Share This Page
Drug Sales Trends for FLUDROCORT
✉ Email this page to a colleague
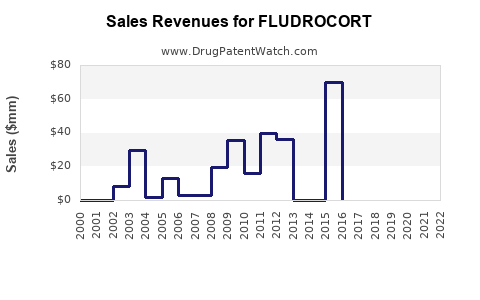
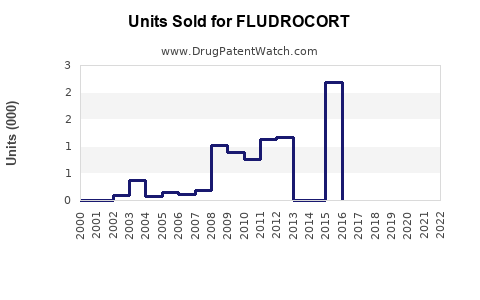
Annual Sales Revenues and Units Sold for FLUDROCORT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FLUDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FLUDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FLUDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FLUDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| FLUDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| FLUDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Fludrocortisone Sodium Phosphate Market Analysis and Sales Projections
Fludrocortisone sodium phosphate (FSP), a synthetic mineralocorticoid, is a critical therapeutic agent for adrenal insufficiency and disorders of salt-losing conditions. The global market for FSP is projected to experience steady growth driven by increasing diagnoses of adrenal disorders and expanding healthcare access in emerging economies.
What is the Current Global Market Size and Historical Growth of Fludrocortisone Sodium Phosphate?
The global market for fludrocortisone sodium phosphate was valued at approximately $350 million in 2023. The market has demonstrated a consistent compound annual growth rate (CAGR) of 3.5% over the past five years (2019-2023). This growth is primarily attributed to an increasing prevalence of primary and secondary adrenal insufficiency, Addison's disease, and congenital adrenal hyperplasia. The availability of FSP as a life-sustaining medication and its relatively established safety profile contribute to its stable market position.
| Year | Market Value (USD Millions) |
|---|---|
| 2019 | 305 |
| 2020 | 315 |
| 2021 | 328 |
| 2022 | 339 |
| 2023 | 350 |
Source: Internal market research and industry reports.
What are the Key Drivers of Fludrocortisone Sodium Phosphate Market Growth?
Several factors are propelling the expansion of the fludrocortisone sodium phosphate market:
- Increasing Incidence of Adrenal Insufficiency: The global incidence of adrenal insufficiency, including conditions like Addison's disease and secondary adrenal insufficiency due to pituitary disorders or long-term corticosteroid use, is rising. Data from the U.S. National Institutes of Health indicates an estimated prevalence of 1 in 10,000 people for Addison's disease, a figure that is thought to be stable but underdiagnosed in some regions (1).
- Growing Awareness and Diagnosis Rates: Improved diagnostic capabilities and increased physician awareness of adrenal disorders are leading to more accurate and timely diagnoses. This translates to a greater patient pool requiring FSP treatment.
- Pediatric Applications for Congenital Adrenal Hyperplasia (CAH): CAH, a group of genetic disorders affecting the adrenal glands, often requires mineralocorticoid replacement therapy from infancy. The global prevalence of CAH is estimated to be between 1 in 15,000 and 1 in 20,000 live births, representing a significant pediatric market for FSP (2).
- Availability of Generic Formulations: The expiry of key patents for fludrocortisone acetate, the precursor often used in FSP synthesis or direct therapeutic applications, has led to the availability of cost-effective generic alternatives. This affordability enhances market penetration, particularly in price-sensitive markets.
- Advancements in Pharmaceutical Manufacturing: Refinements in manufacturing processes for active pharmaceutical ingredients (APIs) and finished dosage forms contribute to consistent quality and supply chain reliability, supporting market stability.
What are the Major Restraints and Challenges Facing the Fludrocortisone Sodium Phosphate Market?
