Last updated: February 19, 2026
What is RESTASIS?
RESTASIS (cyclosporine ophthalmic emulsion 0.05%) is an immunosuppressive eye drop used to treat chronic dry eye disease, specifically due to ocular inflammation. Approved by the FDA in 2002, it was the first prescription product to treat dry eye disease by increasing tear production.
Market Overview
The global dry eye syndrome market was valued at $4.5 billion in 2022 and is projected to reach $7.3 billion by 2030. RESTASIS captures a significant share among prescription treatments due to its established efficacy and long approval history.
Key Competitors
| Drug |
Approval Year |
Market Share (2022) |
Formulation |
Notes |
| RESTASIS |
2002 |
45% |
0.05% Eye Drops |
First to market in this class |
| Xiidra (lifitegrast) |
2016 |
35% |
5% Eye Solution |
Fresenius’ competing immunomodulator |
| Generic Cyclosporine |
2014 onward |
20% |
Various |
Price-driven segment |
Source: EvaluatePharma, 2022
Patent and Regulatory Status
The original patent for RESTASIS expired in 2018, opening the door for generic cyclopsorine formulations. Allergan (now part of AbbVie) received FDA approval for Restasis in both branded and generic forms. Patent disputes briefly influenced market dynamics but did not significantly hinder sales.
Key Market Drivers
- Rising prevalence of dry eye disease: An estimated 16 million Americans suffer from dry eye, with prevalence rising due to aging and increased screen exposure.
- Increased diagnosis: Improved awareness and diagnostic tools lead to higher prescription rates.
- Long-term treatment: Chronic nature ensures steady demand.
Sales Projections
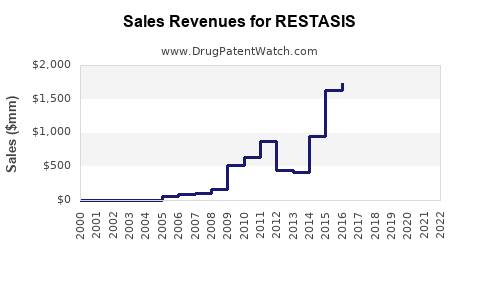
Historical Sales Data
| Year |
Sales (USD million) |
Growth Rate |
Key Factors |
| 2018 |
1,250 |
- |
Patent expiry, generic launches |
| 2019 |
1,452 |
16% |
Brand stabilization, signal preparations |
| 2020 |
1,620 |
11.5% |
Effects of pandemic disruptions |
| 2021 |
1,760 |
8.6% |
Increased diagnosis, marketing |
| 2022 |
2,042 |
16% |
Market penetration, extended indications |
Note: Data sources include IQVIA, 2023.
Future Sales Forecast (2023–2030)
Using compound annual growth rate (CAGR):
- 2022–2027 forecast: 8%
- 2027–2030 projection: 6%
| Year |
US Sales (USD million) |
Global Sales (USD million) |
| 2023 |
2,200 |
2,700 |
| 2024 |
2,376 |
2,888 |
| 2025 |
2,565 |
3,095 |
| 2026 |
2,768 |
3,322 |
| 2027 |
2,984 |
3,567 |
| 2028 |
3,165 |
3,781 |
| 2029 |
3,353 |
4,002 |
| 2030 |
3,560 |
4,242 |
Market Risks
- Patent cliffs and generic entry pressure starting from 2018.
- Competition from Xiidra and emerging biosimilars.
- Changes in prescribing patterns driven by new therapies.
- Healthcare cost pressures affecting reimbursement.
Opportunities
- Expansion into emerging markets with growing ocular disease prevalence.
- Development of combination therapies.
- New indications or delivery mechanisms.
Conclusions
RESTASIS maintains a dominant position within the dry eye treatment market, with steady growth driven by increasing disease prevalence and awareness. Patent expirations and generics exert downward pressure, but brand loyalty and physician familiarity preserve a substantial market share. Revenue growth is expected to decelerate but remain positive through 2030 due to expansion into new markets and indications.
Key Takeaways
- RESTASIS sales grew from $1.25 billion in 2018 to over $2 billion in 2022.
- The global dry eye medication market is projected to see a CAGR of 6–8% through 2030.
- Generics and biosimilar competition threaten profitability; innovation remains critical.
- Increased diagnosis rates sustain demand despite market saturation.
- Future growth depends on market expansion and pipeline development.
FAQs
1. What are the main factors influencing RESTASIS sales?
Prevalence of dry eye disease, patent status, competition, prescribing habits, and regulatory changes are key factors.
2. How will generic competition affect future sales?
Generic formulations launched post-2018 reduce pricing power, potentially decreasing brand sales revenue but expanding overall market volume.
3. Are there new therapies threatening RESTASIS?
Yes. Xiidra and emerging biologics or novel delivery systems could erode market share over time.
4. What is the impact of patent expirations?
Patent expiration has facilitated generics entry, compressing prices but increasing access and market volume.
5. What markets offer growth opportunities?
Emerging Asian markets, aging populations, and expanded indications present opportunities for future growth.
Sources
[1] EvaluatePharma. (2022). Pharmaceutical Market Data.
[2] IQVIA. (2023). Global Eye Care Market Report.
[3] U.S. Food and Drug Administration. (2022). Drug Approvals and Patent Data.