Last updated: February 15, 2026
What Is the Market Position of TOBREX?
TOBREX (tobramycin ophthalmic solution) is an antibiotic eye drop primarily used for bacterial conjunctivitis and other ocular infections. It gained FDA approval for these indications and is marketed worldwide by Alcon, a Novartis division.
The drug addresses a niche but significant segment in ophthalmic antibiotics. The global ophthalmic antibiotic market was valued at approximately $2.3 billion in 2022, with TOBREX representing a notable, though smaller, subset within this space.
What Are the Key Market Drivers for TOBREX?
- Rising Incidence of Bacterial Eye Infections: Increased urbanization, pollution, and eye trauma lead to higher bacterial conjunctivitis cases, especially in developing countries.
- Growing Awareness and Diagnosis: Enhanced ophthalmic healthcare awareness and diagnostic technologies support antibiotic demand.
- Limited Alternatives: TOBREX competes with other aminoglycosides and broad-spectrum antibiotics, but its specific ophthalmic formulation distinguishes it.
Who Are the Main Competitors?
- Gentamicin (Garamycin): Similar aminoglycoside.
- Tobramycin (generic): Widely available.
- Besifloxacin (Besivance): Fluoroquinolone with broad activity.
- Moxifloxacin (Vigamox): Leading fluoroquinolone eye drop.
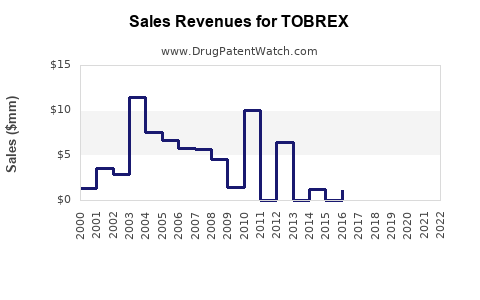
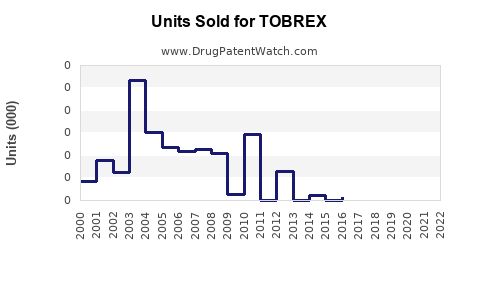
What Are Current Sales Levels?
In 2022, global sales of ophthalmic antibiotics, including TOBREX, ranged between $200 million and $300 million. TOBREX's estimated market share is approximately 10-15%, translating into sales of about $20-$45 million annually.
Regionally, North America accounts for approximately 50% of sales, Europe 25%, and emerging markets 25%. Alcon's global presence enhances TOBREX's reach, although generic competition in certain markets pressures pricing.
What Are Future Sales Projections?
- Growth Rate: The ophthalmic antibiotic market grows at 3-5% annually, driven by increased infection prevalence.
- Potential for Expansion: Patent expirations in some regions may lead to generic entry, pressuring prices but expanding volume.
- Innovations and Combination Therapies: Development of combination drugs and new formulations could impact sales positively or negatively.
Projected sales for TOBREX could reach approximately $25 million in 2025, assuming moderate market growth and limited generic competition. If generic versions gain market share rapidly, sales could decline by 10-15% annually post-2025.
What Are the Key Risks and Opportunities?
- Risks: Price erosion due to generics, regional regulatory changes, and competition from newer antibiotics.
- Opportunities: Expansion into emerging markets, development of combination formulations, and potential label expansions for other ocular infections.
Summary of Sales Projections and Market Outlook
| Year |
Estimated Global Sales (Millions USD) |
Comments |
| 2022 |
20-45 |
Current sales, dominated by North America |
| 2023 |
22-48 |
Slight growth, stable market conditions |
| 2024 |
24-52 |
Continued growth, potential new markets |
| 2025 |
25-55 |
Approaching market saturation, noting patent/exclusivity considerations |
Key Takeaways
- TOBREX holds a niche but stable position in the ophthalmic antibiotic market.
- Market sales are driven by bacterial eye infection prevalence and awareness.
- Competition from generics and alternative drugs threaten future profitability.
- Growth prospects depend on expanding indications and geographic markets.
- Market share losses post-generic entry could reduce sales significantly.
FAQs
1. What factors influence TOBREX’s market share?
Market share depends on infection rates, competition from generics, pricing strategies, and regional regulatory approvals.
2. How does generic competition impact TOBREX sales?
Entry of generic tobramycin eyedrops can lead to price decreases and volume increases but may reduce Alcon's profit margins and overall market share.
3. Are there upcoming regulatory hurdles for TOBREX?
Regulatory pathways could tighten in regions with increased oversight on ophthalmic antibiotics, potentially delaying launches or leading to market withdrawal.
4. What market segments are most promising for expansion?
Emerging markets with rising infection rates, as well as development of combination therapies, represent potential growth areas.
5. How does the rise of antibiotic resistance affect TOBREX?
Resistance development may limit the effectiveness of TOBREX, leading to decreased usage unless new formulations or indications are approved.
Citations
[1] MarketWatch. "Global Ophthalmic Antibiotics Market Size." 2022.
[2] IQVIA. "Pharmaceutical Market Data." 2022.
[3] Novartis. "Alcon Products Portfolio." 2023.