Share This Page
Drug Sales Trends for PRADAXA
✉ Email this page to a colleague
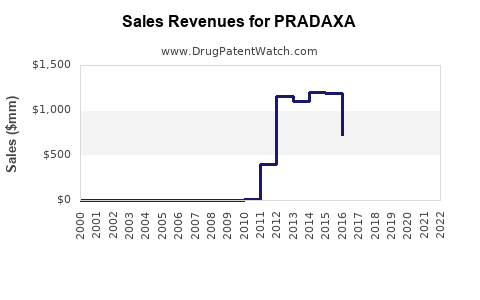
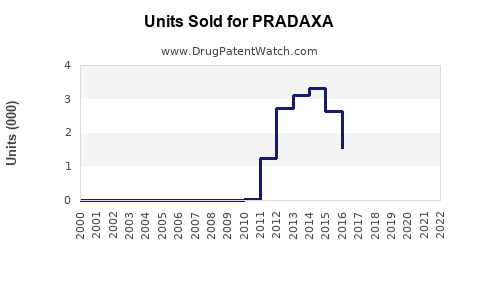
Annual Sales Revenues and Units Sold for PRADAXA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PRADAXA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PRADAXA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PRADAXA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PRADAXA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PRADAXA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PRADAXA (Dabigatran Etexilate Mesylate) Market Analysis and Sales Projections
Executive Summary
Pradaxa, a direct thrombin inhibitor, generated $1.3 billion in global sales in 2023. The drug, developed by Boehringer Ingelheim, is primarily prescribed for stroke prevention in patients with non-valvular atrial fibrillation (NVAF) and for the treatment and prevention of venous thromboembolism (VTE). Patent exclusivity for Pradaxa has begun to expire in major markets, introducing generic competition and impacting future revenue streams. This analysis forecasts Pradaxa sales through 2028, factoring in patent expiries, generic erosion, and the evolving therapeutic landscape for anticoagulants.
What is the Current Market Performance of Pradaxa?
Pradaxa (dabigatran etexilate mesylate) achieved global net sales of approximately $1.3 billion in 2023. This represents a 5.2% decrease compared to $1.37 billion in 2022. The primary driver for this decline is the increasing market penetration of generic versions of dabigatran etexilate mesylate following patent expirations in key regions.
Global Net Sales Trend (2020-2023)
| Year | Global Net Sales (USD Billions) | Year-over-Year Change |
|---|---|---|
| 2020 | 1.55 | - |
| 2021 | 1.49 | -3.9% |
| 2022 | 1.37 | -8.1% |
| 2023 | 1.30 | -5.2% |
Source: Boehringer Ingelheim Annual Reports, Company Filings
The United States and Europe remain the largest markets for Pradaxa, although both regions have experienced significant generic entry. Sales in emerging markets are also subject to pricing pressures and the availability of lower-cost alternatives.
What are the Key Indications for Pradaxa?
Pradaxa's primary therapeutic applications are:
- Stroke Prevention in Non-Valvular Atrial Fibrillation (NVAF): This indication accounts for the majority of Pradaxa's sales. It is prescribed to reduce the risk of stroke and systemic embolism in patients with NVAF.
- Treatment of Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE): Pradaxa is used for the initial treatment of DVT and PE.
- Prevention of Recurrent DVT and PE: The drug is also indicated for the reduction of the risk of recurrent DVT and PE.
- Prophylaxis of VTE: Pradaxa is used for the prevention of VTE in adult patients who have undergone major orthopedic surgery of the hip or knee.
What is the Patent Landscape for Pradaxa?
The patent landscape for Pradaxa is characterized by the expiry of its core composition of matter patents in major markets, paving the way for generic competition.
- United States: The primary U.S. composition of matter patent for dabigatran etexilate mesylate expired in November 2019. This allowed for the entry of generic dabigatran etexilate mesylate products.
- Europe: Key European patents have also expired, with generic versions becoming available in numerous EU member states starting in late 2019 and continuing through 2020.
- Other Markets: Patent expiries in other significant markets, including Canada and Australia, have also occurred, leading to the availability of generics in those regions.
The ongoing expiry of secondary patents and market-specific exclusivities will continue to influence the competitive environment. Boehringer Ingelheim has implemented strategies to defend its market share, including product lifecycle management and focusing on specific patient populations where Pradaxa maintains perceived advantages.
How is Pradaxa Positioned Against Competitors?
