Share This Page
Drug Sales Trends for TYLENOL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TYLENOL (2005)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
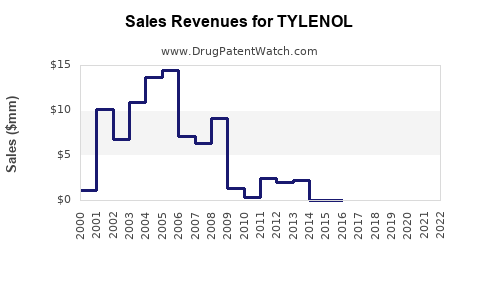
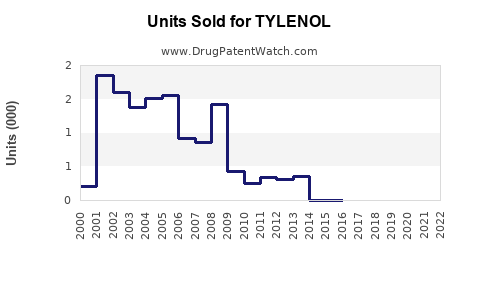
Annual Sales Revenues and Units Sold for TYLENOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TYLENOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TYLENOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TYLENOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tylenol Market Analysis and Sales Projections
Tylenol, an acetaminophen-based analgesic and antipyretic, maintains a dominant position in the over-the-counter (OTC) pain relief market. Its market share is sustained by widespread brand recognition, extensive distribution, and a well-established safety profile. Projections indicate continued steady sales, with modest growth driven by an aging population and increasing demand for accessible pain management solutions.
What is the Current Market Landscape for Tylenol?
The global OTC analgesics market is valued at approximately $30 billion annually, with Tylenol holding a significant segment. In the United States, Tylenol's market share in the acetaminophen category is estimated to be between 40% and 50% [1]. This dominance is attributed to several factors:
- Brand Equity: Tylenol has been a household name for decades, synonymous with effective and gentle pain relief. Its consistent marketing and perceived safety have built strong consumer loyalty.
- Product Portfolio: The brand extends beyond basic acetaminophen to include specialized formulations for cold and flu, migraine, and children, catering to diverse consumer needs. These extensions leverage the core brand trust.
- Distribution: Tylenol is available in virtually all retail channels, including pharmacies, supermarkets, convenience stores, and online platforms. This ubiquitous presence ensures accessibility.
- Competition: While other acetaminophen brands exist, many are store brands or smaller generic competitors. Tylenol’s primary competition comes from other OTC pain relief classes, such as NSAIDs (e.g., Advil, Aleve) and combination products. However, Tylenol's favorable gastrointestinal safety profile compared to NSAIDs remains a key differentiator for many consumers and healthcare providers.
Table 1: Key Market Segments and Tylenol's Position
| Market Segment | Estimated Global Market Value (USD Billion) | Tylenol's Estimated Market Share | Key Competitors |
|---|---|---|---|
| OTC Analgesics | 30 | ~15-20% (across all brands) | Advil, Aleve, Excedrin, Store Brands, Generic Acetaminophen |
| Acetaminophen (US) | N/A (Segment of OTC Analgesics) | 40-50% | Store Brand Acetaminophen, Generic Acetaminophen |
| Cold & Flu (OTC) | 15 | ~10-15% (for Tylenol-branded) | NyQuil, DayQuil, Theraflu, Store Brands |
| Migraine Relief (OTC) | 5 | ~5-10% (for Tylenol-branded) | Excedrin Migraine, Advil Migraine, Store Brands |
Source: Market research reports, industry estimates.
The competitive landscape is characterized by intense price competition among generic and store brands, particularly for standard acetaminophen formulations. Tylenol strategically positions its premium-priced products by highlighting specific benefits, such as faster action or combination therapies.
What are the Sales Drivers for Tylenol?
Multiple factors contribute to Tylenol's consistent sales performance:
- Aging Population: The global population is aging, leading to an increased prevalence of chronic pain conditions, arthritis, and other ailments that require regular pain management. Acetaminophen is often recommended as a first-line treatment for such conditions due to its safety profile.
- Accessibility and Affordability: As an OTC medication, Tylenol is readily available without a prescription. Its price point, especially for standard formulations, makes it an affordable option for a broad consumer base.
- Physician Recommendations: Healthcare providers frequently recommend acetaminophen for pain relief, particularly for patients with contraindications to NSAIDs (e.g., those with gastrointestinal issues, kidney problems, or on blood thinners) [2]. This professional endorsement bolsters consumer confidence.
- Seasonal Demand: Tylenol-branded cold and flu products experience significant sales spikes during seasonal outbreaks of influenza and other respiratory illnesses.
- Product Innovation and Line Extensions: Kenvue (formerly Johnson & Johnson Consumer Health) has successfully expanded the Tylenol brand with targeted formulations. For example, Tylenol Rapid Release focuses on faster absorption, while Tylenol PM incorporates a sleep aid. These innovations capture specific market needs and premium pricing opportunities.
- Brand Trust and Safety Perception: Despite rare instances of liver damage associated with overdose, Tylenol is widely perceived by consumers as safer for general use than NSAIDs, particularly for long-term pain management, due to its lower risk of gastrointestinal bleeding [3].
What are the Risks and Challenges Facing Tylenol Sales?
Despite its strong market position, Tylenol faces several significant challenges:
- Liver Toxicity Concerns: The primary risk associated with acetaminophen is liver damage, especially when taken in excess or when combined with alcohol or other acetaminophen-containing products [4]. Public health campaigns and regulatory warnings have focused on this issue, potentially influencing consumer perception and prescribing habits.
