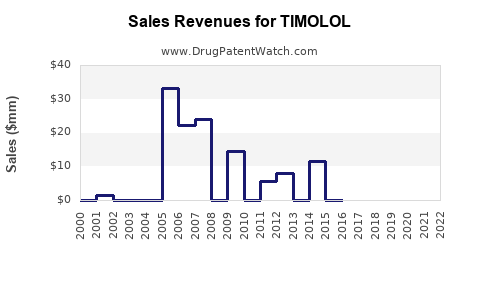
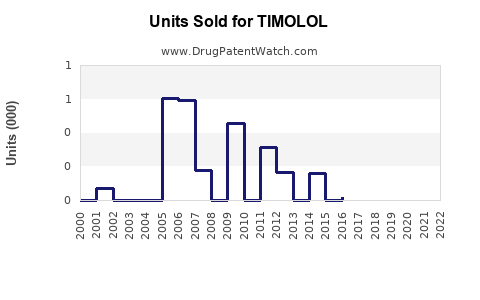
Last updated: February 20, 2026
What is the Current Market Size for Timolol?
Timolol is a non-selective beta-adrenergic receptor blocker primarily used to treat ocular hypertension, open-angle glaucoma, and sometimes systemic conditions like migraine prophylaxis. The global market for timolol eye drops was valued at approximately USD 1.2 billion in 2022. It accounts for a significant share of the anti-glaucoma medication sector, which is projected to grow at a compound annual growth rate (CAGR) of about 4.4% over the next five years.
The key markets include North America, Europe, and Asia-Pacific. North America holds the largest share, driven by high prevalence of glaucoma (approximately 3 million in the US alone) and the widespread use of combination therapies. Europe follows, with increasing adoption of ophthalmic treatments. The Asia-Pacific region shows rapid growth due to rising awareness, healthcare infrastructure improvements, and increasing prevalence of eye diseases.
What Are Driving Factors for Market Growth?
- Increasing glaucoma prevalence: Globally, the number of people with glaucoma is expected to reach 112 million by 2040, up from 76 million in 2020 [1].
- Chronic nature of ocular hypertension: Long-term management sustains steady demand.
- Development of generic formulations: Patent expirations have allowed entry of generics, reducing prices and expanding access.
- Advancements in delivery methods: Eye drop formulations improve patient compliance.
- Emerging markets: Growing healthcare expenditure and awareness in developing economies increase market penetration.
What Are the Key Competitive Landscape Dynamics?
The market is highly consolidated, with top players including:
| Company |
Market Share (Approximate) |
Notable Contributions |
| Novartis |
40% |
Leading with brand-name products like Timoptol. |
| Santen Pharmaceutical |
20% |
Focus on ophthalmic and CNS treatments. |
| Sandoz (Novartis generic division) |
15% |
Significant generic offerings. |
| Others |
25% |
Includes smaller pharma companies and generics. |
Novartis holds a dominant position as the primary provider of branded timolol formulations. The generic segment, led by Sandoz, accelerates competitive pressure and price erosion.
What Are Sales Projections for the Next Five Years?
Based on current trends, global sales of timolol eye drops are expected to reach USD 1.5 billion by 2027. This reflects a CAGR of around 4% from 2022 to 2027.
| Year |
Projected Market Size (USD billions) |
| 2023 |
1.27 |
| 2024 |
1.32 |
| 2025 |
1.36 |
| 2026 |
1.42 |
| 2027 |
1.50 |
Regional growth rates vary: North America maintains a 4.0-4.5% CAGR, Europe approximately 3.5%, while Asia-Pacific leads with upwards of 6% driven by increasing healthcare investments.
What Are Barriers and Risks?
- Patent expirations: Accelerate generic competition, pressure prices downward.
- Regulatory changes: Influence approval processes for new formulations or delivery devices.
- Market saturation: Especially in developed economies with high existing adoption.
- Alternative treatments: Such as prostaglandin analogs or surgical interventions gaining popularity.
What Is the Outlook for New Formulations or Labels?
Innovations like sustained-release formulations and combination eye drops aim to improve patient compliance. These could rejuvenate growth, especially if approved for broader indications or lower scheduled dosing.
Final Thoughts
Timolol remains a core therapy for glaucoma with stable long-term demand. The market will expand, supported by demographic trends and generics. The competitive landscape has consolidated quickly, with growth driven by volume expansion and pricing strategies.
Key Takeaways
- The global timolol eye drop market was valued at USD 1.2 billion in 2022.
- Expected to reach USD 1.5 billion by 2027, with a CAGR of approximately 4%.
- North America leads, but Asia-Pacific shows increasing growth potential.
- Patent expirations and generics are central to market dynamics, influencing pricing and competition.
- Innovation in delivery systems may offer growth opportunities.
FAQs
1. How does patent expiry impact timolol sales? Patent expirations allow generic manufacturers to enter the market, increasing supply and reducing prices, which can lower brand-name sales but expand overall market volume.
2. What are the main competitors to timolol in glaucoma treatment? Prostaglandin analogs like latanoprost and travoprost have gained popularity due to better tolerability, potentially limiting timolol’s growth.
3. Are there emerging markets for timolol? Yes. Countries like India, China, and Brazil are increasing their glaucoma treatment penetration as healthcare infrastructure improves.
4. How do newer formulations influence the market? Sustained-release and combination therapies may extend market lifetime for timolol formulations, especially if they demonstrate improved adherence.
5. What is the main regulatory challenge for timolol? Ensuring formulations meet quality standards across diverse markets and gaining approval for innovative delivery methods.
References
[1] Tham, Y. C., et al. (2020). Global prevalence of glaucoma and projections. Lancet Global Health, 8(6), e1079–e1080.