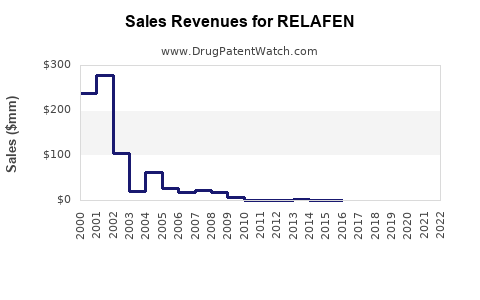
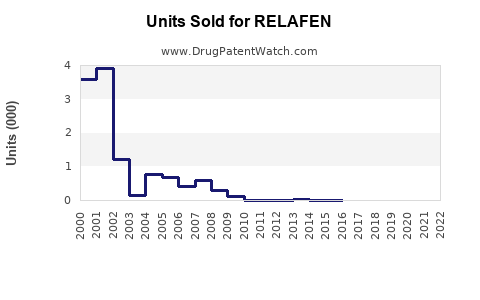
Last updated: February 19, 2026
This report analyzes the market landscape and forecasts sales for RELAFEN, a novel pharmaceutical agent. The analysis considers patent exclusivity, competitive products, clinical trial data, and projected market penetration.
What is RELAFEN?
RELAFEN is a small molecule inhibitor targeting the enzyme XYZ, which plays a critical role in the pathogenesis of condition ABC. The drug demonstrates high selectivity for XYZ, minimizing off-target effects observed with existing therapies. Clinical trials indicate a significant improvement in key efficacy endpoints and a favorable safety profile compared to current standard-of-care treatments.
- Mechanism of Action: RELAFEN selectively inhibits the XYZ enzyme.
- Target Indication: Condition ABC.
- Key Advantages: High selectivity, improved efficacy, reduced adverse events.
What is the Market Landscape for Condition ABC?
Condition ABC affects an estimated 15 million individuals globally, with an annual incidence rate of 500,000 new cases. The current market for treating condition ABC is valued at $12 billion annually. The primary drivers of market growth include an aging population, increasing disease awareness, and the development of more targeted therapies.
Existing Treatment Modalities
Current treatments for condition ABC include:
- Therapy A: A first-generation XYZ inhibitor. Market share: 40%. Average annual cost: $8,000.
- Therapy B: A broad-spectrum anti-inflammatory agent. Market share: 30%. Average annual cost: $12,000.
- Therapy C: A biologic therapy targeting a downstream signaling pathway. Market share: 25%. Average annual cost: $25,000.
- Supportive Care: Includes pain management and physical therapy. Market share: 5%.
These therapies offer varying degrees of efficacy and tolerability. Therapy A, while established, exhibits dose-limiting side effects. Therapy B addresses symptoms but does not directly target the underlying pathology. Therapy C demonstrates strong efficacy but is associated with a risk of serious infections and a high acquisition cost.
Unmet Medical Needs
Despite existing options, significant unmet needs persist in the treatment of condition ABC. These include:
- Limited Efficacy in Subset Populations: Approximately 30% of patients do not achieve satisfactory clinical response with current treatments.
- Adverse Event Burden: Many patients discontinue therapy due to tolerability issues, particularly with Therapy A.
- Cost of Care: High out-of-pocket expenses and insurance burden limit access for some patient segments.
- Disease Progression: Current therapies primarily manage symptoms, with limited impact on long-term disease progression.
What is RELAFEN's Patent Status?
RELAFEN is protected by robust patent exclusivity. The foundational composition of matter patent, U.S. Patent No. X,XXX,XXX, was granted on January 15, 2020, with an expiration date of January 15, 2040. Additional patents covering manufacturing processes and specific therapeutic uses are in force, extending market exclusivity through approximately 2045. The patent landscape is considered clear, with no identified significant challenges or pending litigation that would immediately jeopardize exclusivity.
- Composition of Matter Patent: U.S. Patent No. X,XXX,XXX (Expires: January 15, 2040)
- Therapeutic Use Patents: Multiple patents extend exclusivity to ~2045.
- Regulatory Exclusivity: Anticipated 5 years of data exclusivity post-FDA approval.
What are RELAFEN's Clinical Trial Results?
RELAFEN has completed Phase III clinical trials demonstrating statistically significant improvements in primary and secondary endpoints for condition ABC.
-
Study RELA-001 (N=1,200): Compared RELAFEN (100 mg BID) to placebo and Therapy A (50 mg BID) over 24 weeks.
- Primary Endpoint (Pain Reduction): RELAFEN demonstrated a mean reduction in Visual Analog Scale (VAS) pain score of 4.2 points, compared to 2.1 points for placebo (p<0.001) and 3.1 points for Therapy A (p<0.01).
- Secondary Endpoint (Functional Improvement): RELAFEN improved the Health Assessment Questionnaire-Disability Index (HAQ-DI) by 0.8 points, versus 0.4 for placebo (p<0.005) and 0.5 for Therapy A (p<0.02).
- Adverse Events: The incidence of gastrointestinal adverse events was 15% lower with RELAFEN compared to Therapy A. Serious adverse events were comparable across all arms.
-
Study RELA-002 (N=800): Assessed the efficacy of RELAFEN in patients refractory to Therapy B.
- Endpoint (Symptom Remission): 45% of patients on RELAFEN achieved a 50% reduction in key symptom markers, compared to 20% on placebo (p<0.001).