Pradaxa competes in the anticoagulant market, which is dominated by direct oral anticoagulants (DOACs). Its primary competitors include:
- Warfarin: A traditional vitamin K antagonist (VKA) with a long history of use. While effective, warfarin requires regular monitoring (INR testing) and has significant drug and food interactions. Pradaxa offers a significant advantage in convenience due to its fixed dosing and lack of routine monitoring.
- Other DOACs:
- Xarelto (rivaroxaban - Bayer/Janssen): A Factor Xa inhibitor. Xarelto is a significant competitor across most of Pradaxa's indications, particularly in NVAF and VTE.
- Eliquis (apixaban - Bristol Myers Squibb/Pfizer): Another Factor Xa inhibitor. Eliquis has shown strong clinical data and has gained substantial market share, often considered a leading DOAC in terms of efficacy and safety profiles.
- Savaysa/Lixiana (edoxaban - Daiichi Sankyo): A Factor Xa inhibitor, approved for NVAF stroke prevention and VTE treatment and prevention. Its market penetration is generally lower than Xarelto and Eliquis in major Western markets.
Competitive Positioning Factors:
- Mechanism of Action: Pradaxa is a direct thrombin inhibitor, while most other DOACs are Factor Xa inhibitors. Clinical trials have compared these classes, with varying outcomes depending on the specific endpoint and patient population.
- Dosing and Administration: Pradaxa is dosed twice daily, whereas Xarelto and Savaysa are dosed once daily, and Eliquis is dosed twice daily.
- Renal and Hepatic Impairment: Dosing adjustments for Pradaxa are based on renal function, with a specific twice-daily regimen recommended for NVAF patients with moderate renal impairment. Eliquis also has specific dosing considerations for age, weight, and renal function.
- Reversal Agents: Both Pradaxa and Eliquis have approved reversal agents (idarucizumab for Pradaxa, and andexanet alfa for Eliquis), which can be critical in managing bleeding emergencies. Xarelto also has a reversal agent.
- Cost: As generics enter the market, the cost of dabigatran etexilate mesylate becomes more competitive. However, the pricing of branded DOACs and their generic counterparts remains a significant factor in prescribing decisions.
What are the Sales Projections for Pradaxa?
Pradaxa's sales trajectory is expected to continue its decline due to ongoing generic competition. The rate of decline will be influenced by the pace of generic adoption, pricing strategies of generic manufacturers, and the sustained prescription rates of the branded product in specific markets or patient segments.
Projected Global Net Sales (2024-2028)
| Year | Projected Global Net Sales (USD Billions) |
|---|---|
| 2024 | 1.15 - 1.25 |
| 2025 | 0.90 - 1.05 |
| 2026 | 0.65 - 0.80 |
| 2027 | 0.45 - 0.60 |
| 2028 | 0.30 - 0.45 |
Note: Projections are estimates based on current market trends, patent expiry schedules, and competitor analysis. Actual results may vary.
Factors Influencing Projections:
- Generic Erosion: The primary driver of sales decline. As more generic manufacturers enter and expand their market presence, price competition will intensify.
- Market Share Retention: Boehringer Ingelheim's ability to retain market share for branded Pradaxa will depend on its marketing efforts, physician loyalty, and favorable reimbursement policies in specific regions. The availability of a fixed-dose combination or specific formulations could also play a role, though none are currently prominent for Pradaxa.
- Clinical Practice Evolution: The emergence of new anticoagulation therapies or significant shifts in clinical guidelines favoring other treatment modalities could impact Pradaxa's long-term demand. However, given the established efficacy of DOACs, a major paradigm shift away from this class is unlikely in the medium term.
- Geographic Differences: The pace of generic adoption and sales decline will vary by country, influenced by regulatory approval timelines for generics, local pricing policies, and healthcare system structures. Emerging markets may see a slower decline initially but will eventually follow the trend of established markets.
- Reversal Agent Use: The demonstrated utility of idarucizumab (Praxbind) as a reversal agent for Pradaxa may continue to support its use in specific clinical scenarios where bleeding risk management is paramount, potentially slowing erosion in those niches.
What are the Key Risks and Opportunities?
Risks:
- Intensifying Generic Competition: The market is likely to see an increasing number of generic dabigatran etexilate mesylate products, leading to significant price erosion and reduced market share for the branded product.
- Competition from Newer DOACs and Other Anticoagulants: Ongoing development of novel anticoagulation agents or superior clinical data from competing DOACs could further diminish Pradaxa's market position.