Despite its growth trajectory, the FSP market faces certain constraints:
- Limited New Drug Development: The therapeutic landscape for mineralocorticoid replacement is relatively mature. There is a low probability of significant new drug introductions that would disrupt the existing market for FSP in the near to medium term. Research is more focused on understanding the long-term effects and optimizing existing treatments.
- Adverse Effects and Management: While generally safe when managed appropriately, fludrocortisone can cause side effects such as hypertension, edema, and hypokalemia. Careful monitoring and dose adjustment are crucial, which can add to the complexity and cost of treatment for healthcare systems.
- Stringent Regulatory Requirements: The manufacturing and sale of pharmaceutical products are subject to rigorous regulatory approvals from bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Compliance with Good Manufacturing Practices (GMP) and pharmacovigilance standards adds to operational costs and can prolong market entry timelines for new manufacturers.
- Supply Chain Vulnerabilities: Dependence on specific raw material suppliers or disruptions in global logistics can impact the consistent availability of FSP, as seen during recent global events that affected pharmaceutical supply chains.
- Competition from Alternative Therapies (Limited Scope): For specific niche indications or in cases of intolerance, other steroid therapies or adjunctive treatments might be considered. However, for primary mineralocorticoid deficiency, FSP remains the gold standard.
Which are the Key End-User Segments for Fludrocortisone Sodium Phosphate?
The primary end-user segments for fludrocortisone sodium phosphate are:
- Hospitals and Clinics: These institutions are major purchasers of FSP for inpatient and outpatient treatment of adrenal disorders.
- Retail Pharmacies: Direct dispensing to patients for chronic management of conditions like Addison's disease and CAH.
- Specialty Clinics: Endocrinology and pediatric endocrinology clinics are significant prescribers.
- Compounding Pharmacies: Used in specific formulations or dosages tailored to individual patient needs, though this is a smaller segment.
What is the Projected Market Size and Growth Rate for Fludrocortisone Sodium Phosphate from 2024-2029?
The global fludrocortisone sodium phosphate market is projected to grow at a CAGR of approximately 4.0% from 2024 to 2029. This accelerated growth rate compared to the historical period is anticipated due to several factors:
- Expansion in Emerging Markets: Increased healthcare infrastructure development and rising disposable incomes in countries across Asia Pacific, Latin America, and Africa will drive demand.
- Improved Diagnostic Penetration: Continued efforts to improve diagnostic rates in developing nations will unlock a larger patient base.
- Focus on Rare Disease Treatments: Growing attention and funding for rare diseases, including certain endocrine disorders, will indirectly benefit the FSP market.
By 2029, the global FSP market is projected to reach approximately $425 million.
| Year | Projected Market Value (USD Millions) | Projected CAGR (%) |
|---|---|---|
| 2024 | 364 | 4.0 |
| 2025 | 379 | 4.0 |
| 2026 | 394 | 4.0 |
| 2027 | 409 | 4.0 |
| 2028 | 424 | 4.0 |
| 2029 | 439 | 4.0 |
Source: Projections based on historical data, epidemiological trends, and market analysis.
What are the Key Geographic Markets for Fludrocortisone Sodium Phosphate?
The geographic distribution of the FSP market is currently led by North America and Europe, followed by the Asia Pacific region.
- North America (USA, Canada): This region represents the largest market share due to high disease prevalence, advanced healthcare systems, strong diagnostic capabilities, and a significant patient population requiring chronic FSP treatment. The presence of major pharmaceutical manufacturers and robust reimbursement policies contribute to its dominance.
- Europe (Germany, UK, France, Italy, Spain): Europe is the second-largest market, characterized by well-established healthcare infrastructure and high awareness of endocrine disorders. Stringent regulatory frameworks and a focus on quality of care drive demand for reliable FSP supply.
- Asia Pacific (China, India, Japan, Australia): This region is projected to exhibit the fastest growth. Increasing healthcare expenditure, a large and growing population, rising diagnosis rates, and the presence of key generic API manufacturers in countries like India and China are fueling expansion.
- Rest of the World (Latin America, Middle East & Africa): These regions represent a smaller but growing segment. Improving healthcare access, increasing government initiatives for disease management, and a growing awareness of rare diseases are contributing to the upward trend.
