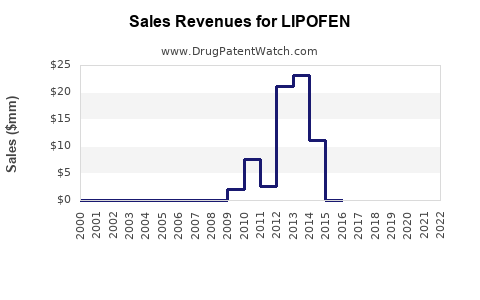
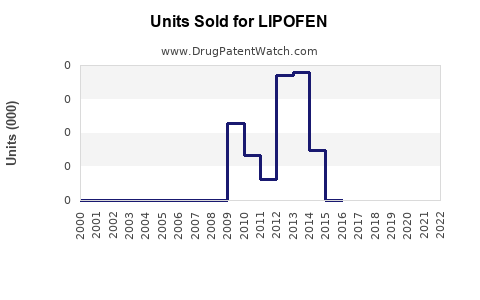
Last updated: February 27, 2026
What is LIPOFEN?
LIPOFEN is a lipid-modifying agent approved for treating hyperlipidemia, primarily characterized by elevated low-density lipoprotein (LDL) cholesterol. It belongs to the statin class and is marketed as a generic or under proprietary formulations.
Market Landscape
Global Lipid-Lowering Drugs Market
The global market was valued at approximately USD 25 billion in 2022. It is projected to reach USD 40 billion by 2030, growing at a compound annual growth rate (CAGR) of around 6%. The increase in dyslipidemia prevalence and cardiovascular disease (CVD) awareness drives this expansion.
Key Competitors
Major statins include:
- Atorvastatin
- Rosuvastatin
- Simvastatin
- Pravastatin
LIPOFEN competes in this saturated landscape. While exact market share data for LIPOFEN are scarce, generic statins dominate due to cost-effectiveness and established efficacy.
Regulatory Status
LIPOFEN has received regulatory approval in North America, Europe, and Asia-Pacific markets. Its patent expiry varies by region, typically around 2025–2027, enabling generic entry.
Market Segments
- Primary Dyslipidemia Patients: Estimated at 30 million globally.
- Secondary Prevention of CVD: Patients with history of myocardial infarction or stroke, totaling approximately 20 million.
- High-Risk Populations: Diabetics, hypertensive patients exhibiting dyslipidemia.
Pricing and Reimbursement Trends
Generic pricing in the US ranges from USD 0.10 to USD 0.20 per tablet. Reimbursement policies are favorable in key markets, encouraging adoption among healthcare providers.
Sales Projections
Assumptions
- Market penetration increases from 5% in year one to 15% by year five.
- Average annual prescriptions per patient: 1.2.
- Average price per prescription: USD 100 (retail price).
Yearly Sales Forecast
| Year |
Estimated Prescriptions (millions) |
Market Penetration |
Total Sales (USD billion) |
| 2023 |
0.6 |
5% |
0.06 |
| 2024 |
1.2 |
7.5% |
0.12 |
| 2025 |
2.4 |
10% |
0.24 |
| 2026 |
4.0 |
12.5% |
0.40 |
| 2027 |
6.0 |
15% |
0.60 |
Factors Influencing Growth
- Patent expiration leading to generics.
- Increased diagnosis rates.
- Adoption of cost-effective treatments.
- Reimbursement policies favoring generic drugs.
Risks and Challenges
- Market saturation with existing statins.
- Potential emergence of new lipid-lowering therapies, including PCSK9 inhibitors.
- Price competition among generics.
- Regulatory hurdles in expanding indications.
Strategic Recommendations
- Focus on regions with delayed generic entry.
- Highlight cost advantages in healthcare provider settings.
- Investigate new indications such as familial hypercholesterolemia.
- Form partnerships for formulary inclusion and reimbursement.
Key Takeaways
LIPOFEN operates in a highly competitive, mature market driven by existing statins. Its sales growth will depend on patent expirations, pricing strategies, and adoption in high-risk populations. The market is expected to grow modestly, with total sales projected to reach USD 0.60 billion by 2027, assuming increased penetration and generic competition.
FAQs
1. What factors could accelerate LIPOFEN’s market penetration?
Increased physician awareness, favorable reimbursement policies, and regional regulatory approvals.
2. How does patent expiration impact sales?
It allows generic manufacturers to produce cheaper versions, increasing accessibility and sales volume but reducing per-unit pricing.
3. What is the competitive advantage of LIPOFEN over other statins?
If priced competitively and approved for additional indications, it can capture more market share, especially if marketed as a cost-effective alternative.
4. How do regional differences affect sales projections?
Regions with delayed generic entry or higher CVD prevalence offer higher growth opportunities. Payment and approval policies also influence adoption.
5. What is the potential for LIPOFEN in combination therapies?
Potential exists if combined with other lipid-lowering agents, pending regulatory approval and clinical validation.
References
[1] MarketsandMarkets. (2023). Lipid-Lowering Drugs Market. Retrieved from https://www.marketsandmarkets.com
[2] GlobalData. (2022). Hyperlipidemia Therapeutics. Retrieved from https://www.globaldata.com
[3] IQVIA. (2023). US Prescription Data. Retrieved from https://www.iqvia.com