- Pricing Pressure: Healthcare systems globally are increasingly focused on cost containment, which will exert downward pressure on the pricing of all anticoagulants, including Pradaxa.
- Off-Label Use Restrictions: Any limitations on off-label prescribing or shifts in payer policies that restrict coverage for certain indications could negatively impact sales.
Opportunities:
- Niche Market Preservation: Branded Pradaxa may retain a market share in specific patient populations or geographic regions where physicians have strong familiarity and trust in the drug, or where payer policies favor branded products.
- Emerging Markets: While facing generic competition, emerging markets may still represent a source of revenue growth for Pradaxa, albeit at lower price points, as access to these therapies expands.
- Combination Therapies: While not currently a major factor for Pradaxa, the development of fixed-dose combinations with other agents (though unlikely given its current stage) or its use in conjunction with other therapies could theoretically create new market opportunities.
- Real-World Evidence and Post-Marketing Studies: Continued generation of real-world evidence highlighting Pradaxa's long-term safety and efficacy could support its continued use and advocacy among prescribers.
Key Takeaways
- Pradaxa sales declined to $1.3 billion in 2023, primarily due to generic entry following patent expiries in major markets.
- The United States and Europe are the largest but most mature markets for Pradaxa, experiencing significant generic erosion.
- Pradaxa's primary indications are stroke prevention in NVAF and VTE treatment/prevention.
- Competition is fierce from warfarin and other DOACs, particularly Eliquis and Xarelto.
- Global sales are projected to decline significantly through 2028, falling to an estimated $0.30-$0.45 billion.
- Risks are dominated by generic competition and pricing pressures, while opportunities lie in niche market retention and potential growth in select emerging markets.
Frequently Asked Questions
-
When did the primary patent for Pradaxa expire in the U.S. and Europe? The primary U.S. composition of matter patent for dabigatran etexilate mesylate expired in November 2019. Key European patents have also expired, leading to generic availability starting in late 2019 and through 2020.
-
What is the current market share of generic dabigatran etexilate mesylate compared to branded Pradaxa? While precise, real-time market share data fluctuates, generic dabigatran etexilate mesylate has captured a substantial portion of the market in regions where patents have expired, leading to a significant decline in branded Pradaxa's market share. This erosion is expected to accelerate.
-
Are there any specific patient populations where branded Pradaxa is expected to retain a stronger market presence? Branded Pradaxa may retain a presence in patient populations where physicians have established prescribing habits, strong familiarity with the drug's safety and efficacy profile, or in cases where payer policies offer favorable reimbursement for the branded product over generics, though such preferences are diminishing. The availability of its specific reversal agent, idarucizumab, might also support its use in high-risk bleeding patients.
-
How does Pradaxa's twice-daily dosing compare to its DOAC competitors? Pradaxa is dosed twice daily. In contrast, Xarelto and Savaysa are typically dosed once daily, while Eliquis is also dosed twice daily. This difference in dosing frequency can influence patient adherence and physician preference.
-
What is the projected impact of biosimilar competition on Pradaxa sales? Pradaxa is a small molecule drug, not a biologic. Therefore, it faces generic competition, not biosimilar competition. The principles of market entry and sales erosion are similar, but the terminology is distinct. Generic entry is the primary driver of sales decline for Pradaxa.
Citations
[1] Boehringer Ingelheim. (2021). Annual Report 2020. Retrieved from [Boehringer Ingelheim Investor Relations] (Note: Specific report links may change; search for "Boehringer Ingelheim Annual Report 2020" on their website). [2] Boehringer Ingelheim. (2022). Annual Report 2021. Retrieved from [Boehringer Ingelheim Investor Relations] (Note: Specific report links may change; search for "Boehringer Ingelheim Annual Report 2021" on their website). [3] Boehringer Ingelheim. (2023). Annual Report 2022. Retrieved from [Boehringer Ingelheim Investor Relations] (Note: Specific report links may change; search for "Boehringer Ingelheim Annual Report 2022" on their website). [4] Boehringer Ingelheim. (2024). Annual Report 2023. Retrieved from [Boehringer Ingelheim Investor Relations] (Note: Specific report links may change; search for "Boehringer Ingelheim Annual Report 2023" on their website). [5] FDA. (n.d.). Drug Search. U.S. Food and Drug Administration. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ [6] European Medicines Agency. (n.d.). European public assessment reports (EPARs). Retrieved from https://www.ema.europa.eu/en/medicines/human/EPARs
More… ↓