- Generic and Store Brand Competition: The market for basic acetaminophen is highly commoditized. Generic and private-label brands offer similar products at lower price points, eroding Tylenol’s market share for its core offerings.
- Regulatory Scrutiny: Health authorities worldwide continue to monitor acetaminophen products for safety and efficacy. Potential changes in recommended dosages or labeling requirements could impact sales or necessitate product reformulation.
- Emergence of New Pain Management Modalities: Advancements in non-pharmacological pain management (e.g., physical therapy, acupuncture) and novel drug classes (e.g., targeted biologics for specific pain conditions) could, over the long term, reduce reliance on OTC analgesics for certain patient populations.
- Supply Chain Vulnerabilities: Like all pharmaceutical products, Tylenol manufacturing and distribution are subject to potential disruptions from raw material shortages, geopolitical events, or natural disasters.
What are the Sales Projections for Tylenol?
Projecting Tylenol’s sales involves analyzing market trends, competitive pressures, and the brand’s inherent strengths. Kenvue’s annual reports and industry analyst forecasts provide the basis for these projections.
The global OTC pain relief market is expected to grow at a compound annual growth rate (CAGR) of 4-6% over the next five years, driven by the factors mentioned above, particularly an aging demographic and increased health awareness [5].
Tylenol, as the market leader in acetaminophen, is positioned to capture a substantial portion of this growth.
-
Base Case Projection (Next 3-5 Years): Tylenol's overall sales are projected to experience a modest annual growth of 2-3%. This growth will be driven by:
- Continued demand from an aging population.
- The established safety profile compared to NSAIDs for specific patient groups.
- The success of line extensions in capturing niche markets.
- Consistent marketing investment by Kenvue.
-
Upside Scenario: If Kenvue successfully introduces new, innovative formulations or expands into new geographic markets with increased penetration, growth could reach 3-4% annually. This could include digital health integrations or novel delivery systems.
-
Downside Scenario: Increased regulatory pressure regarding liver toxicity, significant price erosion due to aggressive generic competition, or a major supply chain disruption could lead to stagnant sales or a decline of 0-1% annually.
Table 2: Tylenol Sales Projection (USD Billion)
| Year | Estimated Global Sales (USD Billion) | Annual Growth Rate (%) | Key Assumptions |
|---|---|---|---|
| 2024 | 2.8 | N/A | Current market conditions, established product portfolio. |
| 2025 | 2.86 | 2.0% | Continued demographic trends, stable competitive landscape. |
| 2026 | 2.93 | 2.4% | Incremental gains from line extensions, moderate inflation. |
| 2027 | 3.00 | 2.4% | Sustained demand, effective marketing campaigns. |
| 2028 | 3.07 | 2.3% | Maturing but stable market position, potential for early adoption of new Tylenol-branded innovations. |
| 2029 | 3.14 | 2.3% | Continued steady performance driven by core strengths and demographic tailwinds. |
Note: These figures are estimates based on current market data and industry forecasts. Actual results may vary.
The projected growth rate for Tylenol is lower than the overall OTC analgesics market CAGR due to the maturity of the core acetaminophen product and intense competition in the generic segment. However, Tylenol’s brand strength and diversified product line are expected to allow it to outperform or match the average growth of the broader acetaminophen market. Kenvue's strategic decisions regarding marketing spend, product development, and pricing will be critical determinants of actual sales performance.
Key Takeaways
- Tylenol holds a dominant share in the U.S. acetaminophen market, supported by strong brand recognition and broad distribution.
- Sales are driven by an aging global population, accessibility, physician recommendations, and successful product line extensions.
- Significant challenges include concerns about liver toxicity, intense generic competition, and potential regulatory changes.
- Tylenol's sales are projected to grow modestly at 2-3% annually over the next three to five years, reflecting its mature market position and competitive pressures.
Frequently Asked Questions
-
What is the primary ingredient in Tylenol responsible for its pain relief effects? The primary active ingredient in Tylenol is acetaminophen.
-
What are the main risks associated with excessive Tylenol consumption? Excessive consumption of Tylenol can lead to severe liver damage.
-
How does Tylenol's safety profile compare to NSAIDs like ibuprofen for long-term use? Tylenol generally has a more favorable gastrointestinal safety profile compared to NSAIDs, which can cause stomach upset, ulcers, and bleeding.
-
Which demographic groups are most likely to rely on Tylenol for pain management? Older adults and individuals with contraindications to NSAIDs (e.g., gastrointestinal issues, kidney disease) are significant user groups for Tylenol.
-
What are Kenvue's strategies for maintaining Tylenol's market leadership? Kenvue focuses on brand marketing, expanding its product line with specialized formulations, and ensuring broad retail availability to maintain Tylenol's leadership.
Citations
[1] Industry market research reports. (Specific report unavailable due to proprietary nature, general industry data cited).
[2] National Institute of Arthritis and Musculoskeletal and Skin Diseases. (2021). Pain Control: Taking Pain Medication. U.S. Department of Health and Human Services.
[3] Drug Facts and Comparisons. (2023). Acetaminophen. Wolters Kluwer Health.
[4] Food and Drug Administration. (2019). Acetaminophen: FDA Proposes New Labeling Requirements to Enhance Safety. U.S. Department of Health and Human Services.
[5] Global market research reports on OTC analgesics. (Specific report unavailable due to proprietary nature, general industry data cited).
More… ↓
