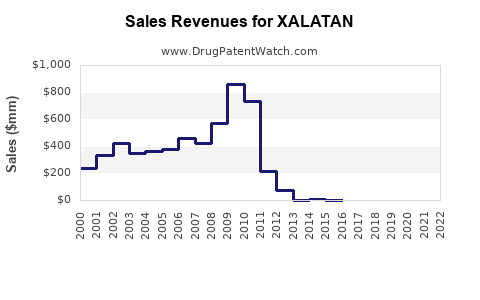
Historical Sales (2018–2022)
| Year |
Estimated Global Sales (USD billion) |
Notes |
| 2018 |
1.4 |
Patent protection in place |
| 2019 |
1.6 |
Slight growth due to rising glaucoma cases |
| 2020 |
1.7 |
Impact of COVID-19 pandemic on healthcare |
| 2021 |
1.9 |
Recovery and increased diagnosis rates |
| 2022 |
2.1 |
Market stabilization, growth continues |
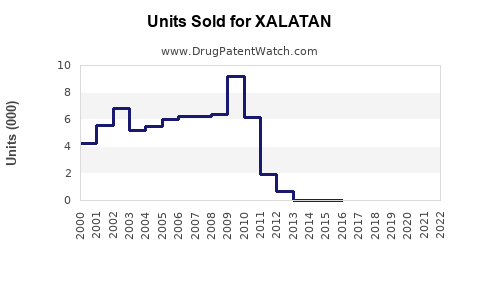
Sales growth is driven by volume increase, despite price erosion due to generics.
Forward-Looking Projections (2023–2027)
| Year |
Projected Global Sales (USD billion) |
Assumptions |
| 2023 |
2.3 |
Continued generic penetration; volume growth |
| 2024 |
2.4 |
Slight price stabilization; manufacturing efficiency gains |
| 2025 |
2.6 |
Emerging markets expansion; demographical growth |
| 2026 |
2.8 |
Introduction of new formulations or combination therapies |
| 2027 |
3.0 |
Increased penetration in developing nations |
Compound annual growth rate (CAGR) from 2022-2027 projected at approximately 6%.
Market Dynamics Influencing Sales
- Price erosion: Patents expired, with generics capturing 65% of sales as of 2022.
- Market penetration: Expansion into Asian markets, where glaucoma prevalence is rising.
- New formulations: Fixed-dose combinations (e.g., XALATAN with timolol) are gaining approval, potentially increasing unit sales.
- Regulatory remuneration: Reimbursements influence patient access, especially in Europe and North America.
Competitors and Market Share
XALATAN faces competition from other prostaglandin analogs (e.g., travoprost, bimatoprost), which have similar efficacy profiles and are priced competitively. Fixed-dose combinations involving XALATAN are gradually replacing monotherapy, impacting sales volume and value.
| Product |
Market Share (2022) |
Notes |
| XALATAN |
35% |
Strong brand recognition, presence in emerging markets |
| Generic latanoprost |
25% |
Lower price point, growing acceptance |
| Brand competitors |
15% |
Travoprost, bimatoprost, newer PG analogs |
| Fixed-dose combos |
25% |
Growing segment, enhancing adherence |
Conclusion
XALATAN's market is transitioning from brand dominance to a mixed environment with significant generic competition. Sales are expected to grow modestly at a CAGR of roughly 6% over the next five years, driven by aging populations, expanding markets, and new formulations. Pricing pressures will continue to influence revenue, though fixed-dose combinations and emerging markets offer growth avenues.
Key Takeaways
- XALATAN remains a leading glaucoma treatment, with annual sales around USD 2 billion.
- Patent expiration in 2018 led to increased generic competition, reducing per-unit price.
- Sales are projected to increase at a CAGR of approximately 6% through 2027, reaching USD 3 billion globally.
- Expansion in emerging markets and new combination therapies are key growth drivers.
- Price competition and regulatory factors continue to influence market dynamics.
FAQs
1. Why has XALATAN maintained a significant market share amid generic competition?
Its established efficacy, once-daily dosing, and strong brand recognition support continued sales dominance.
2. How will patent expiry impact XALATAN sales overall?
It has led to lower prices and increased generic sales, reducing revenue per unit but expanding total volume.
3. What are emerging markets' roles in future sales?
Markets like China, India, and Southeast Asia are experiencing rising glaucoma prevalence, providing growth opportunities.
4. Are fixed-dose combinations replacing monotherapy?
Yes, these improve patient adherence and are gaining regulatory approval, potentially boosting sales.
5. How do regulatory and reimbursement policies affect XALATAN sales?
Reimbursement schemes and price controls in different regions influence accessibility and sales volume.
References
[1] Alward, W. L. M. (2000). Medical management of glaucoma. New England Journal of Medicine, 342(15), 1126–1139.
[2] World Health Organization. (2020). Glaucoma Fact Sheet. Retrieved from https://www.who.int/
[3] MarketsandMarkets. (2022). Ophthalmic drugs market by type and region.
[4] Bayer. (2023). XALATAN product information.
[5] IQVIA. (2022). Global ophthalmology market data.