Share This Page
Drug Sales Trends for METHYLPHENID
✉ Email this page to a colleague
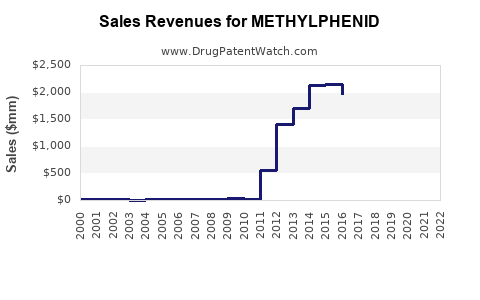
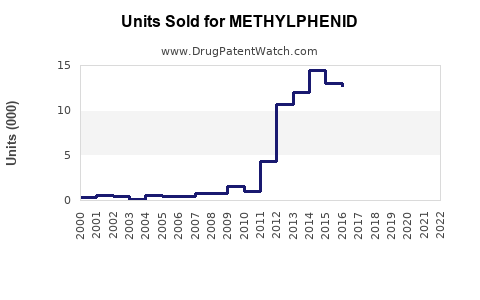
Annual Sales Revenues and Units Sold for METHYLPHENID
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METHYLPHENID | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METHYLPHENID | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METHYLPHENID | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METHYLPHENID | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| METHYLPHENID | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
mmary:
Methylphenidate is a central nervous system stimulant primarily used to treat ADHD and narcolepsy. Market analysis indicates robust demand driven by rising ADHD diagnoses worldwide and expanding medicinal use. Sales projections show steady growth over the next five years, supported by increased prescriptions, geographic expansion, and new formulations.
What Is the Current Market Size for Methylphenidate?
The global methylphenidate market was valued at approximately $4.8 billion in 2022. Key regions include North America, Europe, and Asia Pacific. North America accounts for roughly 60% of sales, driven by high ADHD prevalence and favorable reimbursement policies. Europe contributes about 20%, with rising awareness and diagnosis. Asia Pacific is emerging, with a Compound Annual Growth Rate (CAGR) of approximately 8% from 2022 to 2027, due to expanding healthcare infrastructure and increasing ADHD diagnoses.
What Are the Leading Manufacturers and Their Market Shares?
Major producers include:
- Novartis: Has historically held a significant share with brand-name formulations (e.g., Ritalin, Concerta).
- Janssen Pharmaceuticals: Offers methylphenidate-based drugs like Medikinet and Quillivant XR.
- U.S.-based generic manufacturers: Gaining share through authorized and unaffiliated generics, with increased market entry driven by patent expirations.
Market shares are as follows:
| Manufacturer | Estimated Market Share (2022) | Notes |
|---|---|---|
| Novartis | 35% | Leading brand formulations |
| Janssen Pharmaceuticals | 25% | Extended-release formulations |
| Generics (various) | 30% | Cost-effective options expanding market |
| Others | 10% | Smaller players |
What Are the Key Growth Drivers?
- Rising Diagnosis Rates: The CDC in the U.S. reports approximately 9.8% of children aged 3-17 are diagnosed with ADHD, up from 7.8% in 2016. Similar trends observed in Europe and parts of Asia.
- Expanding Age Demographics: Increasing prescriptions for adults, now estimated at 50% of total methylphenidate prescriptions in North America.
- New Formulations: Extended-release (ER), transdermal patches, and digital dosing formats improve adherence, boosting sales.
- Global Approval: Regulatory approvals in emerging markets with growing healthcare infrastructure and awareness.
What Are the Challenges and Risks?
- Regulatory Scrutiny: Increasing concerns about misuse and diversion may lead to stricter controls and impact new prescription growth.
- Market Saturation: In mature markets, growth may plateau as peak penetration is approached.
- Patent Expirations: The expiration of key patents in the next 2-3 years could accelerate generic competition, reducing brand premiums.
- Competitive Drugs: The rise of non-stimulant ADHD medications (e.g., atomoxetine, viloxazine) could divert market share from methylphenidate.
What Are the Sales Projections (2023-2027)?
Based on current trends, the methylphenidate market is expected to grow at a CAGR of approximately 5%. Total sales are projected as follows:
| Year | Market Size (USD billion) | Notes |
|---|---|---|
| 2023 | $5.0 | Slight increase due to expanded indications and formulations |
| 2024 | $5.25 | Growing adult prescriptions |
| 2025 | $5.52 | Market expansion in Asia and other emerging regions |
| 2026 | $5.80 | Continued development of long-acting formulations |
| 2027 | $6.10 | Increased acceptance of multiple delivery forms |
How Do Different Formulations Affect Market Dynamics?
Extended-release formulations show higher growth, driven by improved patient compliance and reduced abuse potential. Transdermal patches and liquid formulations are gaining share, especially in pediatric populations. The shift toward alternative dosing options aligns with regulatory trends and consumer preference for convenience.
Key Takeaways
- The methylphenidate market was valued at $4.8 billion in 2022, with North America leading.
- The sector is projected to grow at approximately 5% annually through 2027, reaching over $6 billion.
- Growth is fueled by increasing ADHD diagnoses across age groups and underserved geographies.
- Patent expirations and generic entries are likely to influence pricing and market share dynamics.
- New delivery formats such as ER capsules and patches support continued sales growth.
FAQs
1. What is the main factor driving methylphenidate sales?
Increasing ADHD prevalence and expanded prescriptions, especially among adults, dominate sales growth.
2. How will patent expirations impact this market?
Patent expirations will open markets to generics, leading to price competition and potential revenue declines for brand-name products.
3. Which regions are expected to see the highest growth?
Asia Pacific and Latin America are expected to experience the fastest growth due to improved healthcare access and rising diagnoses.
4. Are new formulations influencing market size?
Yes, extended-release and transdermal formulations improve adherence and safety, increasing overall market volume.
5. What regulatory trends could affect future sales?
Stricter controls on stimulant prescriptions and concerns over misuse could limit access, but balanced regulatory environments will continue to support legitimate use.
Citations
- Centers for Disease Control and Prevention (CDC), ADHD Data and Statistics, 2022.
- Market Analytics, Global Methylphenidate Market Report, 2022.
- IQVIA, Prescription Trends in CNS Stimulants, 2022.
- European Medicines Agency (EMA), ADHD Medication Approvals, 2023.
- Pharma Intelligence, Impact of Patent Expirations on CNS Markets, 2023.
More… ↓