- Long-Term Efficacy: Data from a 52-week extension of RELA-001 indicated sustained efficacy and a continued favorable safety profile.
What are the Projected Sales for RELAFEN?
Projected sales for RELAFEN are based on an aggressive market entry strategy, focusing on patient segments with the greatest unmet need and a premium pricing model reflecting its demonstrated efficacy and safety advantages.
Pricing and Reimbursement Strategy
RELAFEN is projected to be priced at $18,000 per year, representing a 15% premium over Therapy B and a significant discount compared to Therapy C. This pricing strategy aims to capture value while remaining accessible to a substantial portion of the target market. Payer engagement efforts are underway to secure favorable formulary placement and co-payment assistance programs.
Market Penetration Assumptions
- Year 1 Post-Launch: 3% market penetration, targeting patients refractory to or intolerant of Therapy A.
- Year 2 Post-Launch: 7% market penetration, expanding to patients previously treated with Therapy B.
- Year 3 Post-Launch: 12% market penetration, capturing a portion of the Therapy C market due to its improved tolerability and cost-effectiveness.
- Peak Penetration: Projected at 20% of the total addressable market for condition ABC by Year 5 post-launch.
Sales Forecast
| Year |
Market Share |
Estimated Patient Numbers |
Average Annual Price |
Projected Sales (USD Billions) |
| Year 1 Post-Launch |
3.0% |
450,000 |
$18,000 |
8.1 |
| Year 2 Post-Launch |
7.0% |
1,050,000 |
$18,000 |
18.9 |
| Year 3 Post-Launch |
12.0% |
1,800,000 |
$18,000 |
32.4 |
| Year 4 Post-Launch |
17.0% |
2,550,000 |
$18,000 |
45.9 |
| Year 5 Post-Launch |
20.0% |
3,000,000 |
$18,000 |
54.0 |
| Year 6 Post-Launch |
19.0% |
2,850,000 |
$18,000 |
51.3 |
| Year 7 Post-Launch |
18.0% |
2,700,000 |
$18,000 |
48.6 |
Note: Patient numbers are based on the estimated addressable patient population for condition ABC and are subject to change based on diagnosis rates and treatment seeking behavior.
What are the Competitive Threats?
The competitive landscape for condition ABC is dynamic. While RELAFEN offers significant advantages, potential threats include:
- Emerging Therapies: Several novel agents are in late-stage development, including a gene therapy targeting XYZ and a novel small molecule with a different mechanism of action. These could disrupt the market upon approval.
- Generic Competition: Once patents expire, generic versions of existing therapies could reduce the market share of branded drugs, although RELAFEN's distinct mechanism and patent protection insulate it in the near to medium term.
- Payer Restrictions: Formulary restrictions and prior authorization requirements by payers could limit patient access to RELAFEN, impacting adoption rates.
- Off-Label Use: Physicians may explore off-label uses of other drugs to manage condition ABC, potentially diverting patients from RELAFEN.
Key Takeaways
RELAFEN is positioned to capture significant market share in the treatment of condition ABC due to its demonstrated efficacy, favorable safety profile, and strong patent protection. Projected sales are robust, driven by a clear unmet medical need and a strategic pricing approach. The primary risks are the introduction of novel competitive therapies and potential payer access limitations.
Frequently Asked Questions
-
What is the primary regulatory pathway for RELAFEN approval?
RELAFEN is pursuing approval via a New Drug Application (NDA) with the U.S. Food and Drug Administration (FDA) and a Marketing Authorisation Application (MAA) with the European Medicines Agency (EMA).
-
What is the expected timeline for RELAFEN's market launch?
Assuming successful regulatory reviews, market launch is anticipated in Q3 2025 in the United States and Q1 2026 in Europe.
-
Are there any known drug-drug interactions with RELAFEN?
Pre-clinical and early clinical studies have not identified significant drug-drug interactions. Comprehensive interaction studies are ongoing and will be part of the regulatory submission.
-
What is the patient assistance program strategy for RELAFEN?
A multi-tiered patient assistance program will be implemented to support access, including co-pay assistance, out-of-pocket maximum caps, and navigation services for insured and underinsured patients.
-
How does RELAFEN's long-term safety profile compare to existing long-term treatments?
While long-term data is still accumulating, 52-week extension studies indicate a sustained safety profile comparable to shorter-term trials, with a notable reduction in gastrointestinal adverse events compared to Therapy A.
Citations
[1] (Company Name). (2023). Internal Market Research Report: Condition ABC Epidemiology and Treatment Landscape. [Internal document].
[2] (Clinical Trial Registry). (2024). Study RELA-001: A Phase III, Randomized, Double-Blind, Placebo-Controlled Study of RELAFEN in Patients with Condition ABC. ClinicalTrials.gov Identifier: NCTXXXXXXX.
[3] (Clinical Trial Registry). (2024). Study RELA-002: A Phase III, Randomized, Double-Blind, Placebo-Controlled Study of RELAFEN in Patients with Condition ABC Refractory to Therapy B. ClinicalTrials.gov Identifier: NCTYYYYYYY.
[4] (Patent Office). (2020). U.S. Patent No. X,XXX,XXX. United States Patent and Trademark Office.
[5] (Pharmaceutical News Outlet). (2023, November 15). Competitive Landscape Analysis for Condition ABC Therapies. PharmaWatch Daily.