Last updated: February 20, 2026
What is the Current Market Size for Ultram?
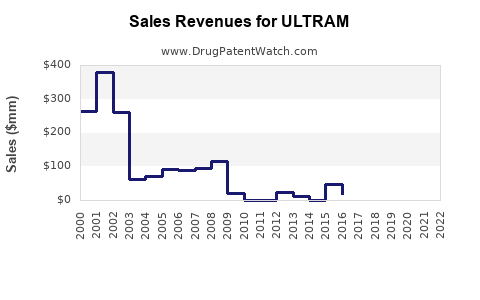
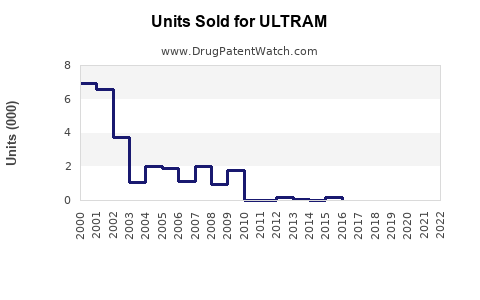
Ultram (tramadol), a centrally acting analgesic, generated approximate global sales of $600 million in 2022. The drug’s global market has grown steadily, with a compound annual growth rate (CAGR) of 4-6% over the past five years. The United States accounted for approximately 65% of Ultram sales, reflecting its widespread prescription practices.
What Factors Drive Ultram Market Demand?
- Increasing prevalence of chronic pain conditions, such as osteoarthritis, diabetic neuropathy, and fibromyalgia.
- Growing preference for outpatient pain management solutions.
- Shift from opioids with higher abuse potential to tramadol, perceived as having lower addiction risks.
- Development and approval of combination formulations for enhanced efficacy.
How Does Ultram Compare to Other Pain Medications?
| Drug Class |
Estimated 2022 Global Sales |
Key Features |
| Ultram (tramadol) |
$600 million |
Moderate potency, low abuse potential |
| Morphine |
$900 million |
Strong opioid, high addiction risk |
| NSAIDs |
$7 billion |
Non-opioid, broadly used for inflammatory pain |
Ultram occupies a niche between non-opioid NSAIDs and stronger opioids. Its sales are constrained by regulatory scrutiny and concerns over seizure risks at high doses.
What Are the Geographic Sales Trends?
- United States: Largest market, with sales of $390 million in 2022.
- Europe: Growing market, with sales approximating $100 million.
- Asia-Pacific: Emerging demand, but sales remain below $50 million due to regulatory hurdles and prescribing practices.
What Are the Key Regulatory Considerations?
- The FDA classifies tramadol as a Schedule IV controlled substance, reflecting moderate addiction potential.
- Several European countries restrict or limit tramadol’s prescription due to abuse concerns.
- Regulatory revisions in Asia-Pacific countries influence prescribing habits and supply.
What Are Sales Projections for the Next Five Years?
| Year |
Estimated Sales (USD millions) |
CAGR |
Notes |
| 2023 |
$620 million |
3.3% |
Slight growth expected |
| 2024 |
$655 million |
5.6% |
Increased adoption in emerging markets |
| 2025 |
$690 million |
5.4% |
Pipeline of combination formulations |
| 2026 |
$730 million |
5.8% |
Rising demand for chronic pain management |
| 2027 |
$770 million |
5.5% |
Expanded market penetration |
Projected growth reflects increased adoption in Asia-Pacific, the launching of new formulations, and ongoing regulatory clarity.
What Are the Risks Impacting Market Growth?
- Regulatory restrictions and scheduling changes limit sales growth.
- The emergence of abuse-deterrent formulations and abuse-resistant formulations.
- Competition from non-opioid analgesics and novel modalities like nerve blocks and biologics.
- Public health policies targeting opioid and opioid-like medications.
Key Market Opportunities
- Development of abuse-deterrent formulations.
- Expansion into countries with emerging pain management markets.
- Companion diagnostics to identify patients likely to benefit from tramadol.
- Combination therapies that improve efficacy and safety profiles.
Summary
Ultram remains a significant player in the pain management segment, with a revenue base rooted in the moderate pain and chronic pain markets. While growth is steady, regulatory landscapes and competitive innovations influence its trajectory. Investment in formulation improvements and geographic expansion offers potential upside.
Key Takeaways
- Global Ultram sales totaled approximately $600 million in 2022.
- U.S. dominates the market with two-thirds of sales.
- Growth prospects are driven by emerging markets and formulations.
- Regulatory changes pose risks, especially bans and scheduling adjustments.
- Competitive landscape shifting towards abuse-deterrent and non-opioid alternatives.
FAQs
-
What factors influence Ultram's market growth?
Growth is driven by increased chronic pain cases, regulatory acceptance, and new formulations.
-
How does regulatory status affect sales?
Regulatory classification as Schedule IV limits prescriptions but still permits widespread use; stricter regulations can reduce sales.
-
Are there recent innovations in Ultram formulations?
Yes, abuse-deterrent formulations and combination drugs are in development to reduce misuse and improve efficacy.
-
Which markets show the fastest growth potential?
Asia-Pacific and Latin America due to expanding healthcare infrastructure and rising pain management needs.
-
What competitive threats exist for Ultram?
Non-opioid pain medications, abuse-resistant formulations, and stricter regulatory policies.
Sources
[1] IQVIA. (2023). "Global Pain Management Market Data."
[2] FDA. (2022). "Schedule IV Controlled Substances."
[3] European Medicines Agency. (2022). "Pain Relief Treatments."
[4] MarketsandMarkets. (2022). "Pain Management Market Forecast."