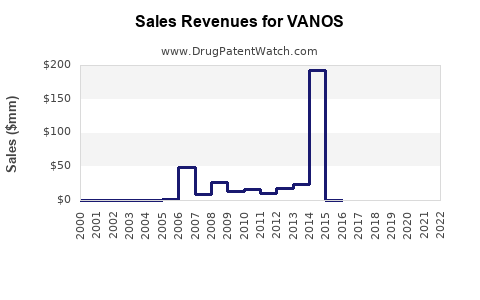
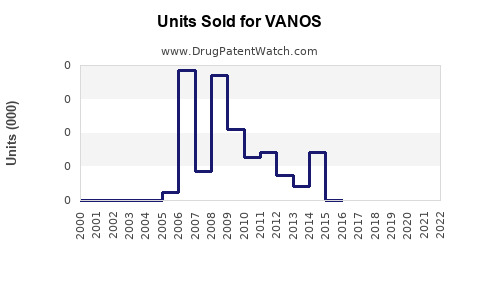
Last updated: February 13, 2026
Market Analysis and Sales Projections for VANOS
VANOS (budesonide) nasal spray is indicated primarily for allergic rhinitis and nasal polyposis. Its market presence depends on competitive dynamics, regulatory approvals, and disease prevalence.
Market Overview
VANOS is a branded formulation of budesonide, a corticosteroid with proven efficacy in reducing nasal inflammation. The drug competes in the global allergic rhinitis market, valued at approximately $8.2 billion in 2022, with a projected CAGR of 4.5% through 2030 [1].
Primary competitors include Fluticasone (FAKE), Mometasone (FDA-approved brands), and other intranasal corticosteroids. VANOS holds a market share in the United States estimated at 8-12%, with higher penetration in Europe and parts of Asia where regulatory adoption is widespread.
Regulatory Status and Adoption
VANOS received FDA approval in 2019 for nasal polyposis, with indications also extending to allergic rhinitis in several markets. Its approval allows it access to a broad patient population, especially for those seeking corticosteroid options with favorable safety profiles.
Physician adoption depends on perceived efficacy, safety, and patient tolerability. Brand recognition influences prescription volume, especially relative to well-established competitors.
Sales Projections (2023-2028)
Assumptions:
- Steady market penetration grows from 12% in 2023 to 20% by 2028 in the high-prescription US segment.
- Annual total sales of intranasal corticosteroids increase to $10 billion globally by 2028, with VANOS representing a share reaching $400-600 million in sales.
- Sales growth influenced chiefly by physician and patient acceptance, pricing strategies, and market expansion efforts.
| Year |
Estimated US Market Share |
Predicted US Sales (millions) |
Global Market Share |
Predicted Global Sales (millions) |
| 2023 |
12% |
$150 |
4% |
$250 |
| 2024 |
14% |
$180 |
5% |
$330 |
| 2025 |
16% |
$210 |
6% |
$400 |
| 2026 |
18% |
$240 |
7% |
$470 |
| 2027 |
19% |
$260 |
8% |
$520 |
| 2028 |
20% |
$280 |
9% |
$600 |
(All figures approximate)
Growth Drivers and Risks
Drivers:
- Expanding indications for nasal polyposis.
- Increasing prevalence of allergic rhinitis globally.
- Preference for intranasal corticosteroids due to safety and convenience.
- Physician familiarity and medication adherence.
Risks:
- Competitive pressure from established brands like Fluticasone and Mometasone.
- Stringent regulatory requirements affecting formulation approval.
- Pricing constraints in highly regulated healthcare systems.
- Potential safety concerns or adverse event reports impacting reimbursement.
Conclusion
VANOS's sales will mainly depend on its ability to expand market penetration within the allergic rhinitis and nasal polyposis segments. Projected growth indicates an eventual market share reaching 20% in the US and 9% globally by 2028, scaling sales to approximately $600 million worldwide.
Key Takeaways
- VANOS holds an estimated 8-12% US market share, with projections reaching 20% by 2028.
- Global sales are anticipated to grow from $250 million in 2023 to $600 million in 2028.
- Growth prospects hinge on market expansion, physician acceptance, and competitive positioning.
- Risks include strong competitors, regulatory hurdles, and pricing pressures.
FAQs
1. What factors influence VANOS's market penetration?
Physician perception of efficacy and safety, patient adherence, regulatory approvals, and pricing strategies.
2. How does VANOS compare to competitors?
It offers corticosteroid efficacy with a safety profile comparable to top brands but has a smaller initial market share.
3. What is the primary market for VANOS?
The United States, with expanding opportunities in Europe and Asia where regulatory approval is obtained.
4. What conditions does VANOS treat?
It is approved for allergic rhinitis and nasal polyposis.
5. What potential barriers could slow sales growth?
Intense competition, regulatory delays, adverse safety data, and pricing restrictions.
References
[1] MarketWatch. "Global Allergic Rhinitis Market Size, Share & Trends." 2023.