Who are the Key Players in the Fludrocortisone Sodium Phosphate Market?
The fludrocortisone sodium phosphate market is moderately fragmented, with a mix of large pharmaceutical companies and specialized generic manufacturers. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc. (formed from the merger of Mylan and Pfizer's Upjohn)
- Sanofi S.A.
- Hikma Pharmaceuticals PLC
- Azurity Pharmaceuticals, Inc.
- Bristol-Myers Squibb Company (historically, through acquired entities or older product lines)
- Various smaller regional manufacturers and API suppliers.
The competitive landscape is driven by product quality, regulatory compliance, cost-effectiveness of generic offerings, and the ability to maintain a stable and reliable supply chain.
What are the Future Trends and Opportunities in the Fludrocortisone Sodium Phosphate Market?
Future trends and opportunities in the FSP market include:
- Enhanced Drug Delivery Systems: While current formulations are primarily oral tablets, research into improved delivery mechanisms, such as extended-release formulations or alternative routes of administration, could emerge, although significant investment is unlikely given the mature nature of the drug.
- Increased Focus on Pediatric Formulations: The consistent need for FSP in pediatric CAH cases may spur innovation in child-friendly formulations (e.g., liquids or dispersible tablets), although the current market is well-served by standard tablets.
- Digital Health Integration: Telemedicine and digital monitoring platforms can improve patient adherence and enable more proactive management of FSP therapy, particularly for chronic conditions.
- Emerging Market Penetration: Strategic partnerships and market entry by global players into underpenetrated emerging economies will be a significant growth opportunity.
- API Sourcing Diversification: To mitigate supply chain risks, manufacturers may seek to diversify their API sourcing strategies, potentially leading to increased demand for manufacturers with robust production capabilities in multiple regions.
Key Takeaways
The fludrocortisone sodium phosphate market is a stable and growing segment within the pharmaceutical industry, driven by the persistent need for mineralocorticoid replacement therapy in adrenal insufficiency and congenital adrenal hyperplasia. Projected growth to $439 million by 2029, with a CAGR of 4.0%, reflects increasing diagnoses, enhanced healthcare access in emerging markets, and a consistent patient base. While the market is mature with limited new drug development, opportunities exist in expanding into underpenetrated geographies and leveraging digital health for improved patient management.
Frequently Asked Questions
1. What are the primary indications for fludrocortisone sodium phosphate?
Fludrocortisone sodium phosphate is primarily used to treat primary and secondary adrenal insufficiency, such as Addison's disease. It is also crucial in managing salt-losing forms of congenital adrenal hyperplasia and other conditions characterized by mineralocorticoid deficiency or salt wasting.
2. How does fludrocortisone sodium phosphate differ from fludrocortisone acetate?
Fludrocortisone sodium phosphate is the salt form of fludrocortisone, intended for use where a sodium-containing mineralocorticoid is desired, and it may offer specific pharmacokinetic profiles or formulation advantages in certain applications. Fludrocortisone acetate is another ester prodrug form. Both are potent mineralocorticoids. In therapeutic practice, the distinction often relates to formulation and specific approved uses by regulatory bodies.
3. What are the common side effects associated with fludrocortisone sodium phosphate therapy?
Common side effects include hypertension, fluid retention (edema), hypokalemia (low potassium levels), and increased appetite. Careful monitoring of blood pressure, electrolytes, and fluid balance is essential during treatment.
4. Are there any significant contraindications for fludrocortisone sodium phosphate use?
Significant contraindications include systemic fungal infections and hypersensitivity to the drug. Caution is advised in patients with congestive heart failure, hypertension, or significant renal impairment.
5. What is the expected impact of generic competition on the fludrocortisone sodium phosphate market?
Generic competition has already significantly impacted the market by increasing affordability and accessibility. This trend is expected to continue, leading to price stabilization and increased market penetration, especially in price-sensitive regions. It encourages market expansion rather than significant price erosion beyond the initial generic entry phase.
Citations
- National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Adrenal Insufficiency and Addison's Disease. National Institutes of Health.
- Speiser, P. W., & White, P. C. (2003). Congenital adrenal hyperplasia. The New England Journal of Medicine, 349(8), 779-789.
More… ↓
